Structural Isomerism
Lajoy Tucker
Teacher

Introduction and Definitions
Structural isomers are compounds with the same molecular formula but different structural formulae. This means they have the same number of atoms of each element but differ in the way atoms are connected, arranged, or which functional group is present.
Types of Structural Isomerism
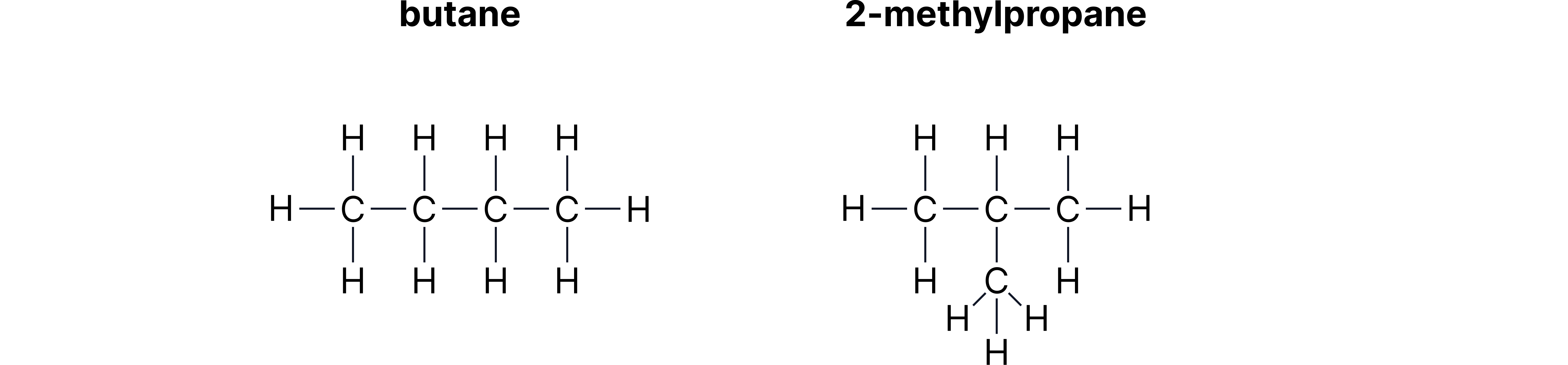
A. Chain Isomerism - same molecular formula, different carbon chain arrangement.
Example:
Butane:
2-methylpropane:
Both have molecular formula
B. Position Isomerism - Functional group is in a different position on the same carbon skeleton.
