Types of Formulae
Lajoy Tucker
Teacher

Contents
Objective
Recognise and construct the empirical, molecular, general, structural, displayed, and skeletal formulae for organic compounds (with chains and rings up to six carbon atoms).
Overview of Formula Types
| Formula Type | Description |
| Empirical |
Simplest whole-number ratio of atoms in a compound |
| Molecular |
Actual number of each type of atom in a molecule |
| Displayed |
Shows all atoms and all bonds in the molecule |
| Structural |
Shows the arrangement of atoms without single bonds |
| Skeletal |
Shows structure with C and H* labels removed (H’s shown when attached to heteroatom (e.g. O or N) |
Examples
Empirical Formula
Simplest whole-number ratio of atoms in a compound.
Example:
has empirical formula
has empirical formula
Molecular Formula
Total number of each atom in the molecule.
Example:
Ethanol =
General Formula
Algebraic formula for a homologous series.
Example:
Alkanes:
Alkenes:
Alcohols:
Displayed Formula
All bonds and atoms shown.
Example:
Ethanol =
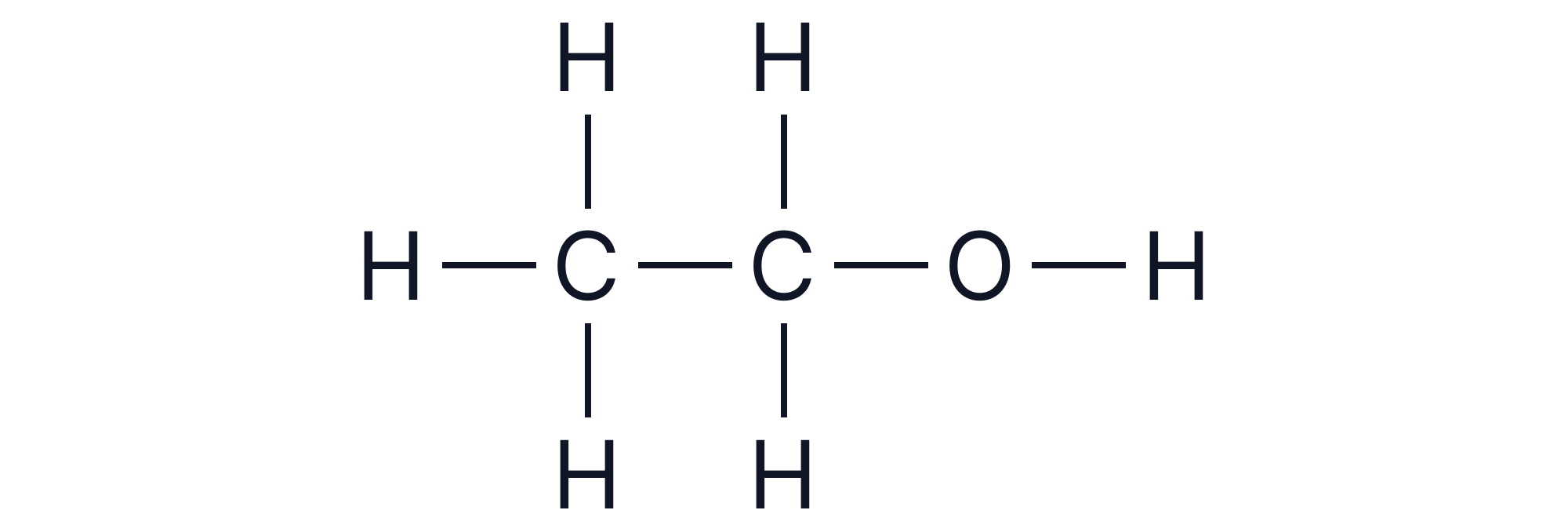
Note - Ensure ALL bonds are shown including O-H bonds
Example: Propanoic acid
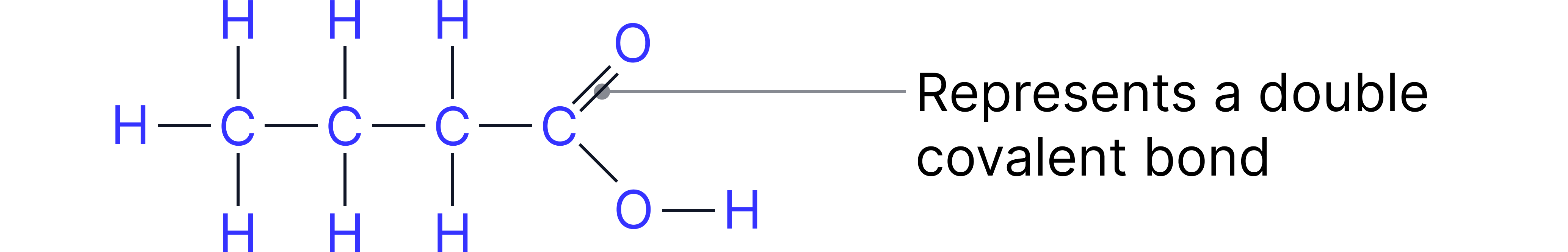
Structural Formula
Shows how atoms are arranged in the molecule in order.
