Structure of Benzene
Lajoy Tucker
Teacher

Contents
Structure of Benzene: Introduction
Benzene has the formula
Its basic structure is six C atoms in a hexagonal ring, with one H atom bonded to each C atom.
The molecule is planar.
Kekulé made a significant breakthrough and was the first chemist to realise that benzene had a ring structure with six carbon atoms each joined to one hydrogen atom.
However, he thought incorrectly that the ring contains three C=C double bonds and three C-C single bonds.
This molecule would be a "triene" ("cyclohexa-1,3,5-triene") with three C=C double bonds rather than a delocalised ring system.
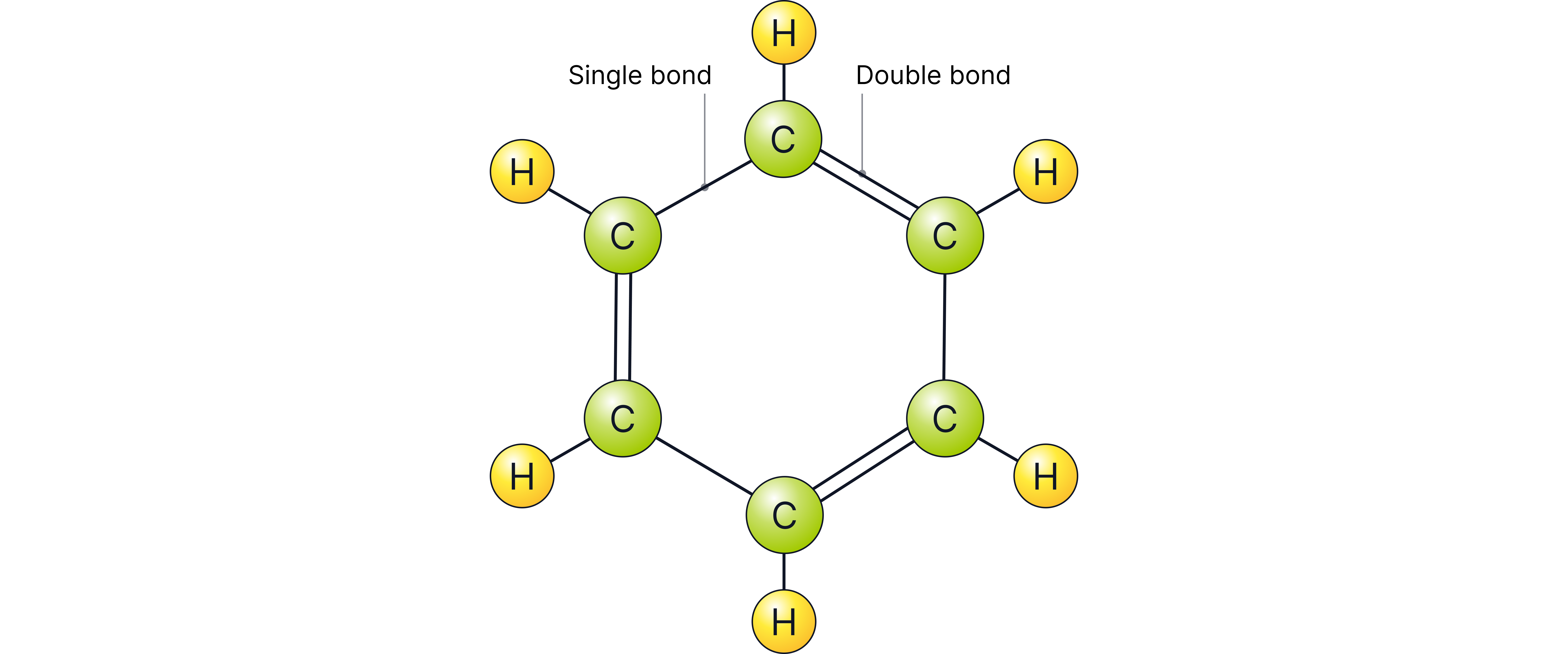
The six C-C bonds are actually the same length – intermediate between single and double.
