Reactions of Benzene
Lajoy Tucker & Dr. Davinder Bhachu
Teachers


Contents
Reactions of Benzene
Benzene reacts according to an electrophilic substitution mechanism
Benzene has a delocalised ring of π-electrons above and below the plane of carbon atoms.
This makes benzene particularly stable
Addition reactions would break this stability, so benzene favours substitution, which preserves the aromatic system.
The π-electrons attack electrophiles. Unlike alkenes, the electron density in benzene is not high enough to react with neutral electrophiles. Generation of a positively charged electrophile is therefore required.
Electrophilic substitution: General mechanism steps
Step 1 – Formation of the electrophile
Benzene reacts with strong electrophiles (positively charged or electron-deficient species),
The electrophile is often generated by a catalyst (e.g. ).
Step 2 – Attack on the ring
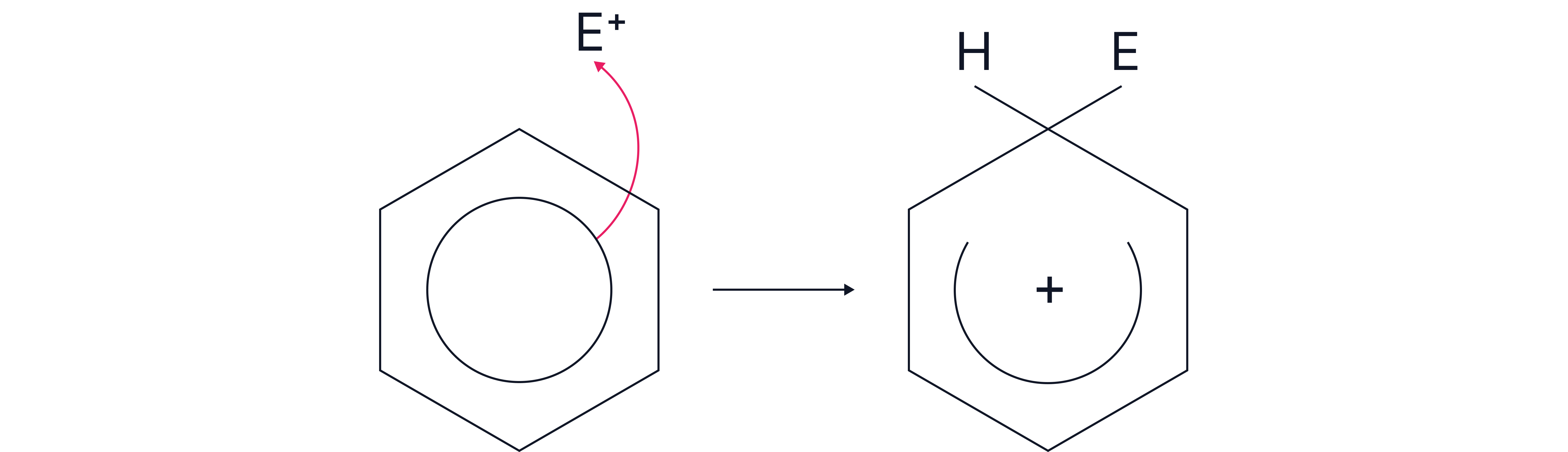
The π-electrons in benzene are attracted to the electrophile.
A new bond forms between one carbon atom and the electrophile.
This breaks the delocalised system temporarily, giving a positively charged arenium ion (carbocation intermediate).
