Melting Point Trends
Lajoy Tucker
Teacher

Contents
Melting Point Trends
Melting points are based on the strength of the forces of attraction holding structure together and as a result the amount of energy required to overcome them.
Giant ionic lattice: A lattice of opposite charged ions held together by many strong electrotastic forces of attraction.
Giant covalent (macromolecular): A 3D network of atoms bonded by strong covalent bonds between atoms throughout the structure.
Simple molecular: Discrete molecules held together by weak intermolecular forces (van der Waals).
Metal oxide | ||||||
Structure | Giant ionic | Giant ionic | Giant ionic (with covalent character) | Giant covalent | Simple molecular | Simple molecular |
Held together by | Strong ionic bonds | Strong ionic bonds | Strong ionic bonds | Strong covalent bonds | Weak intermolecular forces | Weak intermolecular forces |
Relative MP | High↑ | High↑↑ | High↑↑↑ | High | Low↓ | Lowest↓↓ |
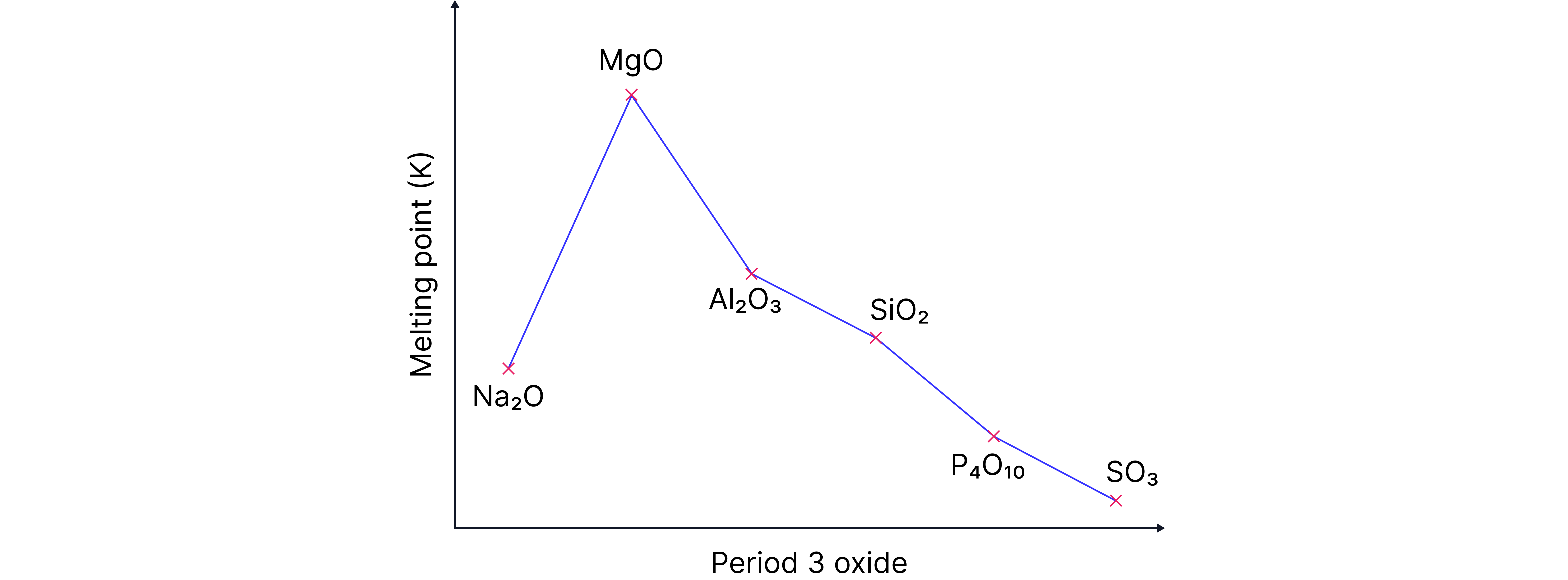
Period 3 Oxides Melting Point Explainer Video
Explanation of Melting Points
Sodium oxide to Magnesium oxide :
Trend: Melting point increases.
Increasing charge and decreasing size of the metal ions . Stronger electrotastic forces of attraction between positive metal ion and negative oxide ion. More energy needed to break bonds higher melting point.
