Nomenclature
Lajoy Tucker
Teacher

Contents
Systematic Nomenclature - Building Understanding from Principles
The Logic Behind IUPAC Naming
Why do we need systematic nomenclature? Common names like "acetone" or "aspirin" tell us nothing about structure. IUPAC names encode the molecular architecture directly into the name, allowing chemists worldwide to communicate precisely.
Core Principle: Every IUPAC name follows the pattern: [Locants] + [Prefix] + [Parent chain] + [Suffix]
What do these terms mean?
Locants: Numbers that show WHERE things are (e.g., the "2" in "2- methylbutane").
Prefix: Groups that aren't the main functional group (e.g., "methyl-", "chloro-").
Parent chain: The longest carbon chain that contains the main functional group.
Suffix: The ending that shows the main functional group (e.g., "-ol" for alcohol, "- one" for ketone).
Step-by-Step Naming Method
1. Find the longest carbon chain containing the functional group.
2. Number the chain to give the functional group(s) the lowest numbers.
3. Name and number all substituents, arrange alphabetically.
Key Rules:
Numbers are separated by commas (2,3-dimethyl).
Numbers and letters are separated by dashes (2-methyl).
When identical groups appear, use di-, tri-, tetra- prefixes.
Alphabetise substituents (ignore di-, tri-, tetra- when alphabetising).
Carbon Chain Stems
|
Carbons |
1C |
2C |
3C |
4C |
5C |
6C |
|
Stem |
meth |
eth |
prop |
but |
pent |
hex |
Functional Group Priority - The Hierarchy of Importance
When multiple functional groups are present, only ONE gets the suffix - the highest priority group. All others become prefixes.
Functional Group Priority Table (from highest to lowest)
|
Priority
|
Functional Group
|
Suffix
|
Prefix
|
Example Structure
|
|---|---|---|---|---|
| 1 | Carboxylic acids | -oic acid | carboxy- | 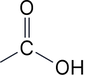 |
| 2a | Esters | -yl -oate | alkoxycarbonyl- | 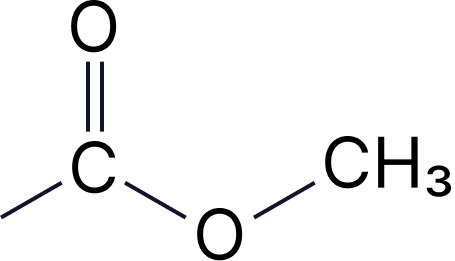 |
| 2b | Acyl chlorides | -oyl chloride | chloroformyl- | 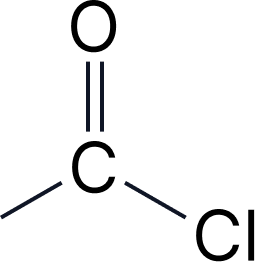 |
| 2c | Amides | -amide | carbamoyl- | 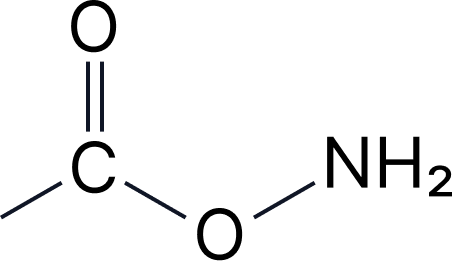 |
| 2d | Acid anhydrides | -oic anhydride | – | 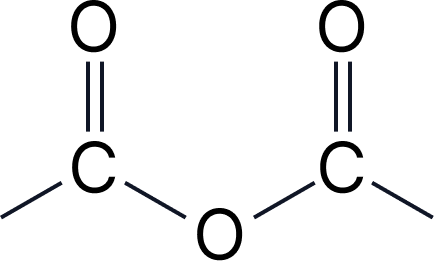 |
| 3 | Nitriles | -nitrile | cyano- | |
| 4 | Aldehydes | -al | formyl- | 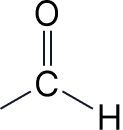 |
| 5 | Ketones | -one | oxo- | 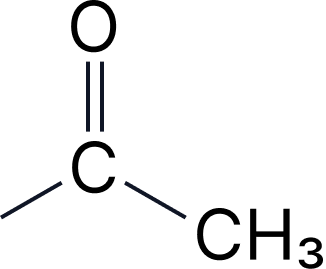 |
| 6 | Alcohols | -ol | hydroxy- | |
| 7 | Amines | -amine | amino- | |
| 8 | Alkenes | -ene | alkenyl- | 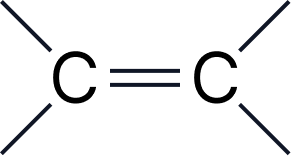 |
| 9 | Halogenoalkanes | – | fluoro-, chloro-, bromo-, iodo- |
Key Rule: The highest priority group gets the suffix and determines chain numbering. All others become prefixes.