Step 3 – Restoration of aromaticity
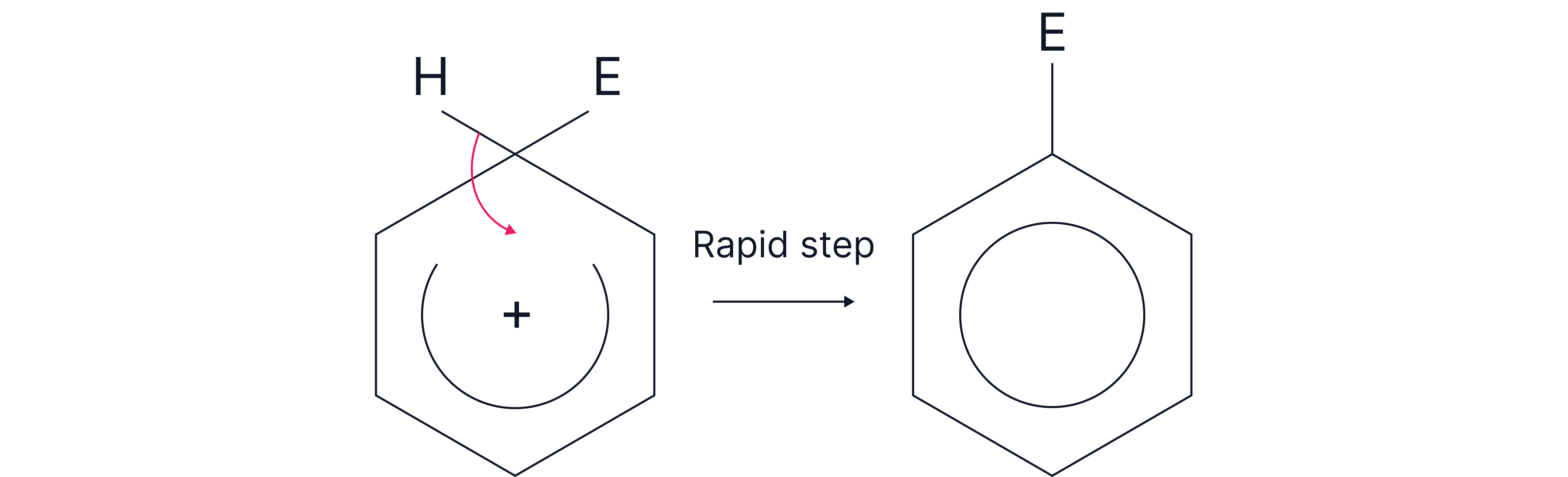
A hydrogen atom is lost from the same carbon.
The electrons from the C–H bond return to the ring, reforming the stable aromatic system.
The catalyst is regenerated if used.
Reaction of Benzene Explained
Nitration
Reaction Overview
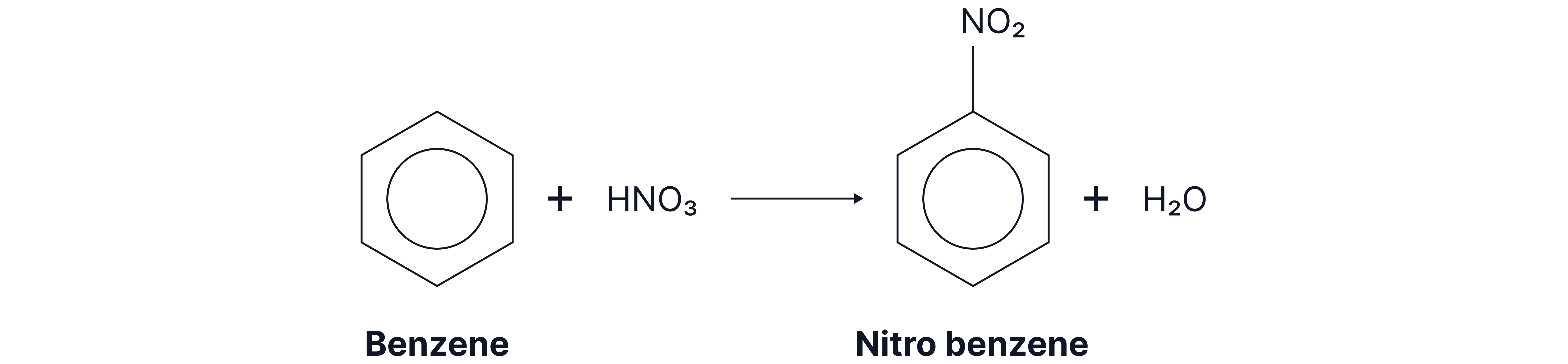
This is done using a mixture of concentrated nitric acid and concentrated sulfuric acid at a temperature of around .
