Reactions with Oxygen
Lajoy Tucker
Teacher

Contents
Reactions with Oxygen
Elements in period 3 from to react with oxygen to produce oxides where the period 3 element is in its highest oxidation state.
The highest oxidation state of an element corresponds to its group in the periodic table e.g. the highest oxidation state of is .
Period 3 Oxides to
Element | Oxide(s) | Name | |
Metals | Na | Sodium oxide | |
Mg | Magnesium oxide | ||
Al | Aluminium oxide | ||
Non-metals | Si | Silicon dioxide | |
P | Phosphorous (V) oxide | ||
S | and | Sulfur dioxide and sulfur trioxide |
Note: The formulae of the metal oxides be derived by considering the charges of the individual ions e.g. Two ions are required to balance the charge of the ion resulting in .
Note: The V corresponds to the oxidation state of the phosphorous atom.
Reactions of Period 3 Elements with Oxygen
Elements in period 3 react with oxygen to produce their oxides.
The equations, conditions, and observations are summarised below.
Element | Description | Equation | Observation |
Na | Burns vigorously | → | Yellow flame, white solid |
Mg | Burns vigorously | → | Bright white flame, white solid |
Al | Burns quickly if powdered | → | White solid (protective oxide layer) |
Si | Reacts slowly when heated | → | White solid |
P | Burns vigorously | → | White smoke (phosphorus pentoxide) |
S | Burns steadily Requires a catalyst and high temperature | Blue flame, choking gas |
Acid–base behaviour changes from basic → amphoteric → acidic across the period.
Period 3 Reactions with Oxygen Explainer Video
Structure and Bonding
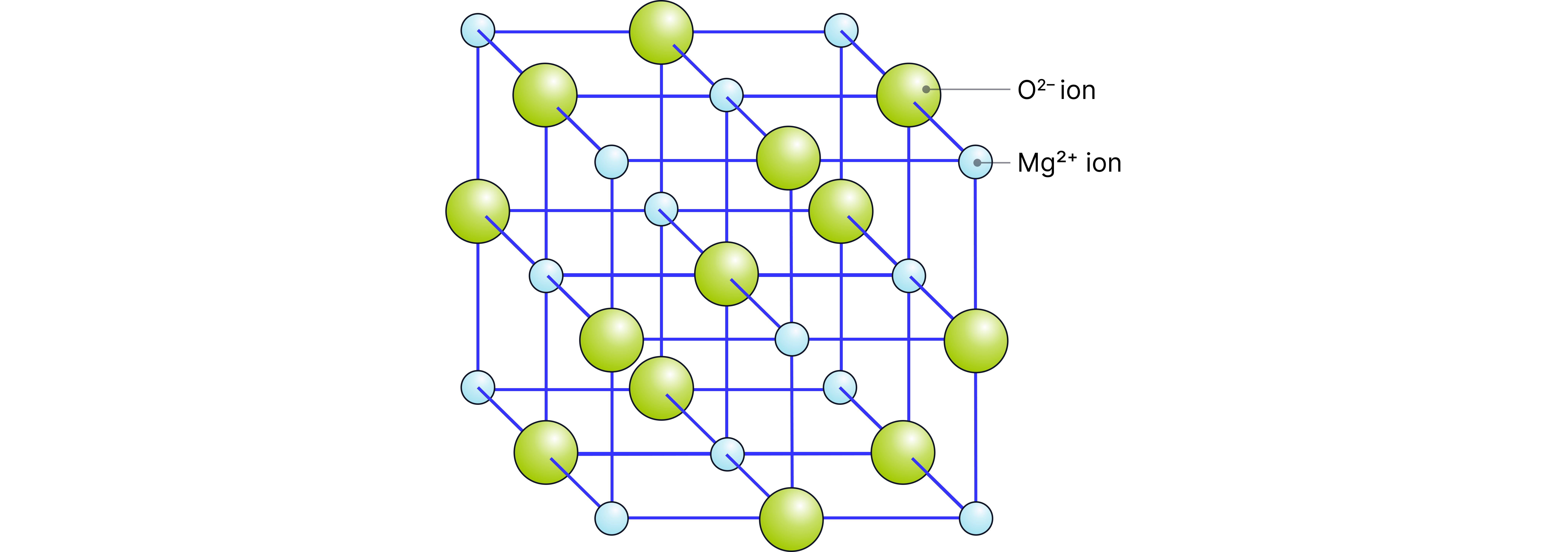
The metallic period 3 elements (sodium, magnesium and aluminum) react with oxygen to form metal oxides. These oxides are held together by ionic bonds where there are strong electrostatic forces of attraction between positive metal ions and negative oxide ions.