Detailed Functional Group Naming
Alkanes
• Suffix: -ane
• Only contain C-H bonds
• Example: CH₃-CH₂-CH₃ = propane
Complex Example: 2-methylpentane
• Longest chain = 5 carbons (pentane)
• Methyl substituent on carbon-2
Alkenes
• Suffix: -ene (needs position number for chains ≥4C)
• Contains C=C double bond
• Example: CH₃-CH₂-CH=CH₂ = but-1-ene
Key Point: Number from end giving C=C the lowest number
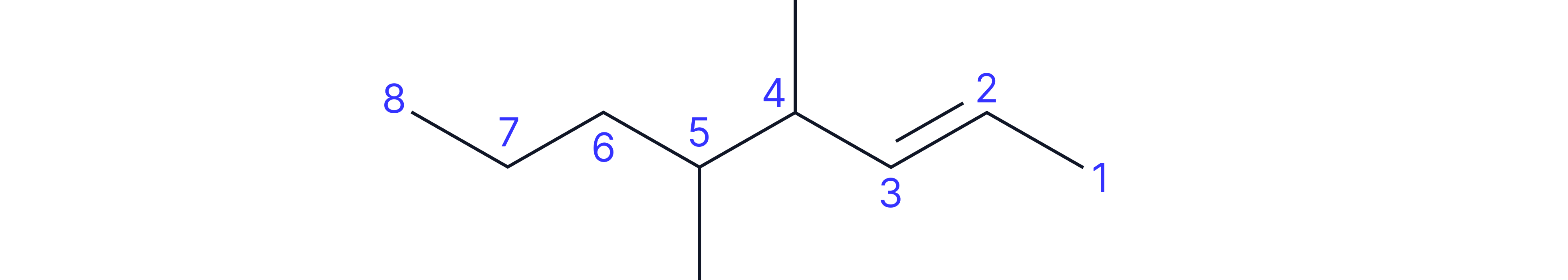
Complex Example: 4,5-dimethyloct-2-ene
• Longest chain containing C=C: 8 carbons (octene)
• Double bond between carbons 2 and 3 (so "2-ene")
• Two methyl substituents on carbons 4 and 5
• Number from end giving double bond lowest number
Halogenoalkanes
• Prefixes: fluoro-, chloro-, bromo-, iodo-
• Always prefixes, never suffixes
• Example: CH₃-CHBr-CH₃ = 2-bromopropane
Simple Examples:
• CH₃-CH₂-I = iodoethane (no number needed - only one position possible)
• CH₃-CHCl-CH₃ = 2-chloropropane
• CH₃-CH₂-CH₂-F = 1-fluoropropane
Multiple Halogens:
• CCl₂H-CH₃ = 1,1-dichloroethane (both chlorines on same carbon)