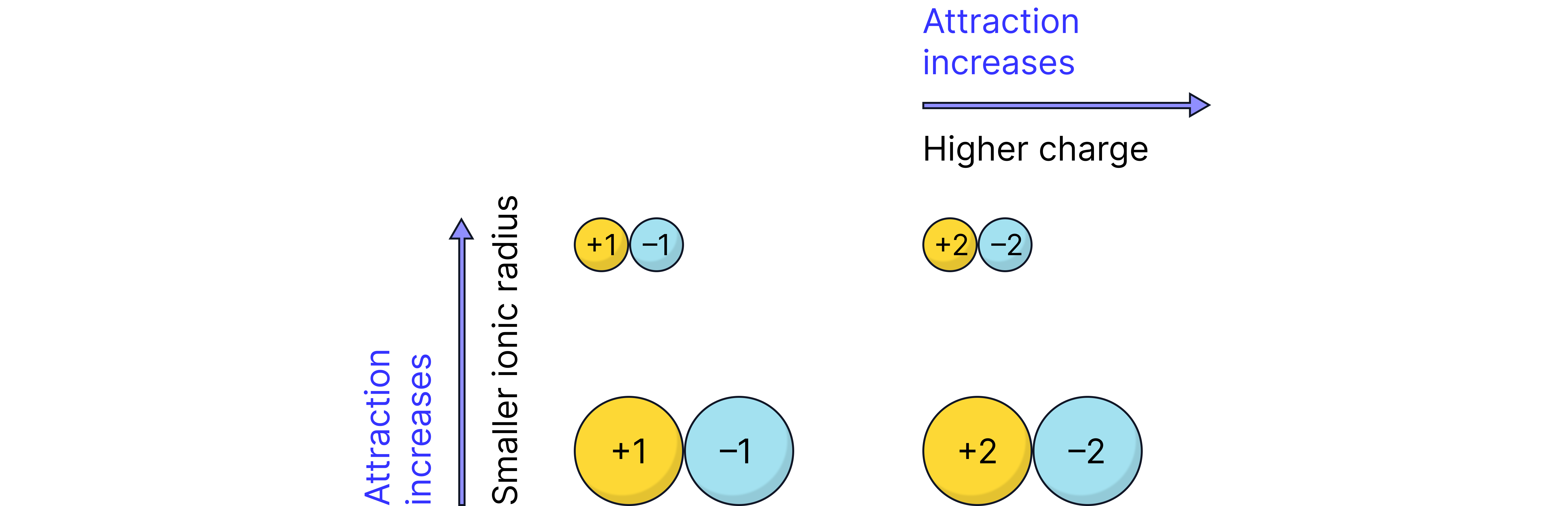
Note: Although ions are the smallest with the greatest charge, the structure of the lattice results in less efficient packing of ions and therefore a lower than expected melting point. This is not assessed by AQA.
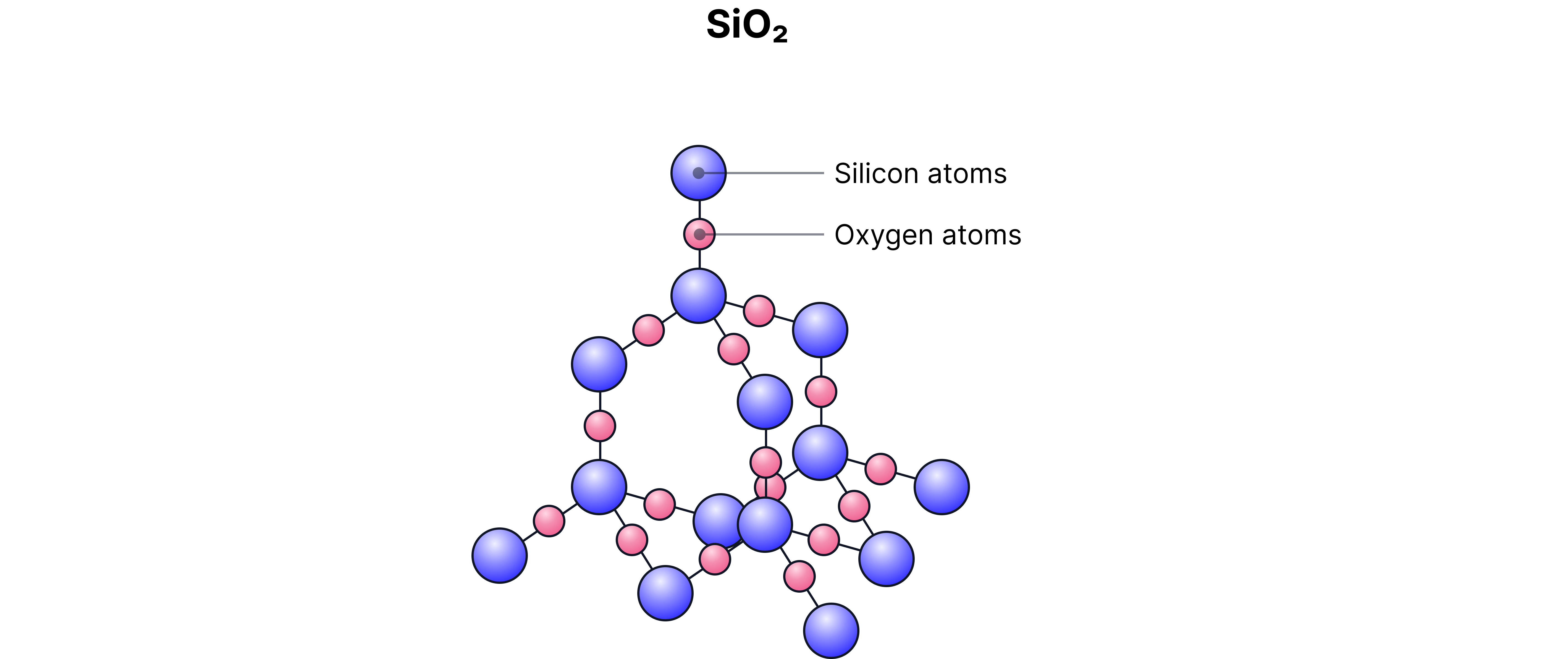
Silicon dioxide :
High melting point
Strong covalent bonds between silicon atoms throughout the structure. A large amount of energy is required to break this bonds.
Phosphorus oxide and oxides of sulfur and :
Trend: Melting point depends on size of molecules . Larger molecules = more electrons = stronger van ders Waals. More energy needed to overcome intermolecular forces higher melting points.
Practice Question
Question:
Which of the oxides below has the strongest ionic bonds?
A.
B.
C.
D.
Answer:
B.
has the strongest bonds, but these covalent.
and are ionically bonded. bonds are stronger due to the smaller size and greater charge of the ion compared to .