Each C atom is bonded to two other C atoms and one H atom by single covalent σ-bonds.
This leaves one unused electron on each C atom in a p orbital, perpendicular to the plane of the ring.
Each p orbital overlaps with the neighbouring p orbitals to form a π-bond.
The overall result is a ring of negative charge ("electron cloud") above and below the plane of the ring.
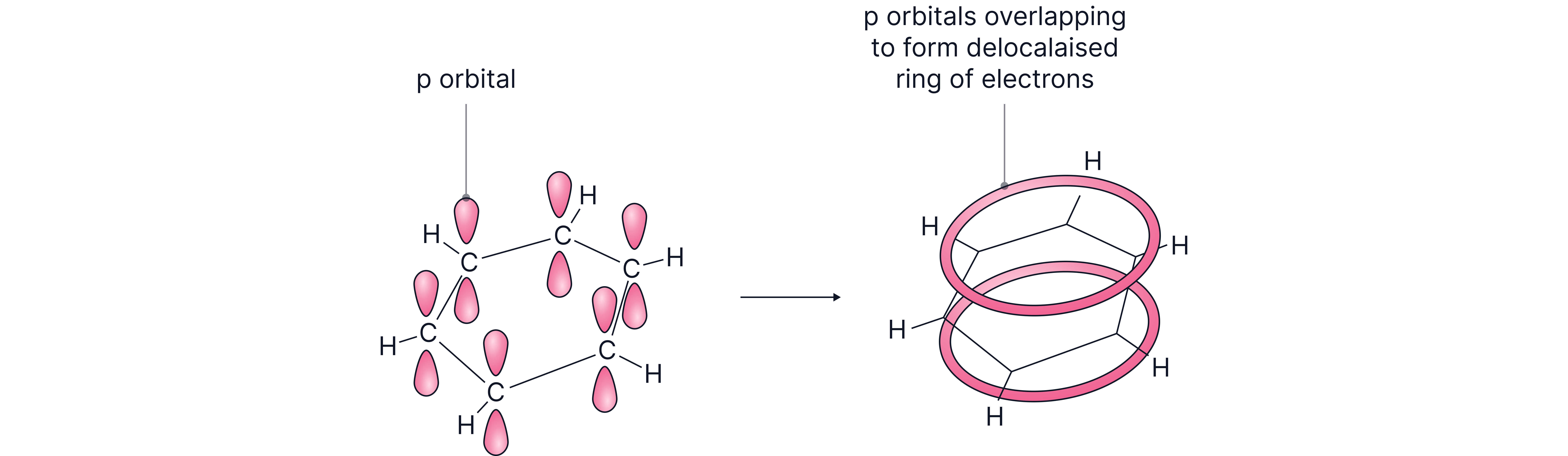
The electrons in the π system do not belong to any particular C atom (or to a bond between two C atoms) – they are free to move throughout the whole π system – i.e. they are delocalised.
As the electrons are delocalised and more spread out, there will be less electron-electron repulsion which makes the molecule more stable.
Due to this delocalisation, the structure of benzene is represented with a circle in the middle of the structure.
There are some key pieces of evidence to support the delocalised structure rather than the Kekulé structure.
1. C-C Bond Length
All the C-C bonds are the same length; and this length is in-between the length of C-C single and C=C double bonds.
C-C single bond:
C=C double bond:
Benzene C-C bonds:
If benzene was a triene we would expect three longer C-C single bonds and three shorter C=C double bonds. Kekulé’s proposed structure would be planar but result in a hexagon that was irregular which is not the case in benzene as all bond lengths are equal.
2. Addition Reactions
Benzene does not readily undergo addition reactions (e.g. benzene does not decolourise bromine water)
If benzene was a triene, we would expect it to readily undergo addition reactions such as this – but it doesn't.
