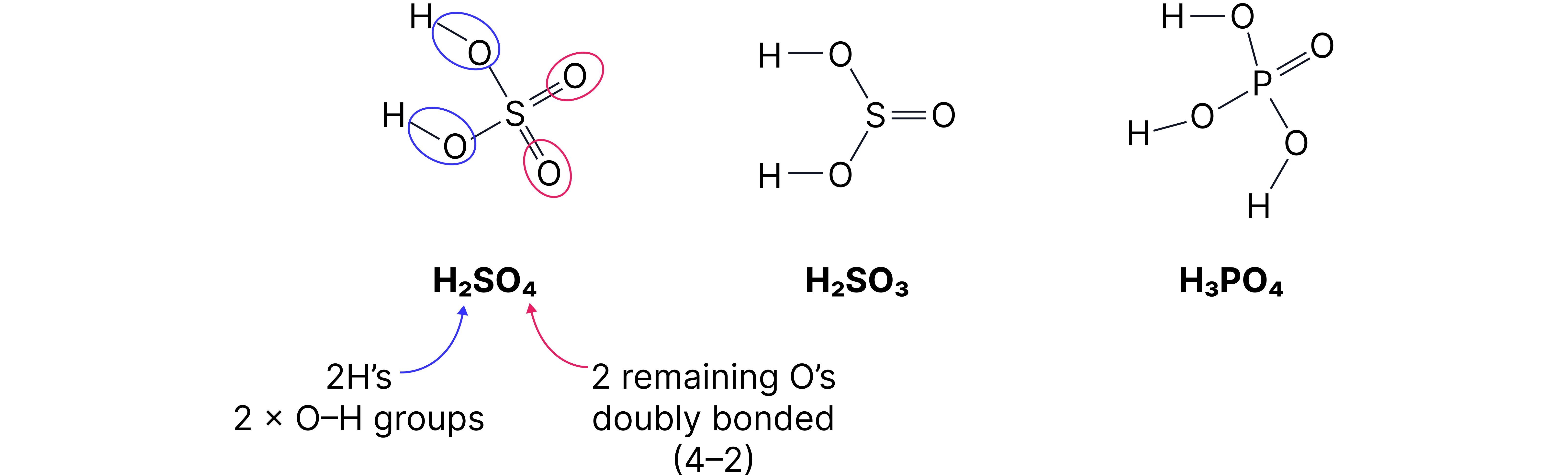Period 3 Oxides with Water
Lajoy Tucker
Teacher

Contents
Reactions of Period 3 Oxides with Water
Period 3 oxides react with water as summarised in the table below.
Oxide | Formula | Type of Oxide | Reaction with Water | pH of solution |
Sodium oxide | Basic | |||
Magnesium oxide | Weak basic | (sparingly soluble) | ||
Aluminium oxide | Amphoteric | Insoluble in water | No effect | |
Silicon dioxide | Acidic | Insoluble in water | No effect | |
Phosphorus pentoxide | Acidic | |||
Sulfur dioxide | Acidic | |||
Sulfur trioxide | Acidic |
Period 3 Oxides with Water Explained
Basic Oxides
Sodium Oxide
Reaction:
Type: Strongly basic oxide
Result: Highly alkaline solution, pH
Magnesium Oxide
Reaction:
Type: Weak base, slightly soluble
Result: Mildly alkaline solution, pH
Comparing Basicity
Sodium oxide and magnesium oxides are basic oxides as the ions combine with ions from water producing (hydroxide ions).
Magnesium ions are smaller and have a higher charge than sodium ions resulting in stronger electrostatic forces of attraction between the metal ions and the oxide ions in magnesium oxide. The oxide ions in magnesium oxide are therefore less available to combine with the ions making the resultant solution less alkaline than with sodium oxide with lower .
Magnesium hydroxide is also only sparingly soluble resulting in a lower concentration in solution.
Insoluble Oxides
Neither aluminum oxide or silicon dissolve in water. This means that despite any acidic/basic nature, there will be no effect on the of the water when these solids are added to it.
Aluminium Oxide
Reaction with water: None (insoluble)
Type: Amphoteric – reacts with both acids and bases, but not water
Effect: No pH change
Silicon Dioxide
Reaction with water: None – insoluble
Type: Weak acidic oxide
Effect: No pH change, but reacts with strong bases (not needed here)
Acidic Oxides
Phosphorus Pentoxide
Reaction:
Type: Strong acidic oxide
Result: Strong acidic solution, pH
Sulfur Dioxide
Reaction:
Product: Sulfurous acid, weak acid
pH:
Sulfur Trioxide
Reaction:
Product: Sulfuric acid, strong acid
pH:
A common exam question is to suggest a method to distinguish between samples of and . As they produce solutions of different acids strengths, a simple approach is to react both with the water and then test the of the resultant solutions using universal indicator or a meter.

Displayed formulae of Acids
The structures of the acids and can be drawn simply by:
1. Centring the period element.
2. Adding as many groups as there are in the formula.
3. Adding remaining groups connected with double bonds.
General Trend Across the Period
Oxides become more acidic across Period 3.
Ionic oxides (left of the period) form alkaline solutions.
Covalent oxides (right of the period) form acidic solutions.
Aluminum oxide is amphoteric (no reaction with water, but reacts with both acids and bases).
Practice Question
Question 1
Which of the following oxides of Period elements reacts with water to form a solution with a pH greater than ?
a)
b)
c)
d)
Answer:
C.
Explanation: produces when reacted with water which is a strongly alkaline solution.
Question 2
Describe and explain the trends in the reactions of Period oxides with water across the period from to . Include the pH of the resulting solutions where appropriate.
Answer:
Oxides across Period
change from basic to acidic.
and react with water to form alkaline solutions ( and ).
and do not react with water (insoluble).
and react with water to form acidic solutions (phosphoric, sulfurous, and sulfuric acids).
pH decreases across the period: from
to
Question 3
Write a balanced equation for the reaction of phosphorus oxide with water. What is the approximate pH of the resulting solution?
Answer:
Equation:
pH: Around
Keys Tips and Reminders
Rather than memorising the equation , just make sure you know the formulae of the oxides and the product with water. This will then make constructing and balancing the equations straight forward.
Do not forget that while the oxides of aluminum and silicon can react with acids and/or bases, they will not react with the water due to insolubility.