Example:

Both have molecular formula
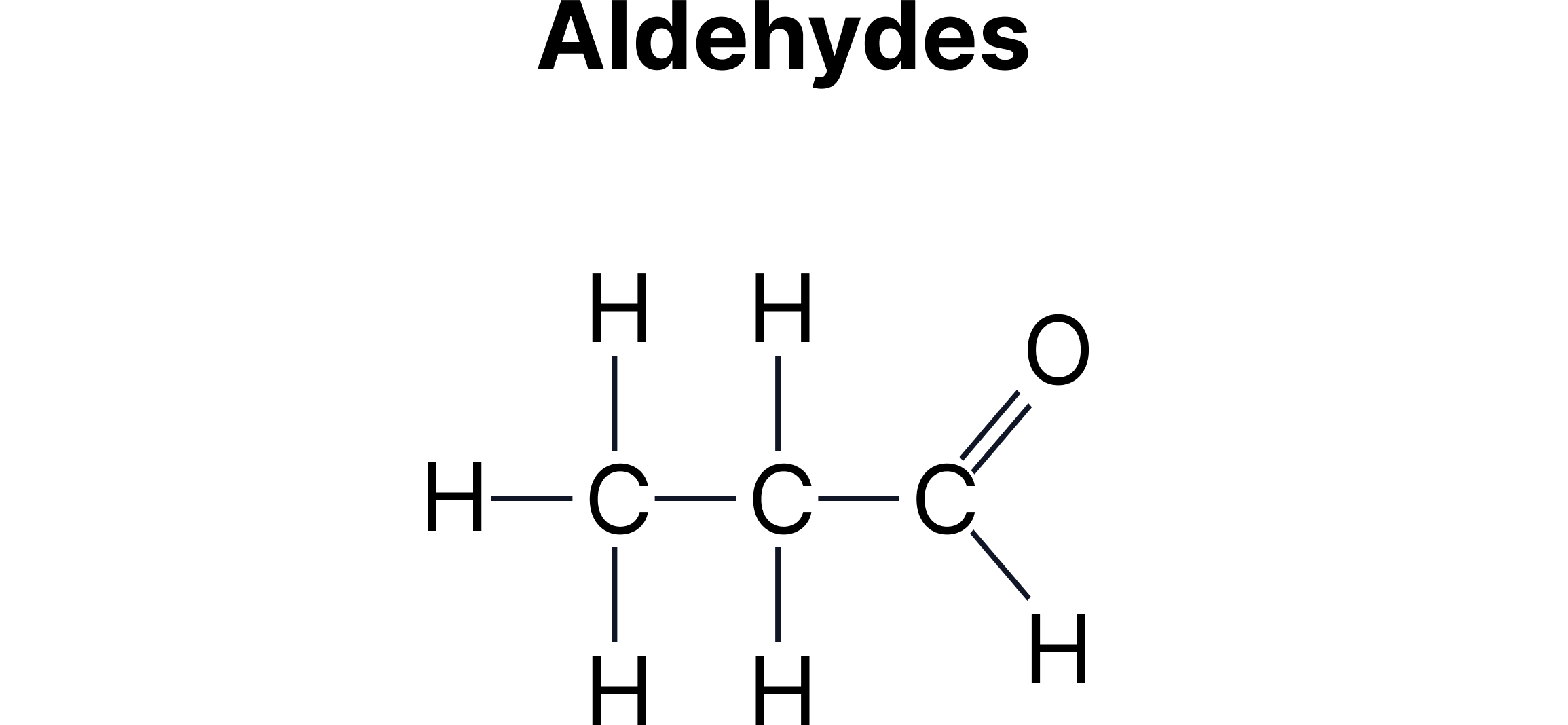
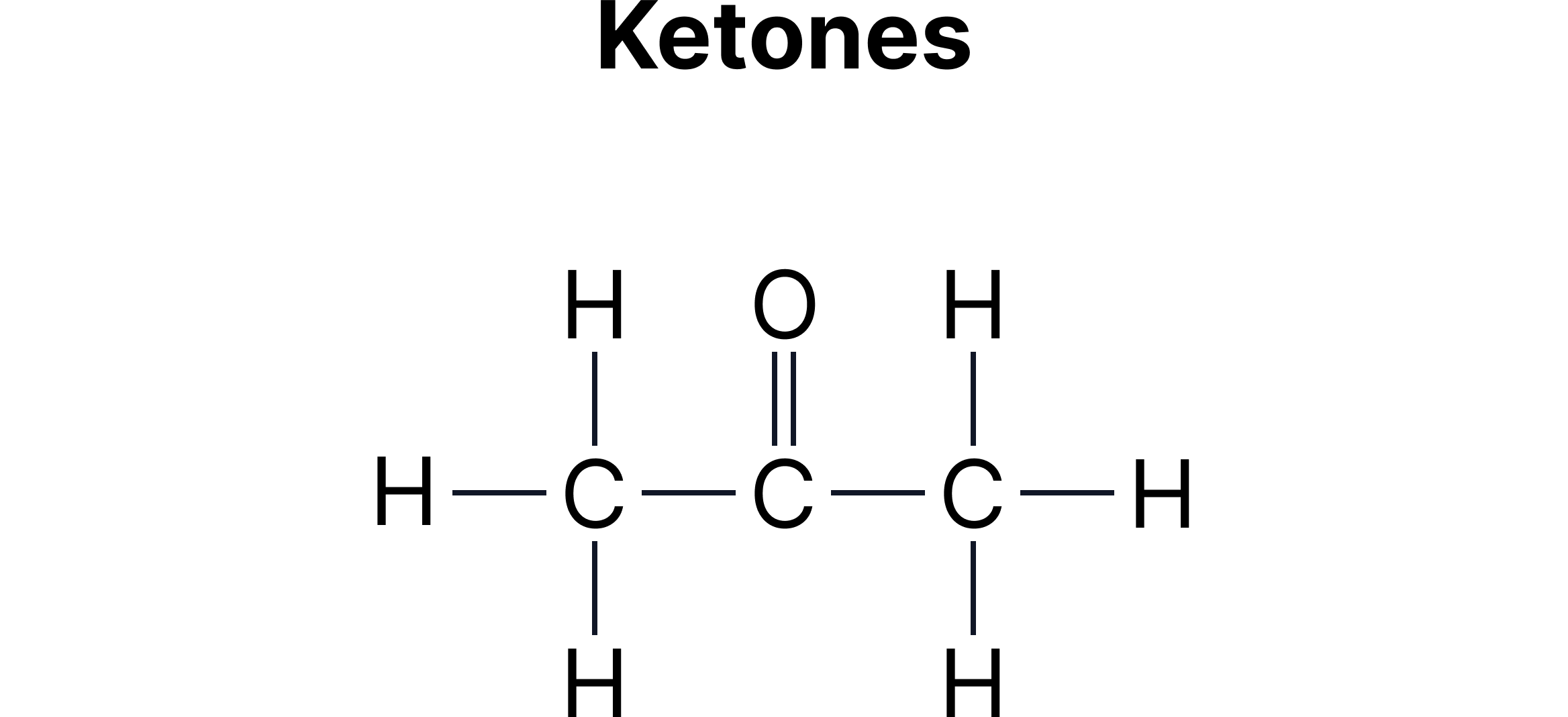
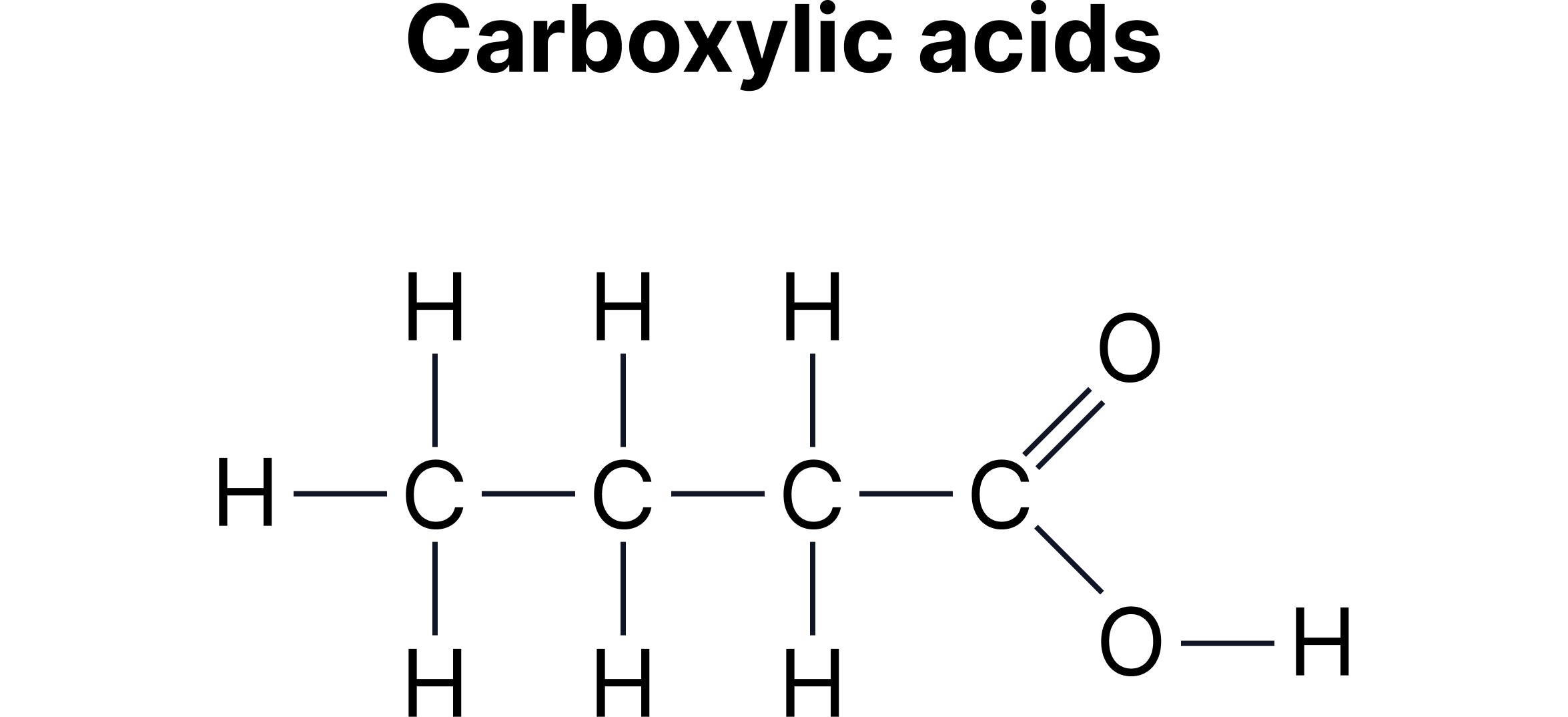
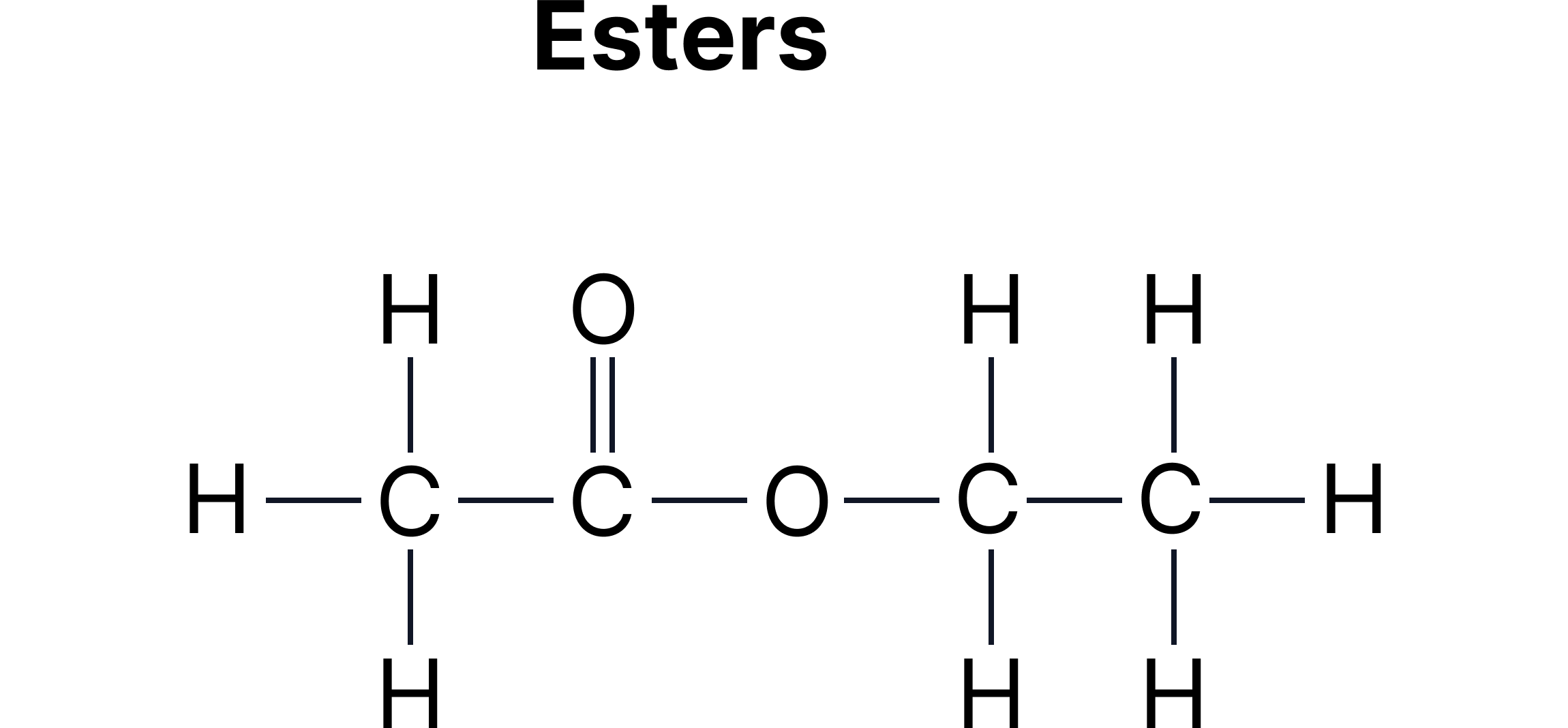
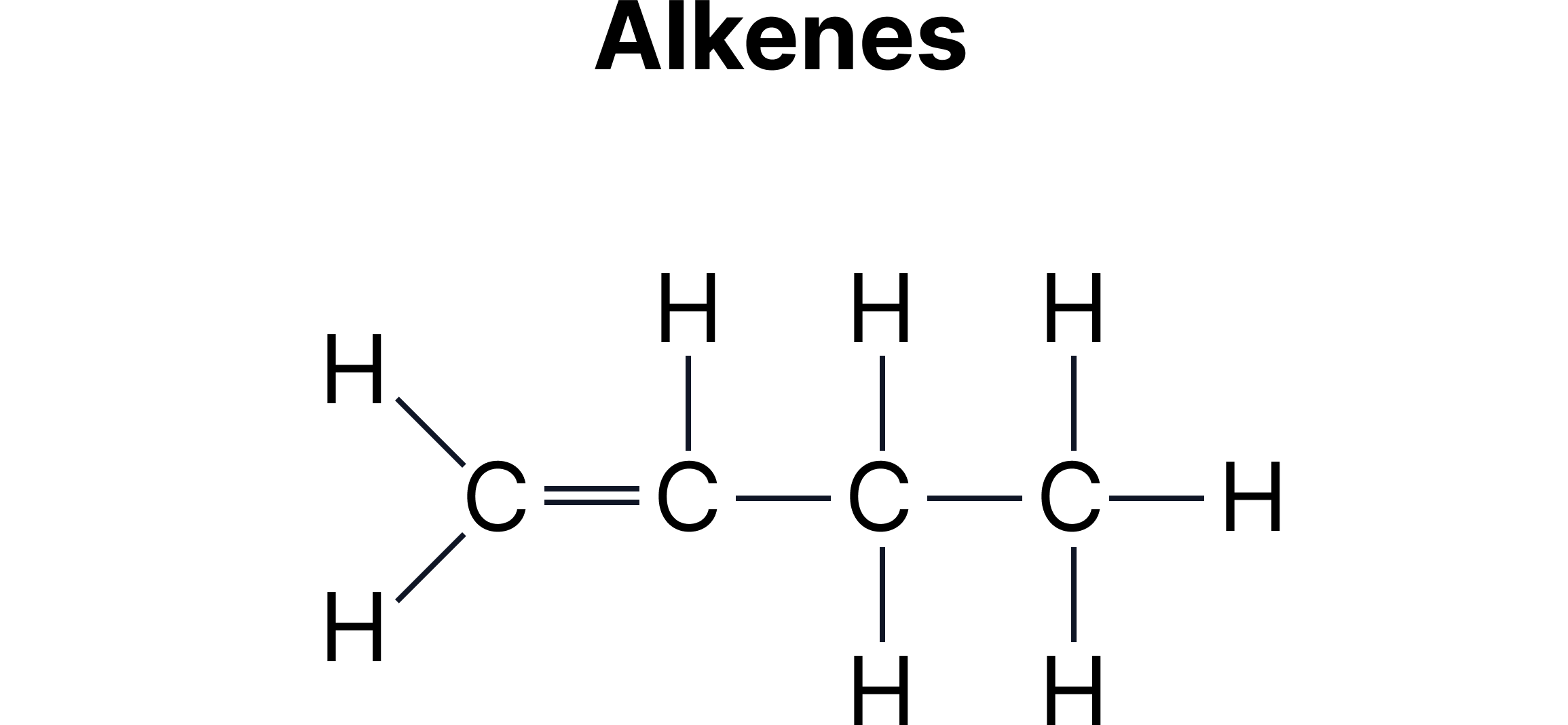
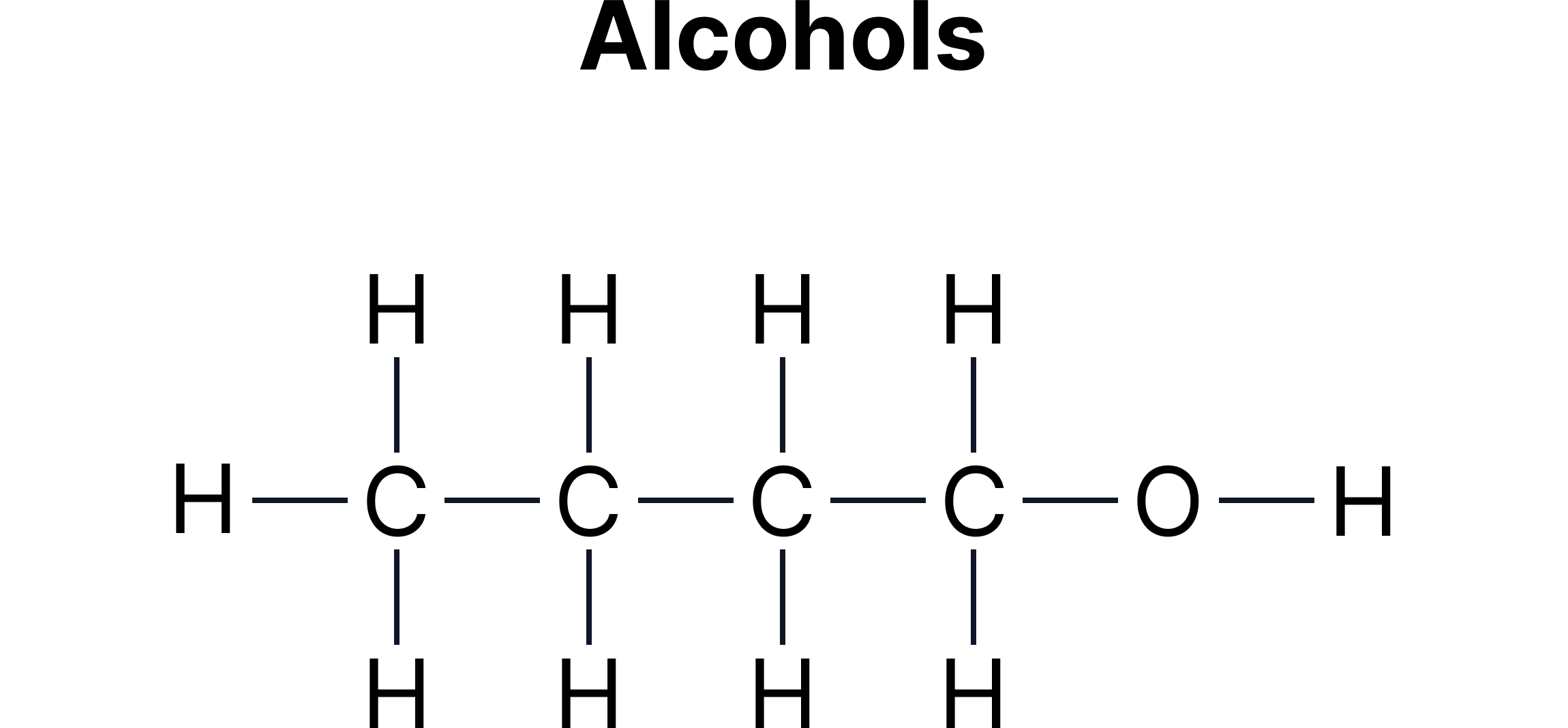