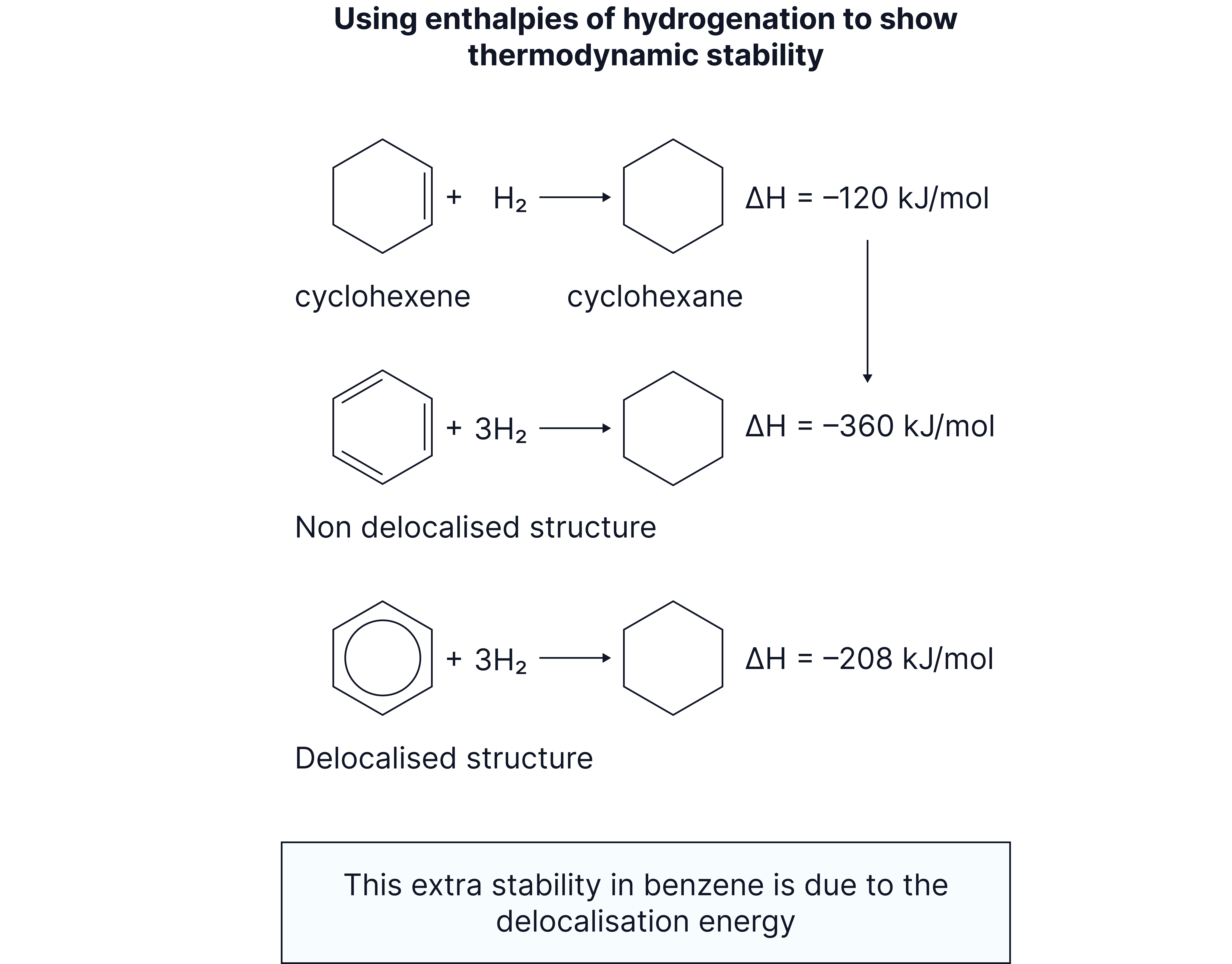
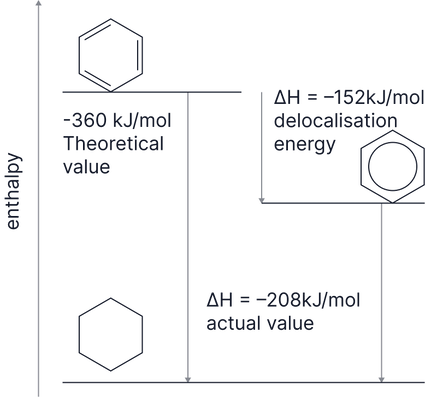
3. Enthalpy of Hydrogenation
When cyclohexene reacts with H₂ to make cyclohexane, the enthalpy change is
When cyclohex-1,3-diene reacts with 2 to make cyclohexane, the enthalpy change is
We might expect a triene to react with 3 to make cyclohexane with an enthalpy change of (i.e. 3 × )
However, the enthalpy change is which is less exothermic than we might expect.