• CH₂F-CH₂Br = 1-bromo-2-fluoroethane (alphabetical order: bromo before fluoro)
• CH₃-CHBr-CHCl-CH₃ = 2-bromo-3-chlorobutane (alphabetical order: bromo
before chloro)
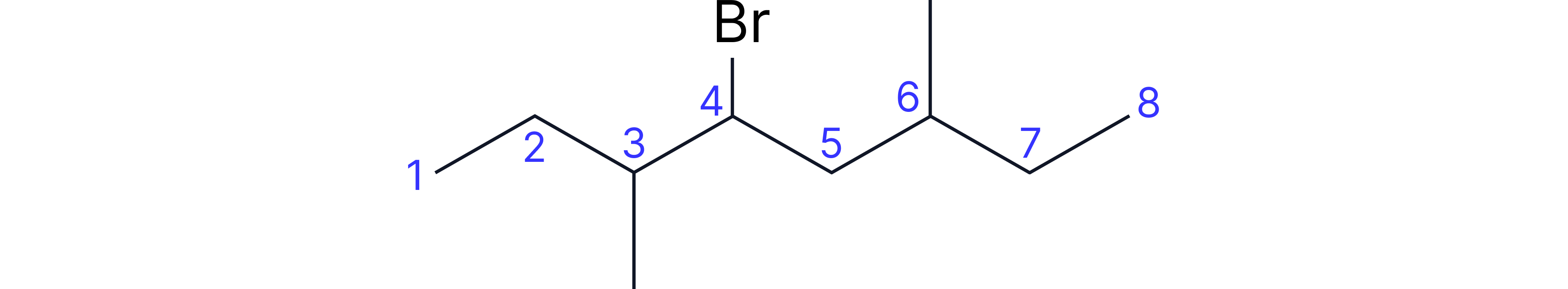
Complex Example: 4-bromo-3,6-dimethyloctane
• Longest chain: 8 carbons (octane)
• Substituents: bromo (carbon-4), methyl (carbon-3), methyl (carbon-6)
• Alphabetical order: bromo, methyl
• Number to minimise ALL substituent positions
Alcohols
• Suffix: -ol (needs position number)
• Prefix: hydroxy-
• Example: CH₃-CH₂-CH₂-OH = propan-1-ol
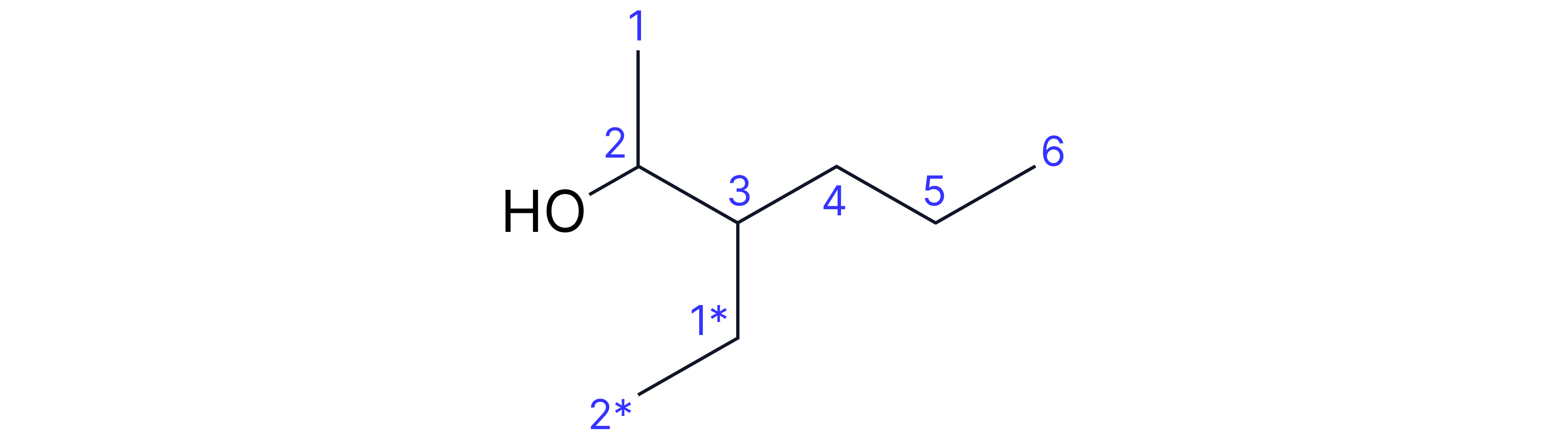
Complex Example: 3-ethylhexan-2-ol
• Longest chain containing -OH: 6 carbons (hexanol)
• Alcohol group on carbon-2 (so "2-ol")
• Ethyl substituent on carbon-3
• Number from end giving alcohol lowest number
Aldehydes
• Suffix: -al
• Prefix: formyl-
• Key rule: Aldehyde carbon is ALWAYS carbon-1
Challenge: How would you name a compound with both aldehyde and carboxylic acid groups? Answer: The carboxylic acid takes priority, aldehyde becomes "formyl-"
Complex Example: 4-chloro-2-methylheptanal