Example 1: Butan-1-ol
Displayed:
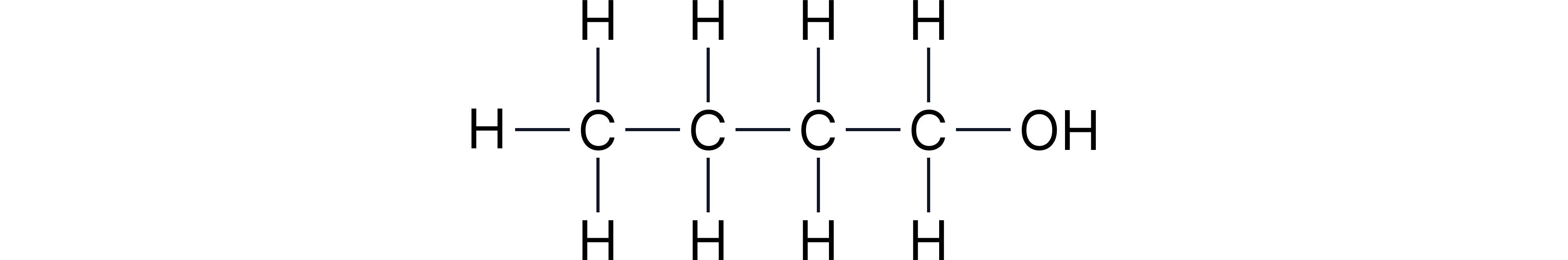
Structural:
Example 2: propene
Single bonds not shown but C=C double bonds often included.
Displayed:
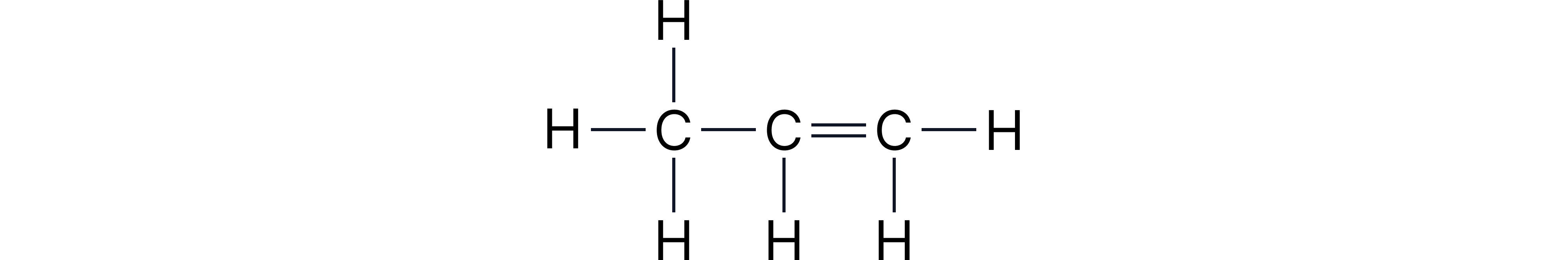
Structural:
Example 3: 2-methylbutane
Parentheses used to show repeating units or groups within a chain and/or to show branches attached to the main chain
Displayed:
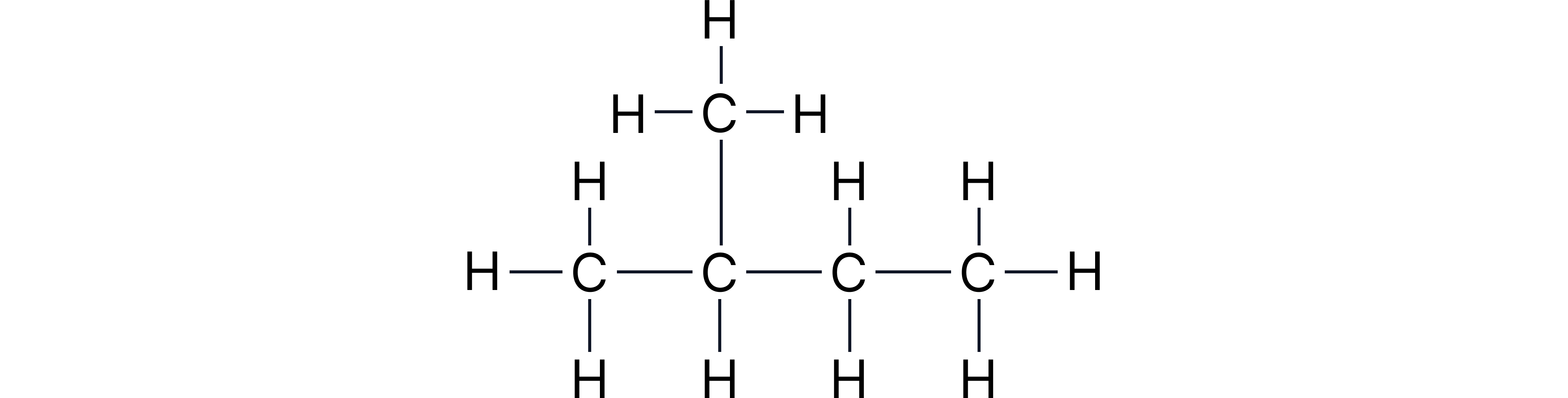
Structural:
Skeletal Formula
Carbon atoms are vertices or line ends.
Hydrogens on carbon atoms are omitted.
Heteroatoms (like O, N, Cl) are shown.
Example 1 = Propan-1-ol
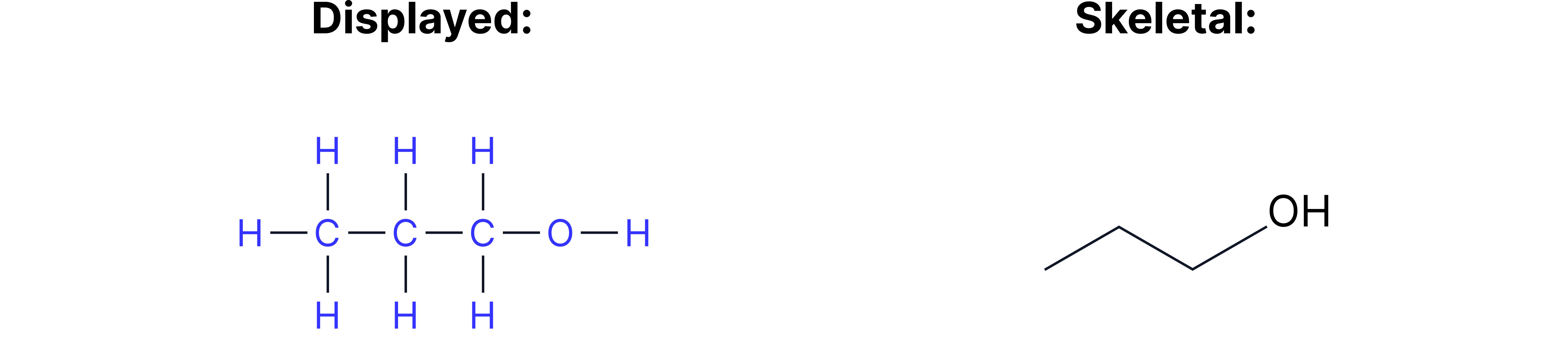
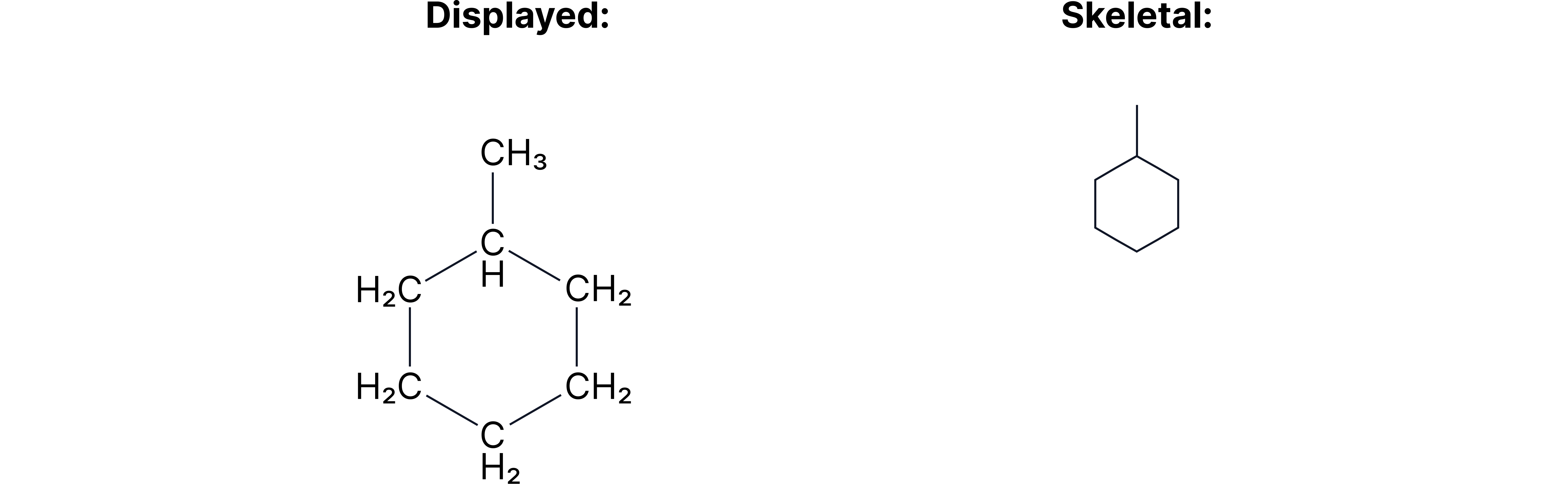
Example 2 = Methylcyclohexane
Skeletal formulae are particularly useful for cyclic compounds