This means that benzene is more stable than the hypothetical triene molecule.
This extra stability is due to the delocalisation of electrons and is known as the delocalisation stability.

Structure of Benzene Explainer Video
Naming Aromatic Compounds
Mono-substituted derivatives of benzene
Aromatic compounds are named in one of three ways:
As benzene with substituents on the ring
Benzene has the highest priority functional group
Short alkyl substituents (methyl, ethyl)
Halogens or other simple substituents
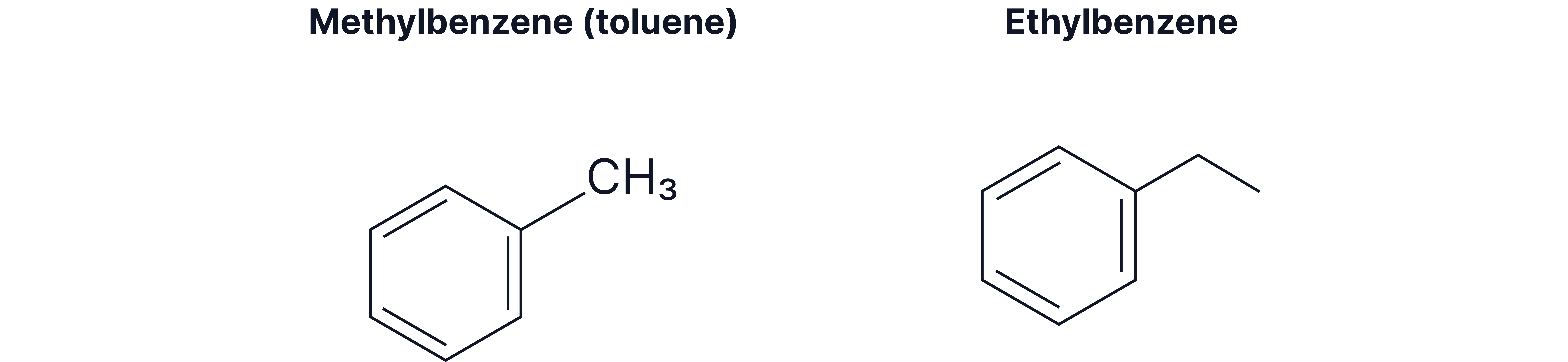
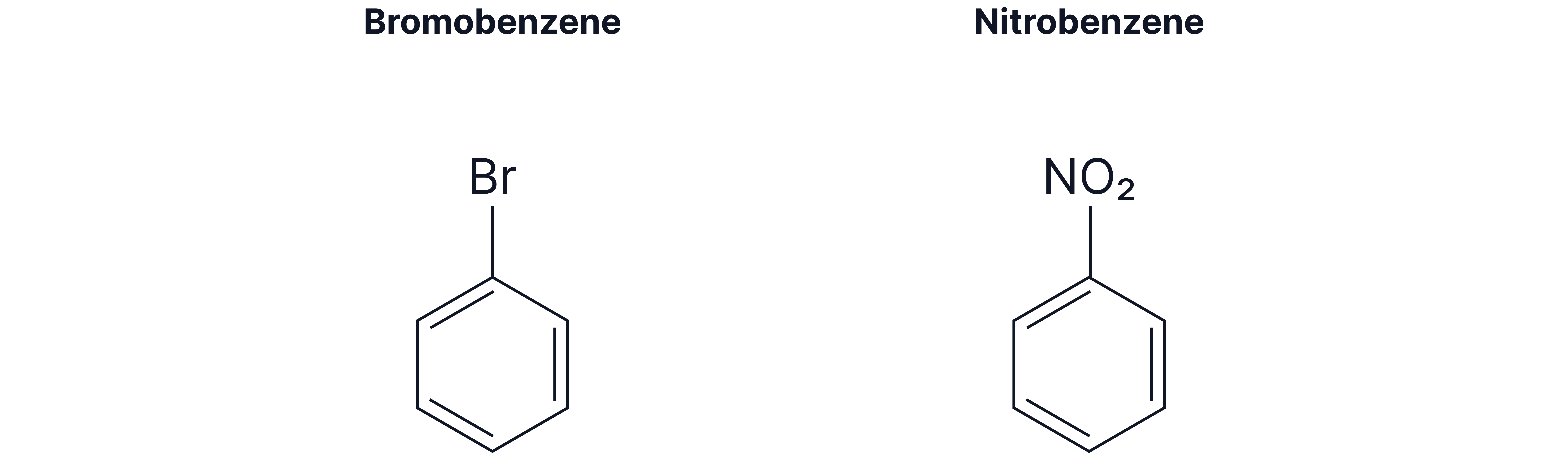
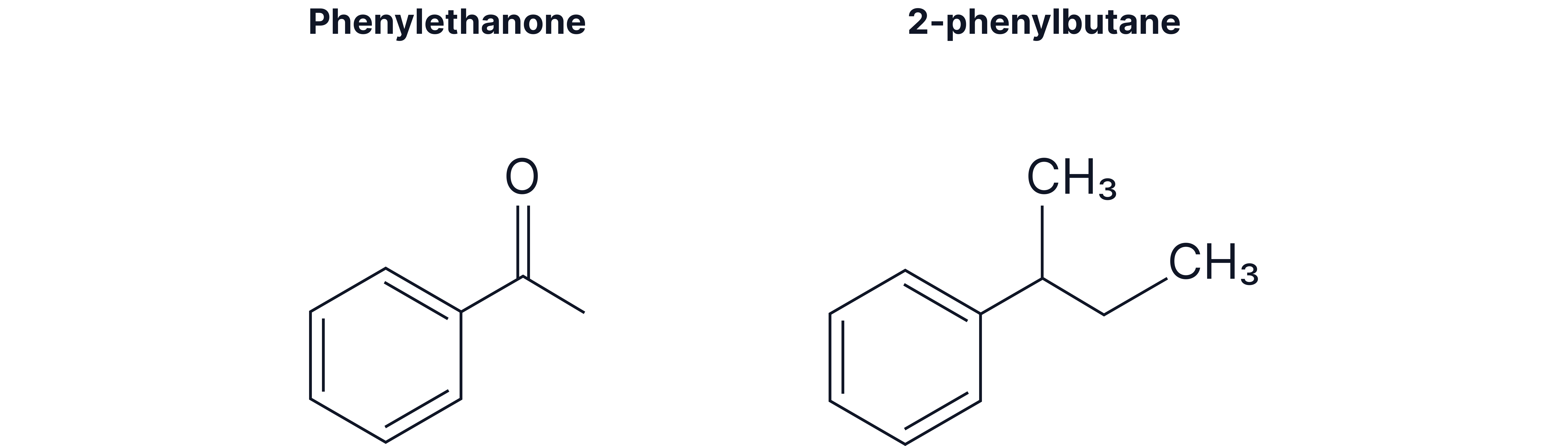
Compounds containing a phenyl group (where is a phenyl group)
Benzene becomes a phenyl substituent () when attached to longer carbon chains or higher priority groups:
Longer alkyl chains (4+ carbons typically)
Multiple functional groups where another takes priority