• Longest chain containing -CHO: 6 carbons (hexanal)
• Aldehyde automatically on carbon-1
• Methyl substituent on carbon-2
• Chloro substituent on carbon-4
• Alphabetical order: chloro, then methyl
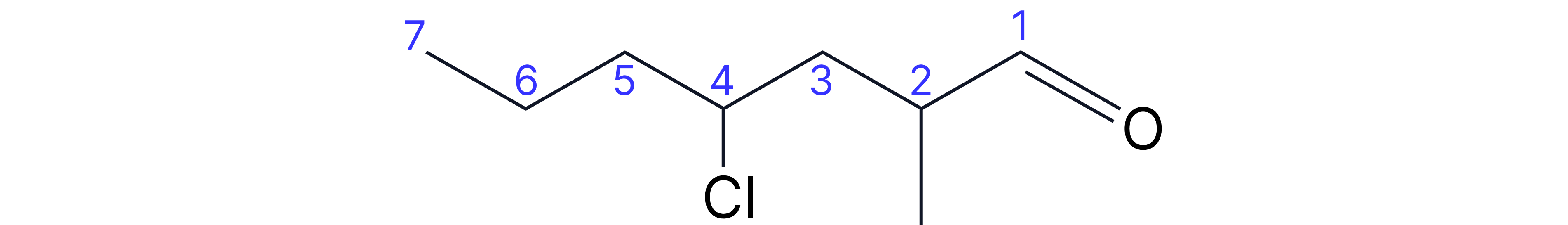
Ketones
• Suffix: -one (needs position number for chains ≥5C)
• Prefix: oxo
Complex Example: 5-bromo-3-methylheptan-2-one
• Longest chain containing C=O: 7 carbons (heptanone)
• Ketone on carbon-2 (so "2-one")
• Methyl substituent on carbon-3
• Bromo substituent on carbon-5
• Number from end giving ketone lowest number
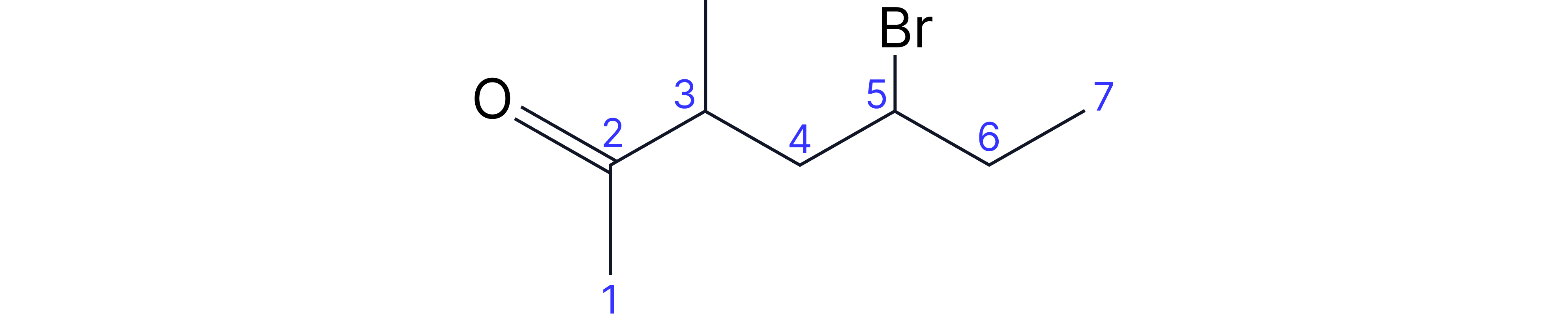
Carboxylic Acids
• Suffix: -oic acid
• Key rule: Carboxylic acid carbon is ALWAYS carbon-1
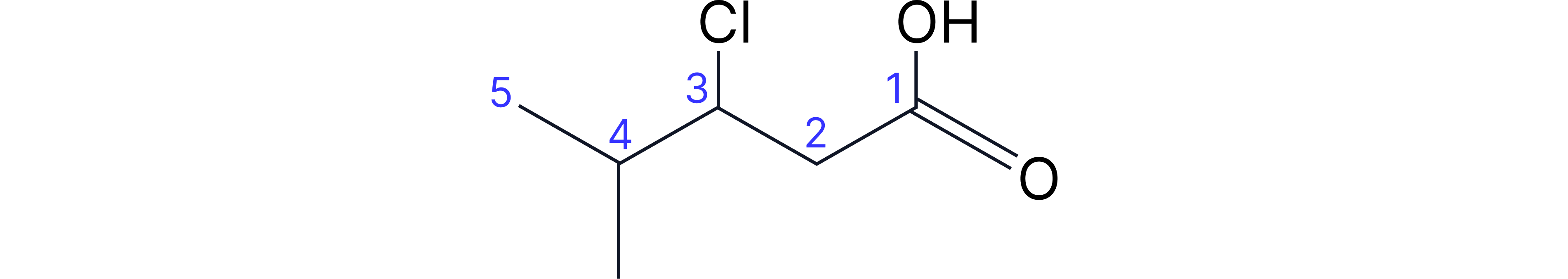
Complex Example: 3-chloro-4-methylpentanoic acid
• Longest chain containing -COOH: 5 carbons (pentanoic acid)
• Carboxylic acid automatically on carbon-1
• Methyl substituent on carbon-4
• Chloro substituent on carbon-3
• Alphabetical order: chloro, then methyl
Nitriles
• Suffix: -nitrile
• Critical point: The cyano carbon (C≡N) counts as carbon-1