1. Generation of the Electrophile
Concentrated sulfuric acid acts as a catalyst, initially reacting with the nitric acid to generate the nitronium ion :
The ion is the electrophile, which attacks the benzene ring.
2. Mechanism: Electrophilic Substitution
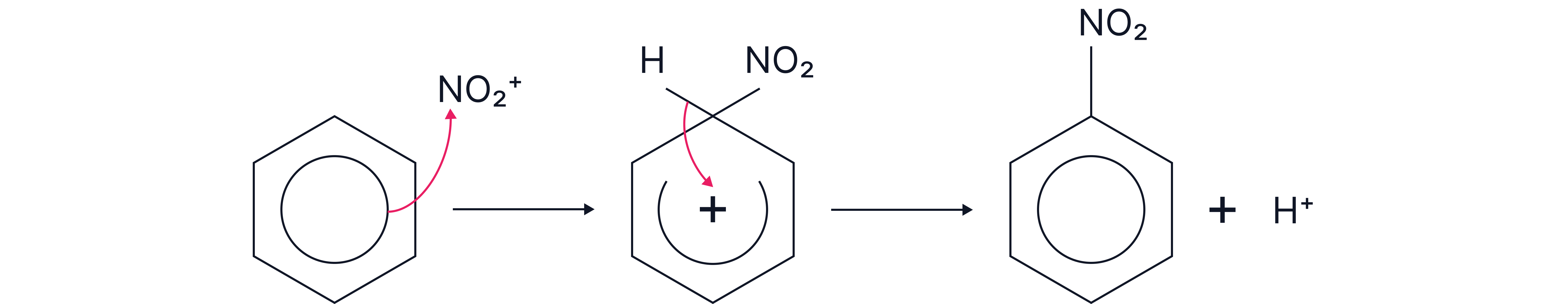
The π-electrons of the benzene ring are attracted to and attack the NO₂⁺ electrophile
A sigma complex (arenium ion) forms with a delocalised positive charge.
A proton is lost from the intermediate.
The aromaticity is restored, and nitrobenzene is formed
3. Regeneration of catalyst
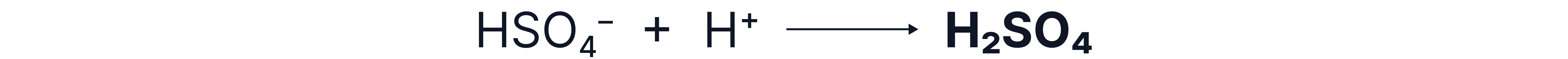
Further reactions:
This is an important first step in the synthesis of aromatic amines. Nitrobenzene is converted to phenylamine through reduction with Sn/HCl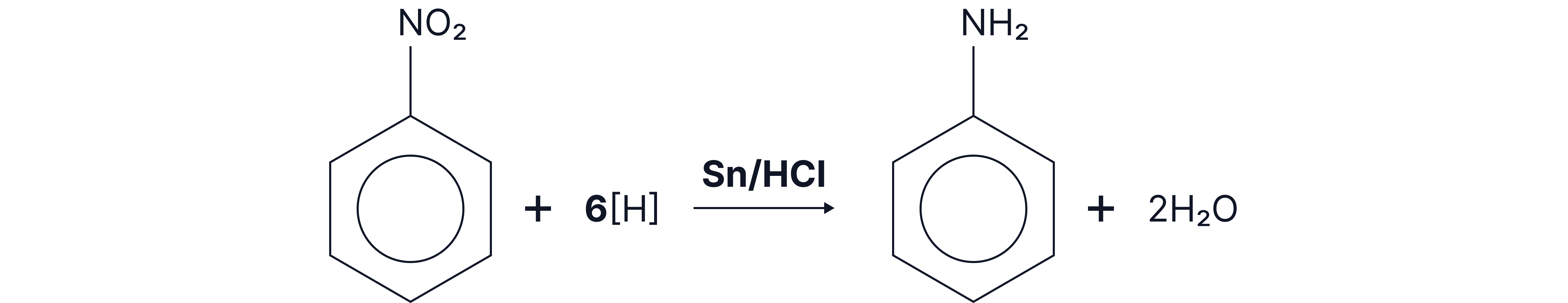