When benzene is clearly the "side chain"
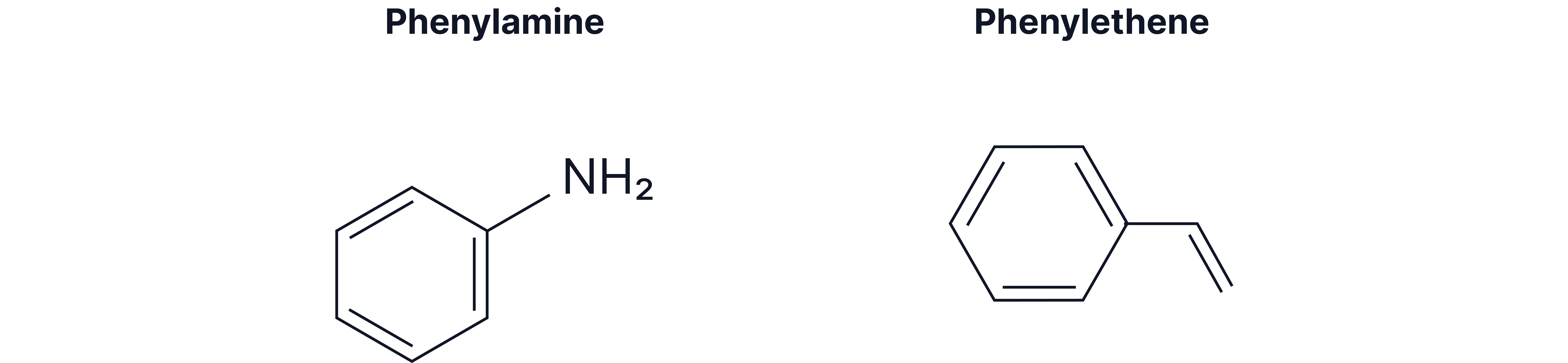
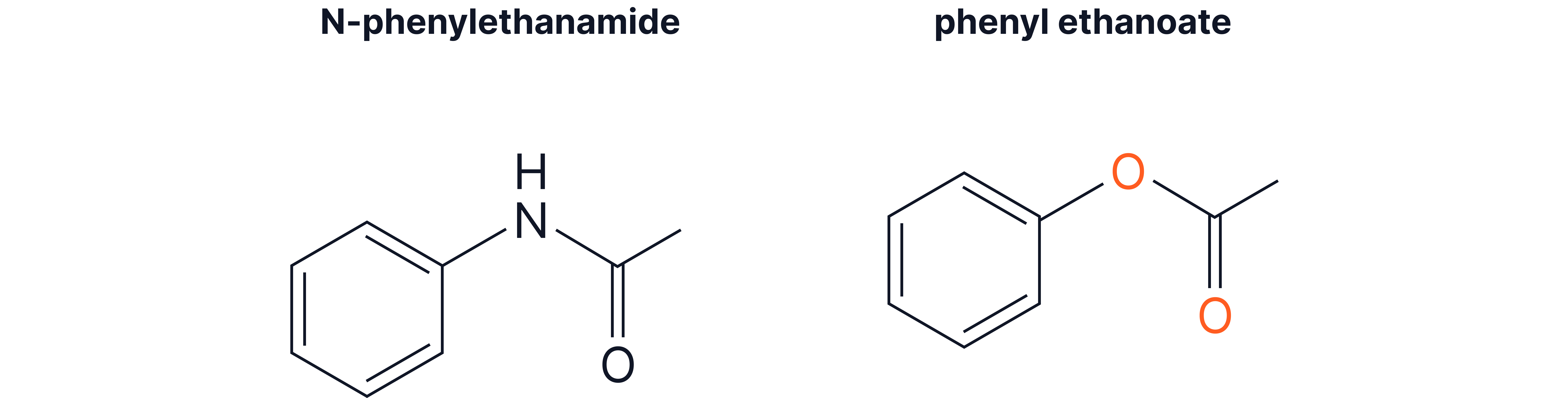
Derivatives of Benzene with more than one substituent
If the groups are all the same
Position of one of them is taken as C-1
Ring is counted in the direction to give the substituents the lowest numbers
Examples:
1,3-dimethylbenzene, 1,2,4-tribromobenzene, benzene-1,4-diol
If the groups are different