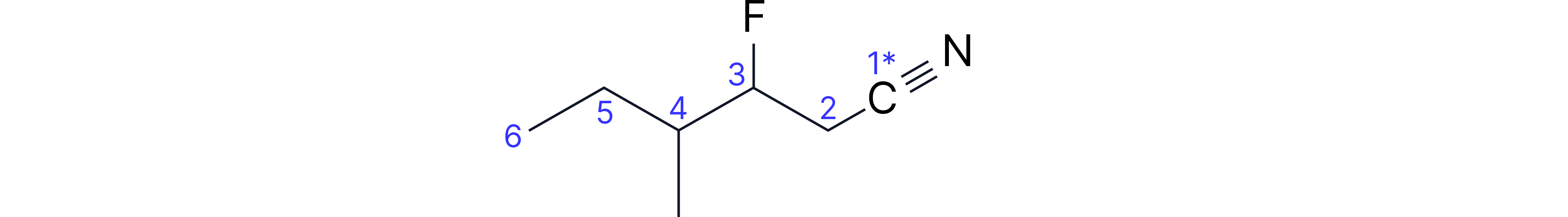
Complex Example: 3-fluoro-4-methylhexanenitrile
• Longest chain containing C≡N: 6 carbons (hexanenitrile)
• Nitrile carbon automatically carbon-1
• Methyl substituent on carbon-4 (from nitrile end)
• Fluoro substituent on carbon-3
• Alphabetical order: fluoro, then methyl
Amines - The Dual System Challenge
Two naming systems exist:
System 1 (Common): -alkylamine

System 2 (IUPAC): -alkan-N-amine

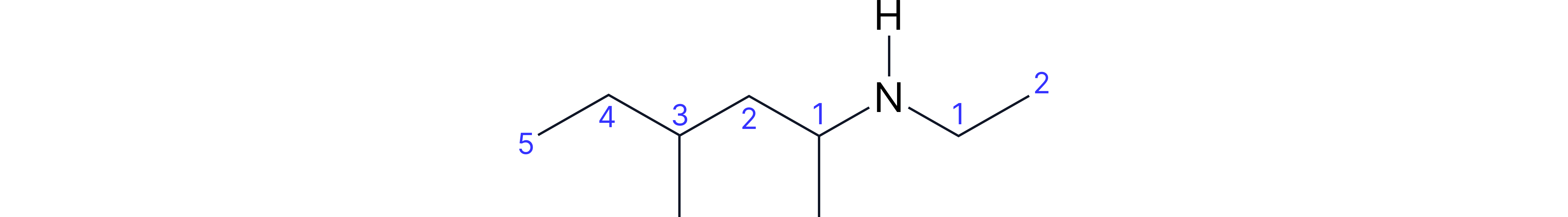
Complex Example: N-ethyl-1,3-dimethylpentan-1-amine
• Longest chain containing NH: 5 carbons (pentanamine)
• Amine on carbon-1 (so "1-amine")
• Methyl substituent on carbon-1 and carbon-3
• Ethyl substituent on nitrogen (so "N-ethyl")
• Order: N-ethyl comes first, then 1,3-dimethyl
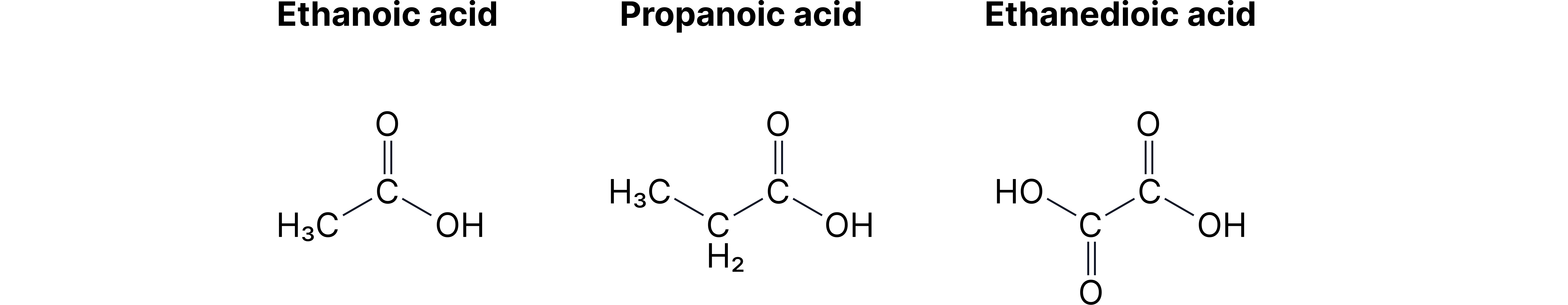
Carboxylic Acid Derivatives
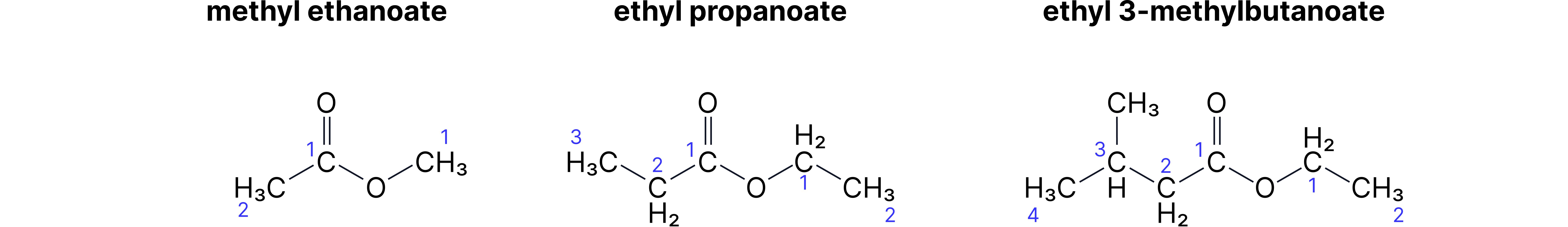
Esters - Two-Part Names
Pattern: [alcohol part]-yl [acid part]-oate
• CH₃-COO-CH₂-CH₃ = ethyl ethanoate
Acyl Chlorides
Pattern: [stem]-oyl chloride

Amides
Primary: [stem]-amide
• CH₃-CONH₂ = ethanamide