Friedel-Crafts acylation of Benzene
Friedel–Crafts Acylation is a type of electrophilic substitution reaction where an acyl group (RCO-) is introduced onto an aromatic ring
It is usually carried out in the presence of a catalyst such as aluminium chloride
This process is commonly used to synthesise aromatic ketones
Reaction overview:
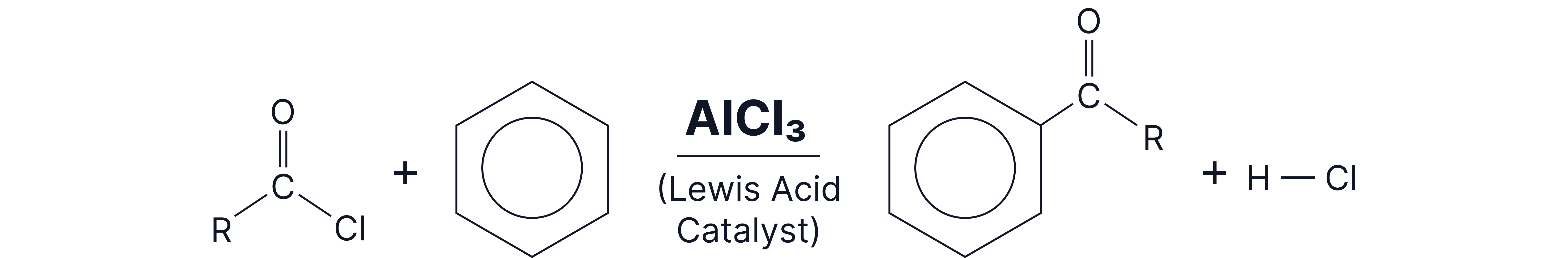
1. Generation of the electrophile -
To carry out this reaction, we need an electrophile (RCO⁺) to attack the benzene ring.
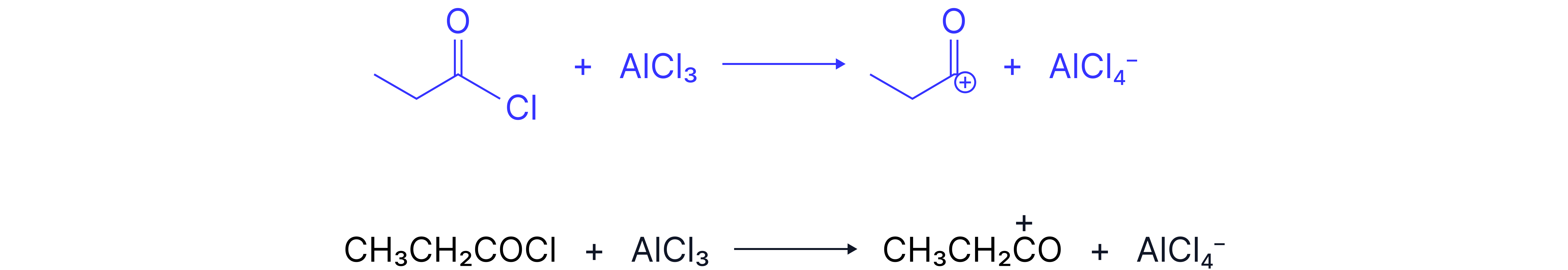
The electrophile is created using an acyl chloride and a halogen carrier, aluminium chloride:
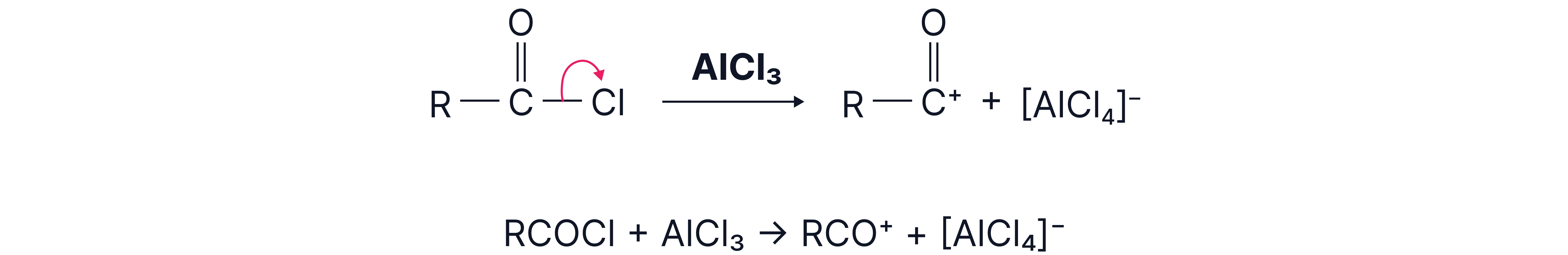
The electrophile is a carbocation
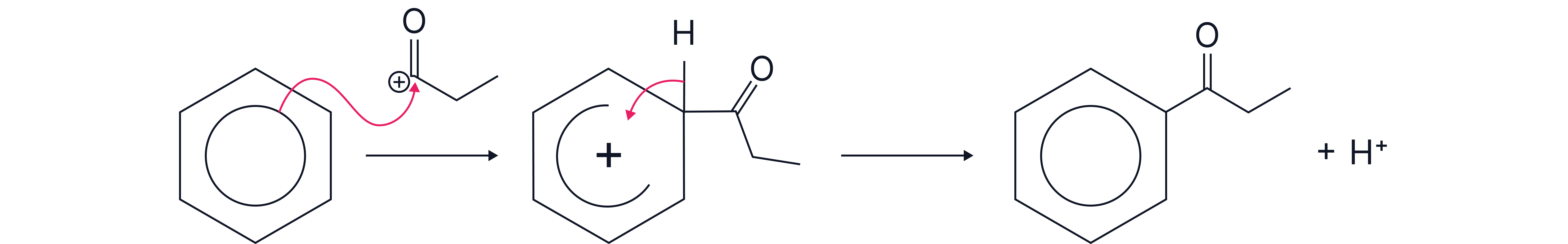
2. Mechanism - Electrophilic Substitution
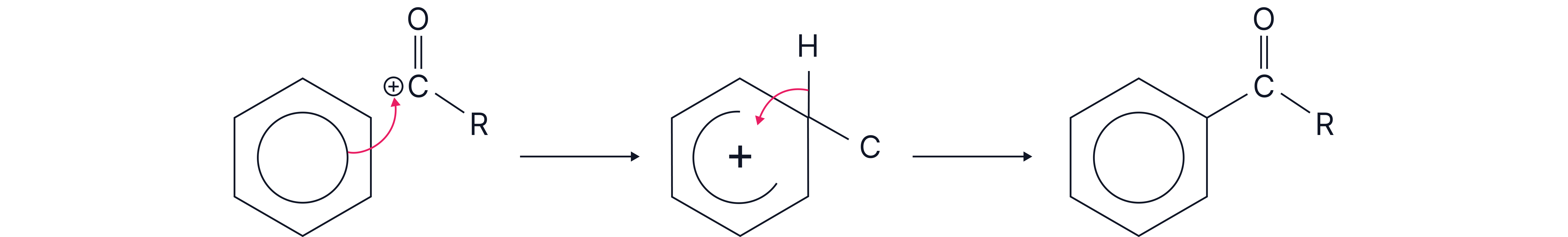
The π-electrons of the benzene ring are attracted to and attack the RCO⁺ electrophile
A sigma complex (arenium ion) forms with a delocalised positive charge.
A proton is lost from the intermediate.
The aromaticity is restored, and the aromatic ketone is formed.
3. Regeneration of catalyst
Further reactions:
The product can then go on to react as a ketone in nucleophilic addition reactions such as addition of hydrogen cyanide and reduction to a secondary alcohol using .
Question 1:
Write the equation for the Friedel–Crafts acylation of benzene using ethanoyl chloride as the acylating agent.
Answer:
Question 2:
Name and outline the mechanism for the reaction of benzene with propanoyl choride. Include an equation for the generation of the electrophile from propanoyl chloride and an appropriate catalyst.
Answer: