Reactions of Ions in Aqueous Solution with Bases
Lajoy Tucker
Teacher

Contents
Introduction
When a metal ion, , dissolves in water, it forms a metal aqua ion. The water molecules coordinate with the metal ion, creating an aqueous complex such as:
Hexaaquacopper(II):
Hexaaquaaluminum(III):
These metal aqua ions are often acidic in solution. This is because the metal ion can polarize the water molecules, making them more likely to lose a proton , resulting in acidic behaviour.
Acid Base Reaction Questions Answered
Reactions of Metal Aqua Ions with Bases
If a base is added to a complex metal ion, the base removes (deprotonates) the until a NEUTRAL INSOLUBLE complex is formed.
The neutral complex will form as a precipitate.
With the condition of an excess of the base, the insoluble complex could react further (or not!).
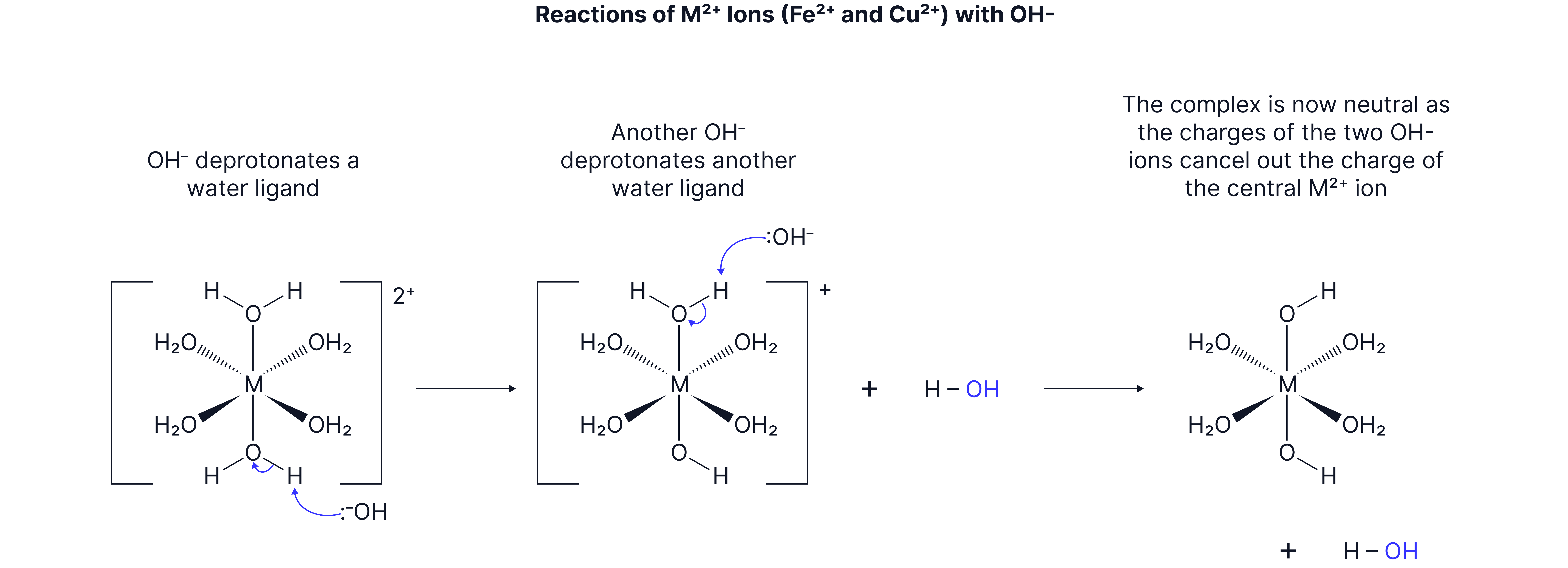
Overall:
This is not a ligand exchange reaction. The water at the end is produced by the ions accepting the from the water ligands.
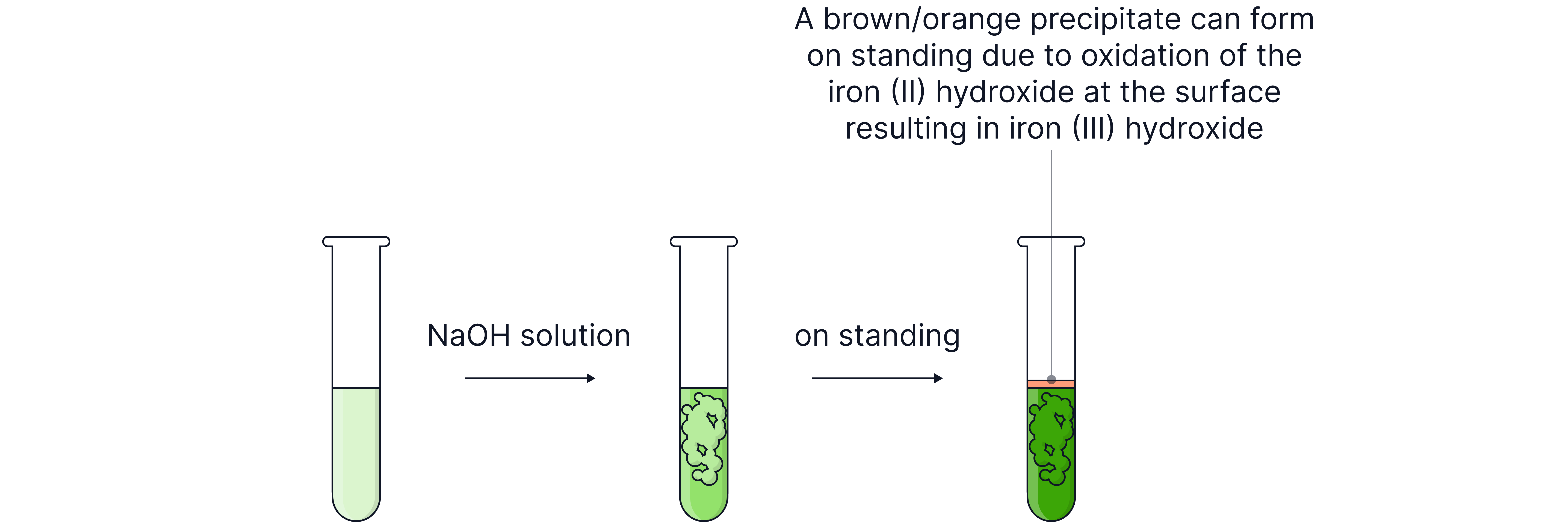
Iron(II)
Iron(II) ions react with alkali to form iron(II) hydroxide, a green precipitate.
Pale green solution:
Pale green precipitate:
Copper (II)
Copper(II) ions react with alkali to form copper(II) hydroxide, a blue precipitate.
Blue solution:
Blue precipitate:
Reactions of Ions ( and ) with
The same principle as above can be applied to metals with a charge. This time, to produce the neutral insoluble complex, three hydroxide ions are required to deprotonate three water ligands.
Overall:
Iron(III)
Iron(III) ions react with alkali to form iron(III) hydroxide, a red-brown precipitate.
Yellow-orange solution:
Brown precipitate:
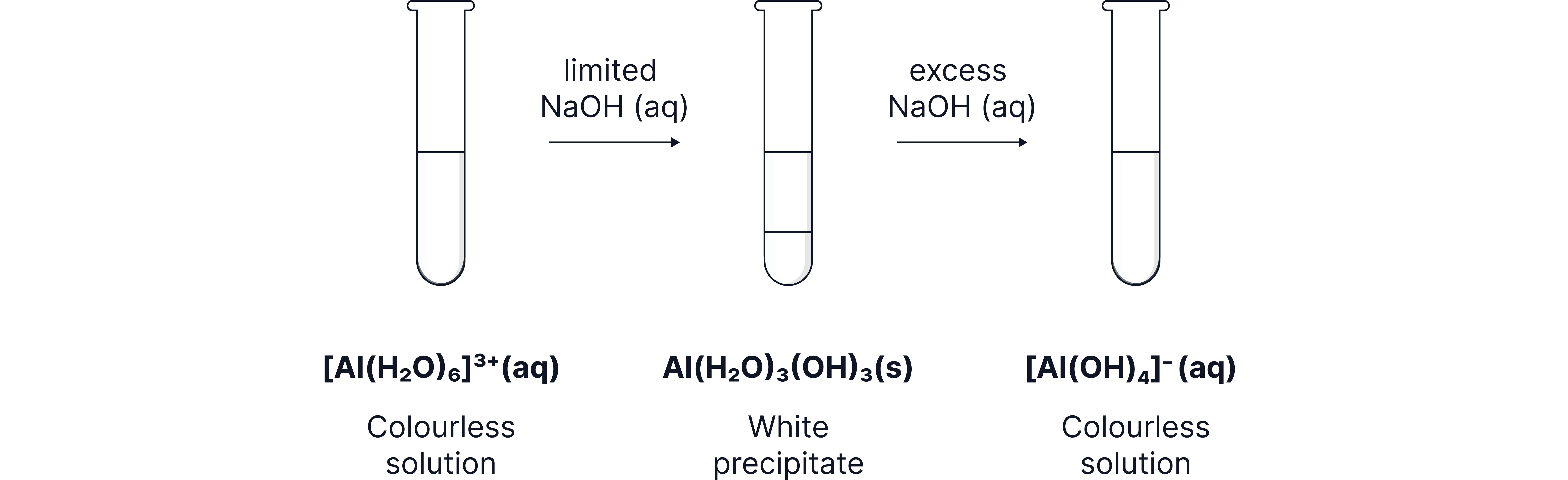
Aluminum(III)
Aluminum ions react with alkali to form aluminum hydroxide, a white precipitate.
Colourless solution:
White precipitate:
The aluminum hydroxide, is amphoteric meaning it can react with both acids and bases.
Reaction with acid produces the original metal-aqua ion.
Reaction with bases (excess ) causes the precipitate to dissolve to form a colourless solution.
White precipitate:
Colourless solution:
Reactions with NH₃ (ammonia)
Ammonia is a weak base. When added dropwise, this forms the neutral insoluble complex and ammonium ions in an acid-base reaction.
Iron(II)
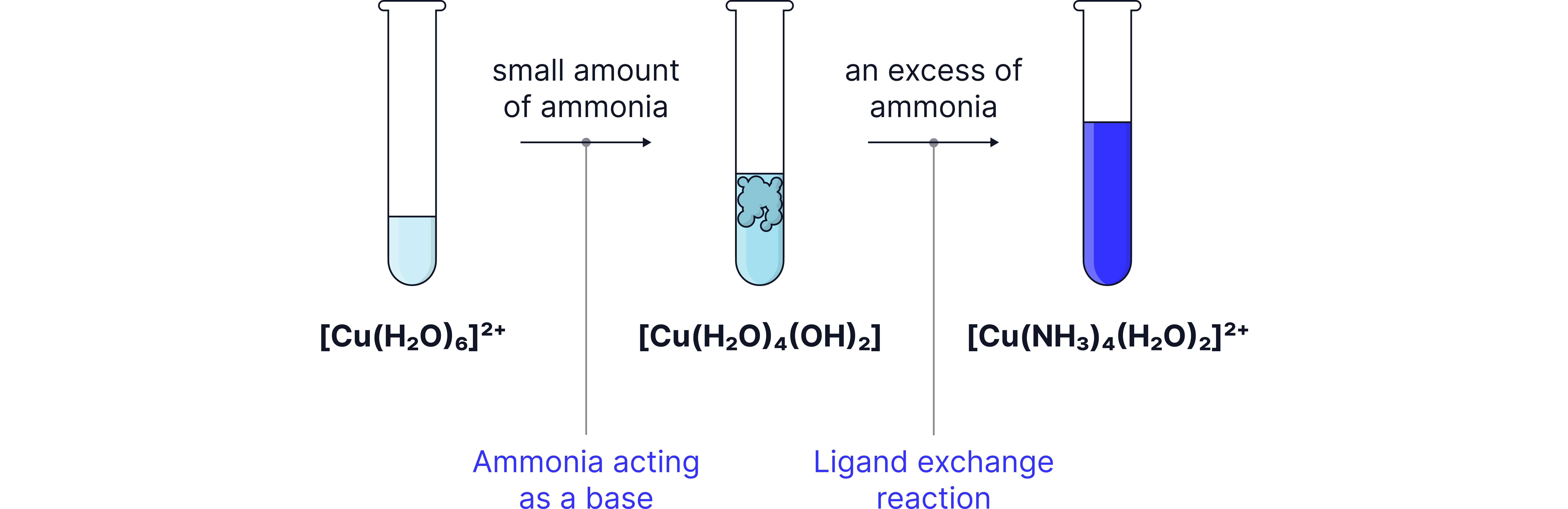
Copper(II)
Like hydroxide ions, ammonia causes the formation of Copper(II) hydroxide.
In excess ammonia reacts further where the ammonia partially substitute the water ligands producing a deep blue solution containing ions.
Blue precipitate:
Deep blue solution:
Iron(III)
Similar to hydroxide ions, ammonia leads to the formation of Iron(III) hydroxide.
Aluminum(III)
Ammonia causes the precipitation of aluminum hydroxide.
Reactions with (carbonate)
ions and
ions are not acidic enough for an acid-base reaction with carbonate ions. Instead, the solid metal (II) carbonate forms as a precipitate.
ions and
ions are more acidic in water due to their greater charge densities. These ions therefore polarise carbonate ions sufficiently to release carbon dioxide. The overall reaction is of the form acid + carbonate neutral metal complex + water + carbon dioxide.
Summary Table
Ion | Colour of aq ion | OH⁻ | NH₃ | CO₃²⁻ |
Fe²⁺ | Pale green | Green precipitate, insoluble excess, slowly turns orange-brown at the surface | Green precipitate, insoluble excess, slowly turns orange-brown at the surface | Green precipitate |
Cu²⁺ | Pale blue | Light blue precipitate, insoluble in excess | Light blue precipitate, deep blue solution when excess added | Blue-green precipitate |
Al³⁺ | Colourless | White precipitate, redissolves in excess to give a colourless solution | White precipitate, insoluble in excess | White precipitate with some bubbles |
Fe³⁺ | Pale orange | Brown precipitate, insoluble in excess | Brown precipitate, insoluble in excess | Red-brown precipitate with some bubbles |
Exam Tips
Make sure to mention colour changes when discussing the formation of precipitates.
Remember, aluminum hydroxide is amphoteric, and can dissolve in excess .
Be able to identify precipitate colours for iron(II), copper(II), aluminum(III), and iron(III) complexes.
Understand the charge/size ratio of metal ions, and how this affects acidity and ligand interactions.