Secondary/Tertiary: Use N- prefix
• CH₃-CONH-CH₃ = N-methylethanamide
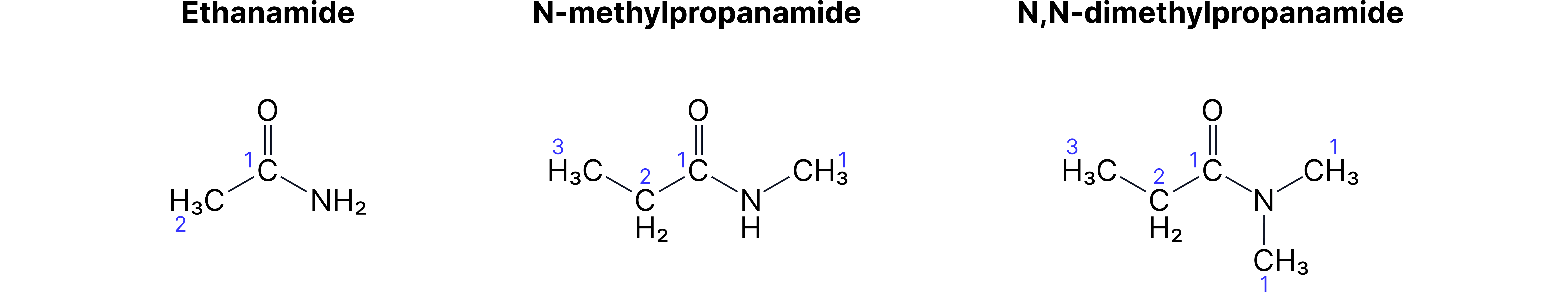
Complex Molecules - Multiple Functional Groups
Priority System in Action
Example: HOOC-CH₂-CHO
1. Identify groups: carboxylic acid (priority 1), aldehyde (priority7)
2. Carboxylic acid gets suffix: -oic acid
3. Aldehyde becomes prefix: formyl
4. Name: formylethanoic acid
Multiple Identical Groups
• Two groups: di- (dimethyl, diol, dione)
• Three groups: tri-
• Four groups: tetra
Examples:
• CH₂OH-CH₂OH = ethane-1,2-diol
Isomerism - Understanding Molecular Relationships
Structural Isomerism Types
Chain Isomerism
Definition: Different carbon skeleton arrangements
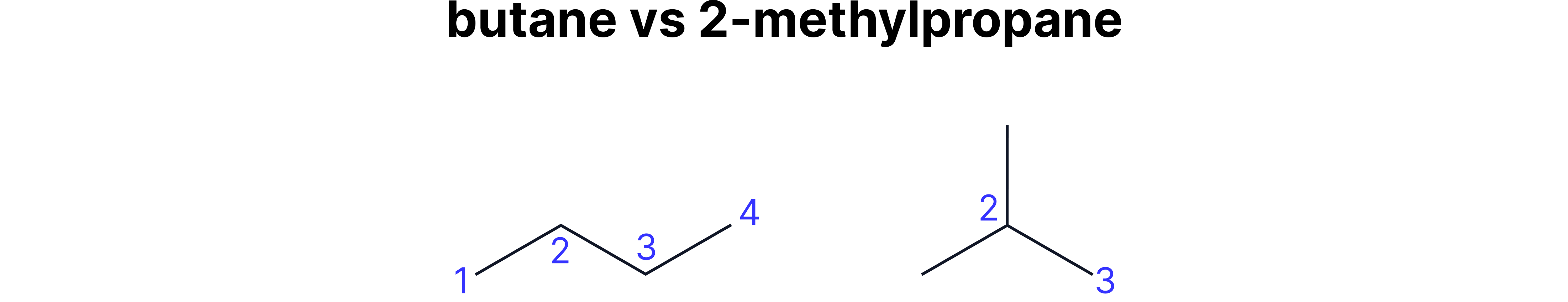
Position Isomerism
Definition: Same functional groups, different positions Simple explanation: It's like having the same furniture in a room, but arranged differently
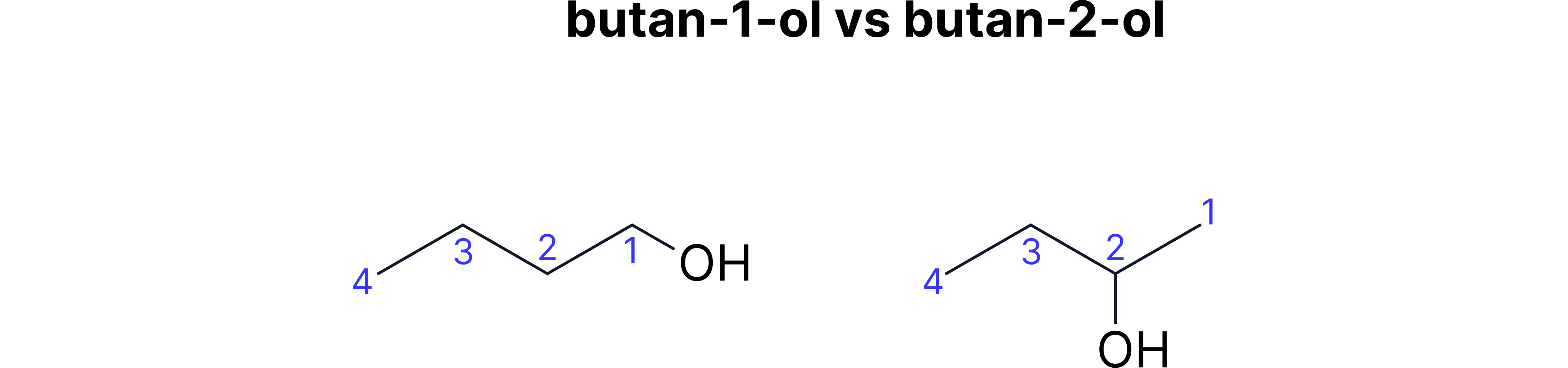
Easy way to remember: The number in the name tells you WHERE the functional group is
Functional Group Isomerism
Definition: Same molecular formula, different functional groups