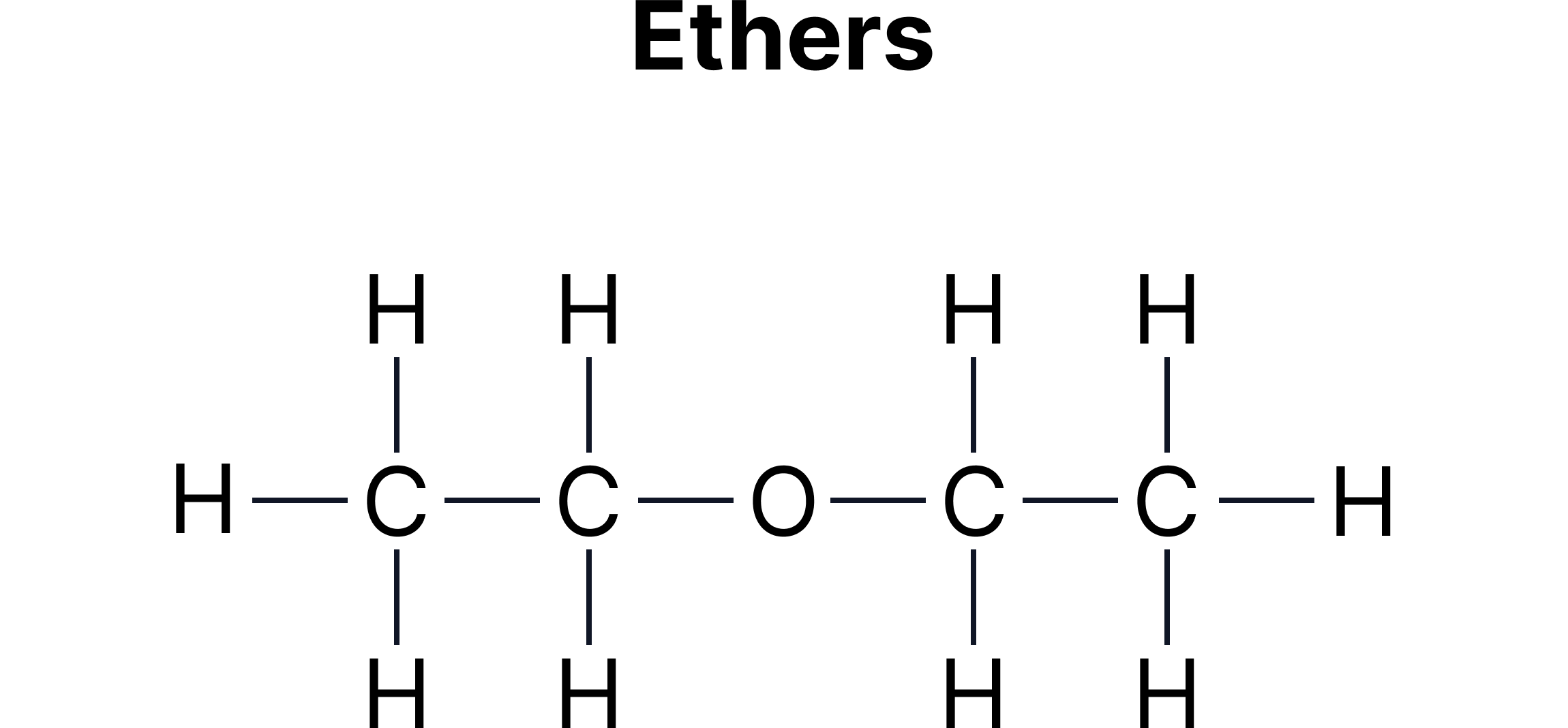
C. Functional Group Isomerism - Same molecular formula but different functional groups.
Example:

Common Functional Group Isomers in A-level Chemistry
|
|
|
|
|
|
|
|
Summary
| Isomerism Type | Difference In |
Example Pair |
| Chain | Carbon backbone structure | Butane & 2-methylpropane |
| Position | Location of a functional group | Butan-1-ol & Butan-2-ol |
| Functional Group |
Type of functional group present |
Propanal & Propanone |
Question 1
Give a chain isomer of pentane.
2-methylbutane or 2,2-dimethylpropane
Question 2
Name two position isomers of
-
1-bromobutane
-
2-bromobutane
Question 3
Give the names and type of isomerism between and
.
- Names: Butane & 2-methylpropane
-
Type: Chain isomerism
Question 4
State the number of structural isomers of molecular formula that are alcohols.
3 isomers:
-
Butan-1-ol
-
Butan-2-ol
-
2-methylpropan-1-ol
Key Tips
Always count the total number of each atom to confirm same molecular formula.
In chain isomerism, be systematic — shift methyl groups without duplicating structures.
Functional group isomerism often appears between:
Alcohols and ethers
Aldehydes and ketones
Carboxylic acids and esters
Alkenes and cycloalkanes