Oxidation States
Lajoy Tucker
Teacher

Introduction and Definitions
An oxidation state (oxidation number) is a number assigned to an atom in a compound or ion that represents its degree of oxidation.
It is a hypothetical charge that shows the number of electrons lost or gained compared to the uncombined element.
Oxidation states are used to determine what is oxidised and what is reduced in redox reactions.
Oxidation = Increase in oxidation state
Reduction = Decrease in oxidation state
Oxidation states are conventionally written with the sign followed the number.
e.g. +3, not 3+
Oxidation State Rules
Rule | Description | Example |
1 | Elements in their uncombined form have oxidation state 0 | O₂, Cl₂, Fe, N₂ all = 0 |
2 | Simple monatomic ions have oxidation state equal to their charge | Na⁺ = +1, Cl⁻ = -1 |
3 | Fluorine is always –1 in compounds | F in HF = –1 |
4 | Group 1 and group 2 metals are +1 and +2 respectively in compounds | In , Na = +1 |
5 | Oxygen is usually –2 | In H₂O, O = –2 |
Exception: In peroxides | In H₂O₂, O = –1 | |
Exception: Bonded to fluorine | In OF₂, O = +2 | |
6 | Hydrogen is usually +1 | In HCl, H = +1 |
Exception: In metal hydrides | In NaH, H = –1 | |
7 | The sum of oxidation states in a neutral molecule = 0 | H₂SO₄: total = 0 |
8 | The sum of individual oxidation states in a complex ion equals the ion’s charge | SO₄²⁻ = –2 overall |
Start with known oxidation states using the rules above.