Key Examples:
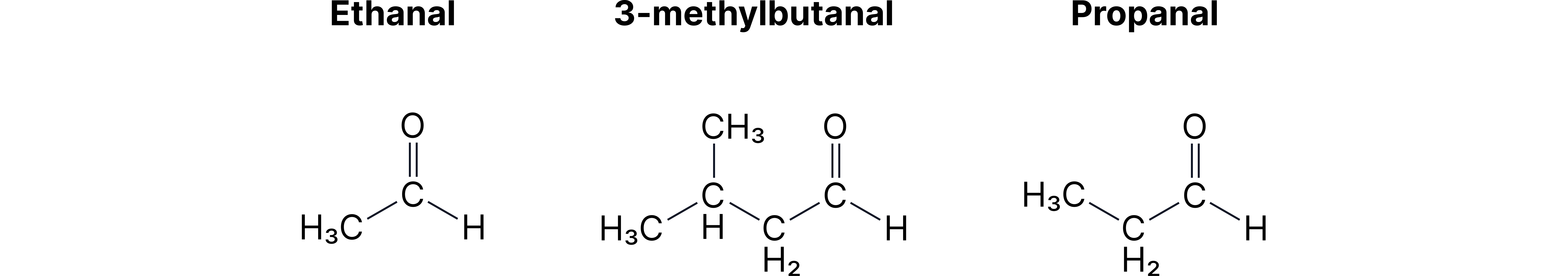
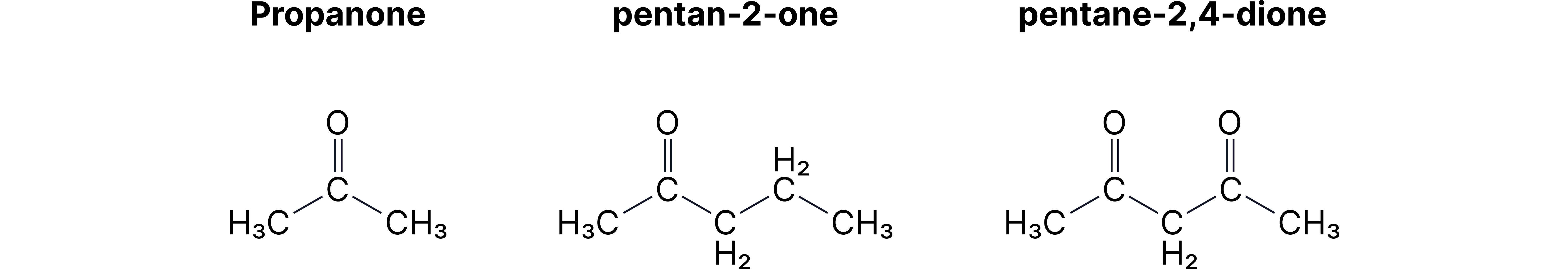
• C₃H₆O: propanal vs propanone.
• C₄H₈O₂: butanoic acid vs methyl propanoate vs ethyl ethanoate.

• C₃H₉N: propylamine vs trimethylamine.
Worked Examples and Problem-Solving
Simple Alkane Naming
Example 1: CH₃-CH₂-CH(CH₃)-CH₂-CH₃
1. Longest chain = 5 carbons (pentane)
2. Methyl substituent on carbon-3 (number from either end gives same result) 3.
Answer:
3-methylpentane
Example 2: CH₃-CH(CH₃)-CH(CH₃)-CH₂-CH₃
1. Longest chain = 5 carbons (pentane)
2. Two methyl groups on carbons 2 and 3
Answer:
2,3-dimethylpentane
Functional Group Priority Examples
Example: CH₃-CH(OH)-CH₂-CHO
1. Functional groups: alcohol (-OH), aldehyde (-CHO)
2. Aldehyde has higher priority → gets suffix (-al)
3. Aldehyde carbon = carbon-1
4. Alcohol on carbon-3 → becomes "3-hydroxy"
Answer:
3-hydroxybutanal
Complex Multi-functional Molecules
Challenge Example: NH₂-CH₂-CH₂-COOH
1. Functional groups: amine (NH₂), carboxylic acid (COOH)
2. Carboxylic acid has higher priority
3. Carboxylic acid carbon = carbon-1
4. Amine on carbon-3 → becomes "3-amino"
Answer:
3-aminopropanoic acid
Common Mistakes and How to Avoid Them
Numbering Errors
Mistake: Not giving the priority functional group the lowest possible number.
Example: Naming CH₃-CH₂-CH(OH)-CH₃ as "butan-3-ol" Correct: "butan-2-ol" (number from the other end).
Wrong Parent Chain
Mistake: Not choosing the longest chain containing the functional group.
Solution: Always find the longest possible chain that includes the priority group.
Ester Naming Confusion
Mistake: Naming the parts backwards Example: CH₃-COO-CH₂-CH₃ incorrectly as "ethanoyl ethyl" Correct: "ethyl ethanoate" (alcohol part first, then acid part)
Extension Topics
Cyclic Compounds
• Prefix: cyclo-
• Examples: cyclohexane, cyclopentanol, cyclohexanone.
• Numbering: Start from functional group, minimize other substituent numbers.
Remember: IUPAC nomenclature is a logical coding system - once you understand the rules, you can decode any molecule's structure from its name, or construct the correct name from any structure.