where showing all the C’s and H’s lead to a structure that is difficult to read and use in mechanisms.
Structural:
Example 3 = But-1-ene
The difference between double and triple bonds are shown through the number of lines between carbon vertices.
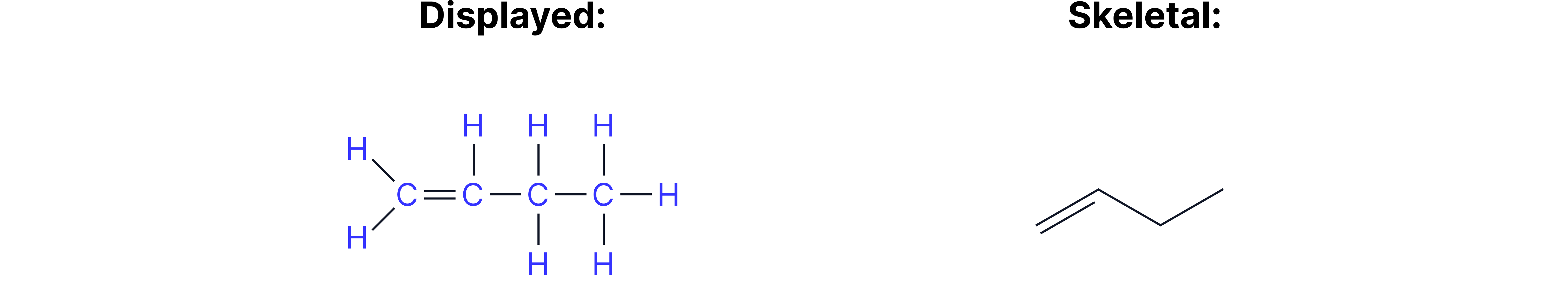
Practice Questions
Question 1
Give the displayed and structural formulae of 2-chloro-2-methylbutane.