Work out the unknown by ensuring the sum equals the molecule or ion’s overall charge.
Tip
Oxygen and hydrogen are often key starting points in most questions as they usually have oxidation states of -2 and +1 respectively in compounds.
Worked Examples
Assign the following oxidation states
Question 1
Answer:
This is uncombined element therefore its oxidation state is 0.
Question 2
in
Answer:
The overall compound is neutral so the individual oxidation states must add to 0.
Hydrogen usually has a +1 oxidation state.
Therefore, Br must have an oxidation state -1.
Question 3
in
Answer:
The overall compound is neutral so the individual oxidation states must add to 0.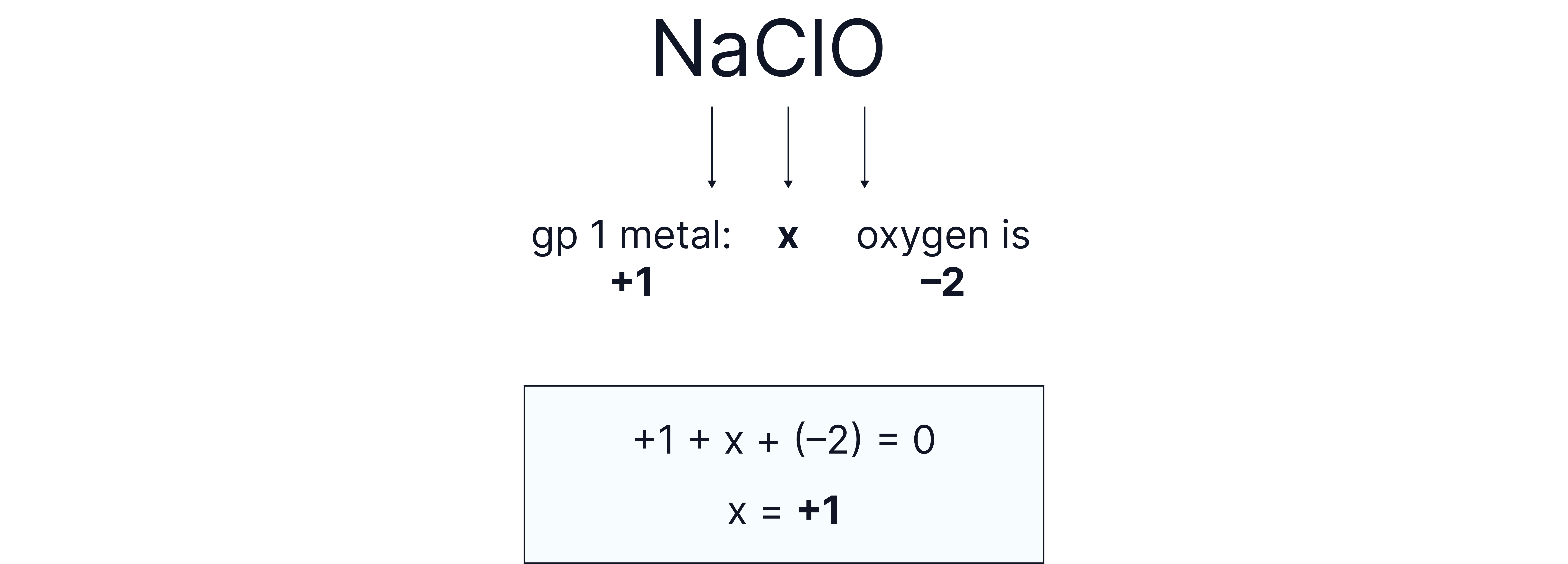
in
Answer:
The ion has a 2- charge so the individual oxidation states must add to -2.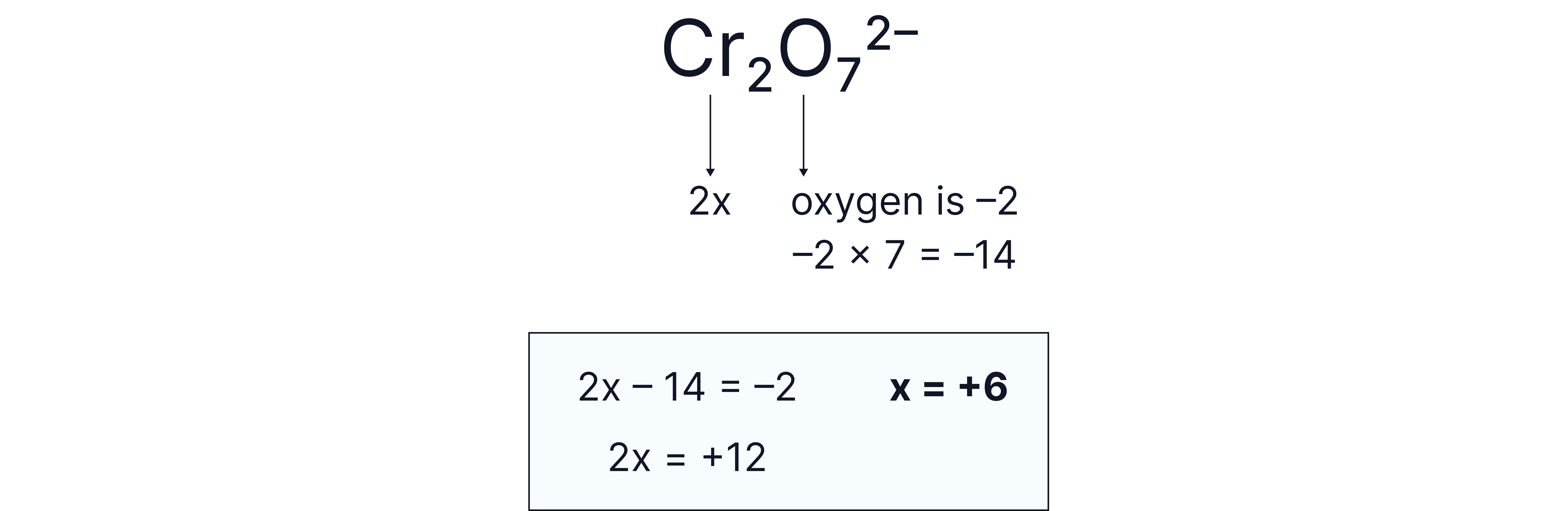
Practice Questions
Question 1:
What is the oxidation state of in ?
Answer:
Let =
is
Question 2
Which species has an oxidation state of +5 on the central atom?
A.
B.
C.
D.
Answer:
: Correct answer: A
Question 3:
What is the oxidation state of sulfur in ?
Answer:
Let
Sulfur is in the oxidation state.
Key Tips
Always assign oxidation numbers to known elements first (H, O, F, group 1 and group 2 metals).
Remember common exceptions: O in peroxides (–1), H in metal hydrides (–1).
In ions, total oxidation states must equal the ion’s charge.
For molecules, the sum must equal 0.
Use oxidation states to determine what is oxidised/reduced, and hence find the agents.