Redox Equations
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
A redox equation is a balanced chemical equation that shows the transfer of electrons during a redox reaction, combining both oxidation and reduction half-equations.
You will be expected to:
Write oxidation and reduction half-equations.
Combine half-equations to form balanced full redox equations.
Use oxidation states to determine what is being oxidised and reduced.
Half Equations
Half equations show the process of oxidation or reduction through the loss or gain of electrons (link to Oxidation and Reduction note)
Constructing half equations:
Step-by-step process:
1. Balance atoms (except and ).
2. Balance by adding .
3. Balance by adding ions (in acidic solutions).
4. Balance charge (or oxidation states) by adding electrons to the appropriate side.
Always check that all species and charges are balanced.
Worked Example
1. to
Here the Cu atoms are already balanced and there are no ’s or ’s so we can skip directly to step 4

To balance the charges, 2 electrons must be placed on the right hand side

Charges and species are now balanced.
Oxidation or reduction?
The electrons on the right hand side (products) have been lost from to produce . The Cu has therefore been oxidised.
2. to
Step 1: Balance the atoms
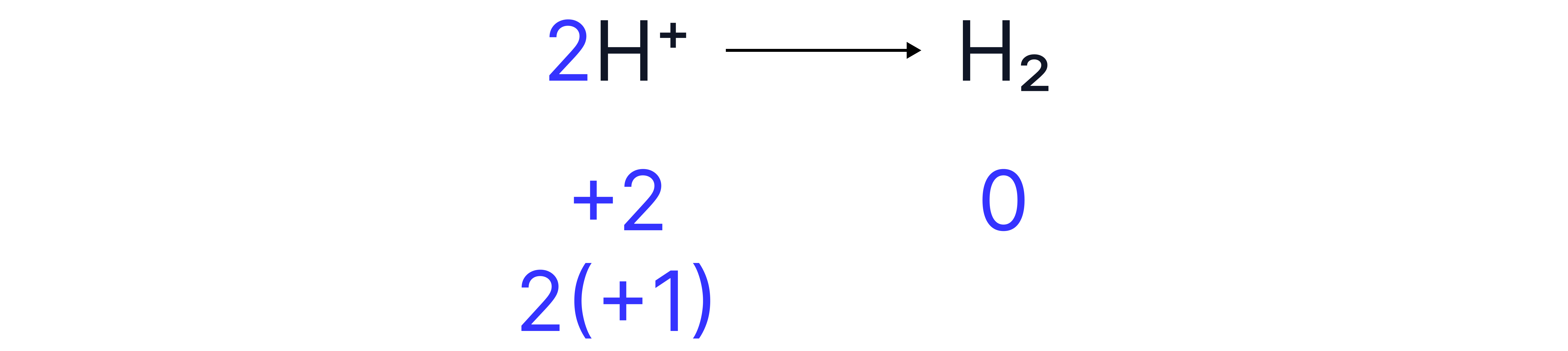
There are no ’s or ’s so we can skip directly to step 4
To balance the charges, 2 electrons must be placed on the right hand side

Charges and species are now balanced.
Oxidation or reduction?
The electrons on the left-hand side (reactants) have been gained by the ions to produce . has therefore been reduced.
We can also think about this as the decrease in oxidation state from +1 to 0
3. to
Step 1: Balance Mn: already 1 on each side
Step 2: Balance on LHS → add to RHS
Step 3: Balance on RHS → add to LHS
Step 4: Balance charge: LHS = , RHS add to LHS
Balancing in terms of the oxidation state of gives the same answer. Decrease in oxidation state from +7 to -2
Combining Half Equations
For the overall redox equation, oxidation and reduction half equations must be combined. To do this,
The number of electrons in both half-equations must be the same. This is because the number of electrons gained by one species must be equal to the number lost by the other to satisfy the law of conservation of mass.
The two half-equations must be added together.
Finally, cancel any species that appear on both sides (e.g. , electrons)
Reaction:
Balance the redox reaction between and in acidic solution.
Oxidation Half-Equation :
Reduction Half-Equation :
Multiply oxidation half-equation ×5 to balance electrons in both half equations:
Add half-equations (keep species on their side of the reaction arrow)
Practice Questions
Question 1
Write a balanced half-equation for the reduction of to in acidic conditions.
Answer
: already balanced
: extra O → add
: add to LHS
Charge: LHS , RHS add to the LHS
Question 2
In acidic conditions, dichromate(VI) ions oxidise iron(II) ions to iron(III) ions. Write the balanced ionic equation for this redox reaction.
Answer
Step 1: Half-equations
Step 2: Multiply Fe equation ×6:
Step 3: Combine:
Key Tips
Always balance atoms first, then charge using electrons.
Acidic solutions use and
When combining half-equations, equalise the electrons lost and gained before adding.
Always check final equations are balanced for atoms and charge.