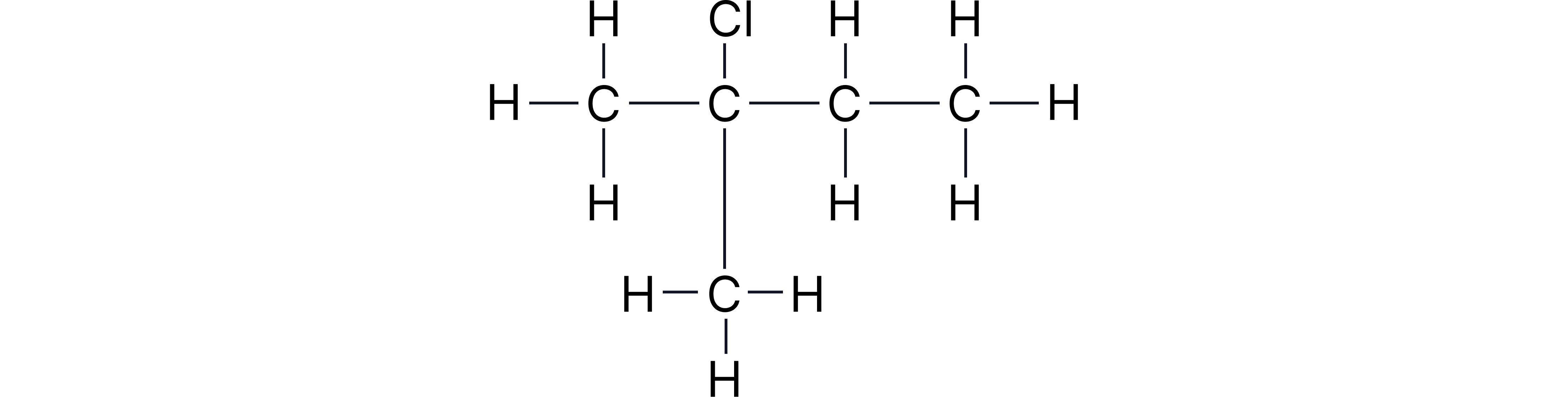
Question 2
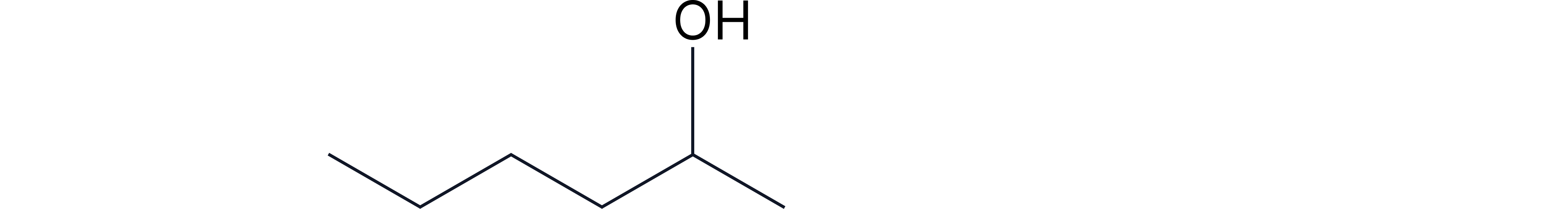
Draw the skeletal formula of hexan-2-ol.
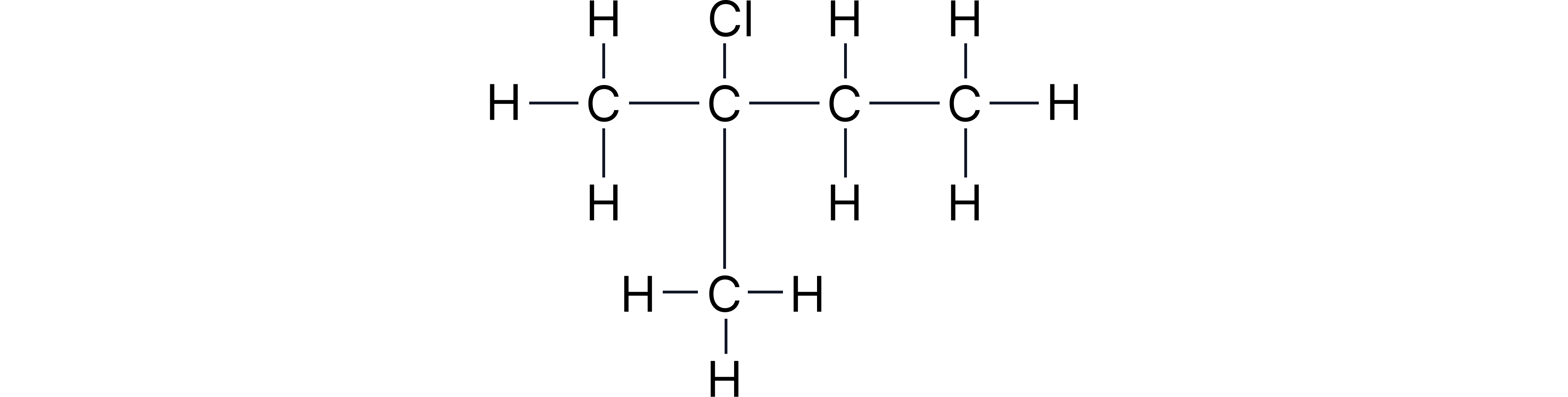
Question 3
Give the structural formula for the compound below.
Tips
Displayed formula = good for showing bonding, but time-consuming to draw. Use brackets in structural formulae where you have branches and/or more of one of the same group attached to a carbon atom
Use skeletal formula for large carbon chains and cyclic compounds to save time and reduce clutter.
Remember: skeletal formula omits H on carbon, but not heteroatoms like O or N.