Functional Groups
Lajoy Tucker
Teacher

Contents
Definition
A functional group is a specific group of atoms within a molecule that is responsible for the characteristic chemical reactions of that molecule.
Common Functional Groups
|
Functional Group |
Suffix/Prefix |
Example Name |
|
|
Alkane |
|
–ane |
Methane |
|
Alkene |
 |
–ene |
Ethene |
|
Alcohol |
–ol |
Ethanol |
|
|
Halogenoalkane |
|
chloro–, bromo– etc |
2-chloropropane |
|
Aldehyde |
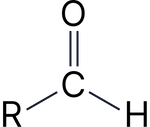 |
–al |
Ethanal |
|
Ketone |
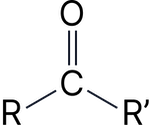 |
–one |
Propanone |
|
Carboxylic acid |
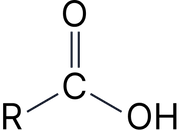 |
–oic acid |
Ethanoic acid |
|
Amine (primary) |
|
–amine / amino– |
Ethanamine |
|
Nitrile |
|
–nitrile |
Propanenitrile |
|
Ester* |
 |
–oate |
Methyl ethanoate |
|
Amide (primary)* |
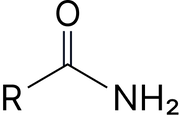 |
- amide |
Ethanamide |
|
Acyl chloride* |
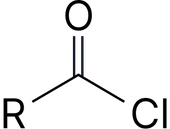 |
-oyl chloride |
Ethanoyl chloride |
|
Acid anhydride* |
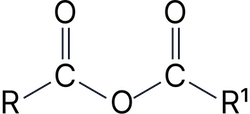 |
-oic anhydride |
Ethanoic anhydride |
R = an alkyl group e.g.
* = Y13 only
Homologous Series
A homologous series is a family of organic compounds with:
The same functional group
The same general formula
Similar chemical properties
Gradually changing physical properties
Members differ by units
E.g. Methane, ethane, propane etc
Questions
Question 1
Identify the functional group in the molecule
Alcohol – contains the group.
Question 2
Identify the functional group in the molecule
Name: Propanone
Functional group: Ketone (–CO–)
Question 3
Which homologous series contains compounds with the general formula ?
Alcohols
Question 4
State the functional group present in each of the following:
a.
b.
c.
(a) Carboxylic acid
(b) Halogenoalkane
(c) Nitrile
Question 5
Which of the following has a ketone functional group?
A. Propanal
B. Butanone
C. Ethanoic acid
D. Butanol
B. Butanone
Question 6
Give the name and functional group of the compound with structure
Name: Propanoic acid
Functional group: Carboxylic acid (–COOH)
Tips
Don't confuse aldehydes () with alcohols ()
Check for alkenes (C=C) in skeletal formulae – no hydrogen atoms are shown.
Functional groups determine the compound’s reactivity, naming, and classification.
Recognise the general formulae for alkanes () and alkenes () to predict the functional group from just the molecular formula