These attractive forces extend in three-dimensions to give a giant ionic lattice structure.
e.g. magnesium oxide
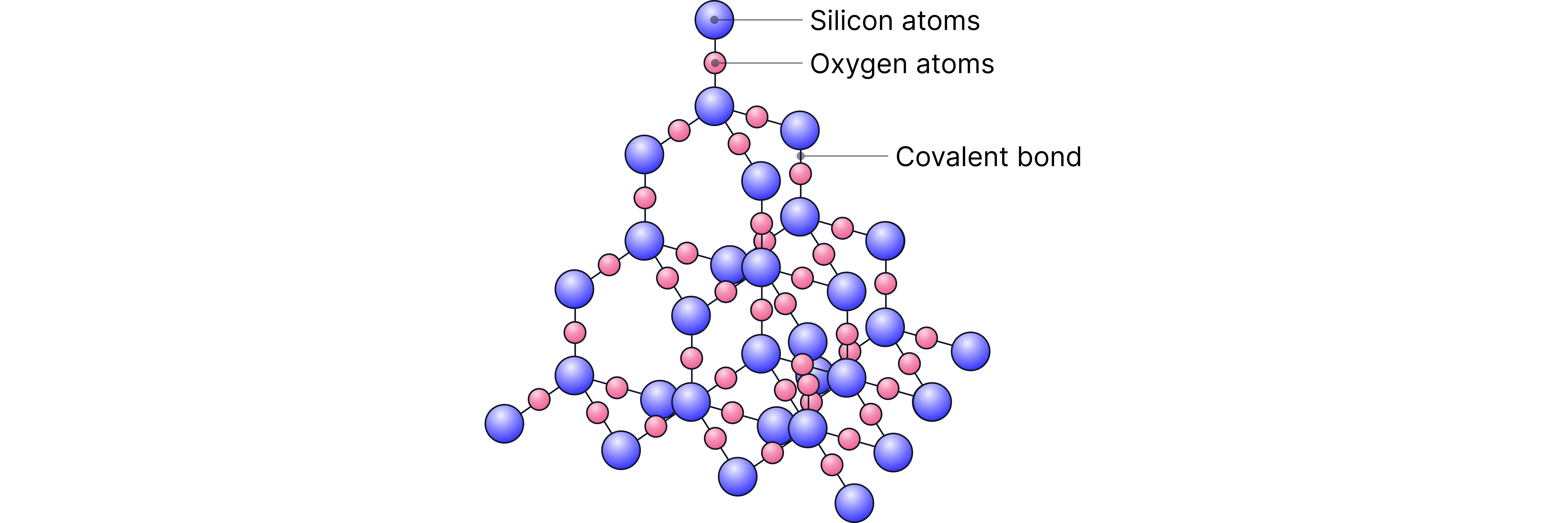
Silicon forms covalent bonds with oxygen where each silicon is covalently bonded to four oxygen atoms in a tetrahedral structure, and each oxygen atom is bonded to two silicon atoms.
This results in a giant covalent lattice (macromolecular) structure where covalent bonds extend in three dimensions.
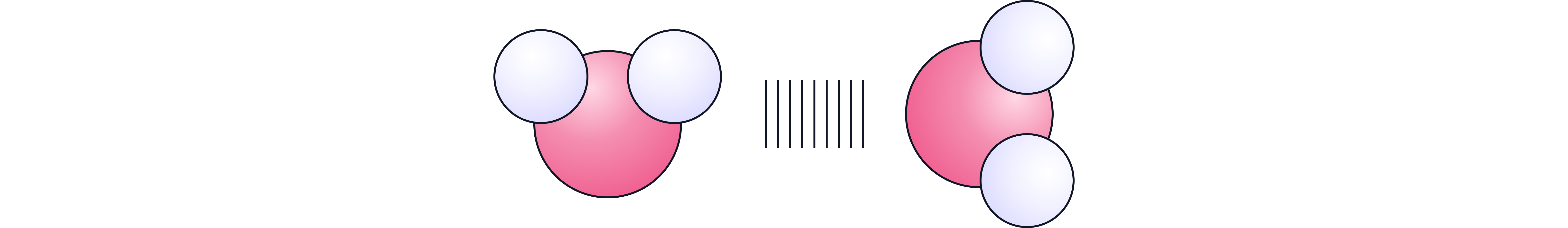
Phosphorous and sulfur form covalently bonded non-metal oxides where atoms share pairs of electrons forming small molecules.
These molecules have weak forces between them (intermolecular forces). The impact of the strengths of these intermolecular forces on melting points is covered in the `Melting Point Trends` note.
Structure and Bonding Summary
Oxide | Bonding | Structure |
Na₂O | Ionic | Giant ionic |
MgO | Ionic | Giant ionic |
Al₂O₃ | Ionic with covalent character | Giant ionic |
SiO₂ | Covalent | Giant covalent (macromolecular) |
P₄O₁₀ | Covalent | Simple molecular |
SO₂ | Covalent | Simple molecular |
SO₃ | Covalent | Simple molecular |
Practice Questions
Question 1
Write an equation for the reaction of phosphorus with oxygen.
Answer:
→
Question 2
Write an equation for the reaction of magnesium with oxygen and state the type of bonding in the oxide formed.
Answer:
→
Bonding: Ionic