Choose the parent compound (giving the position of this substituent C-1)
Add the substituent(s) to that name counting in the direction to give the substituents the lowest numbers
When choosing which group determines the parent compound, choose the most significant group
Substituents are listed in alphabetical order
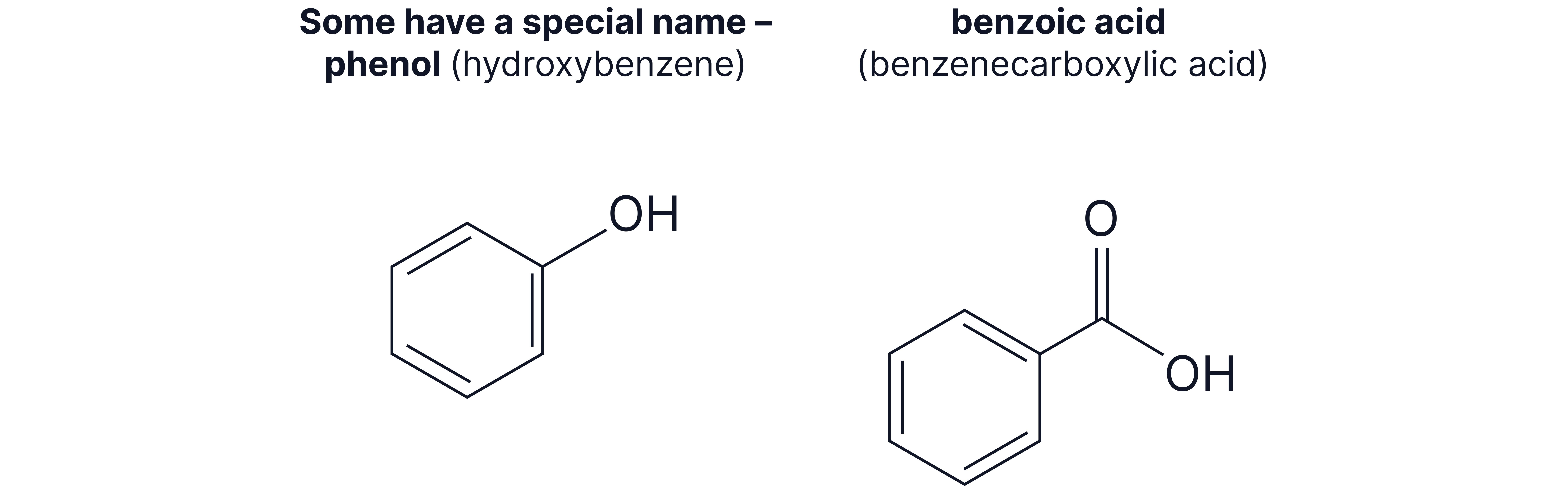
Example 1: 2-methylphenol
Parent compound: phenol (OH group has higher priority than )
Numbering: OH at position 1, at position 2
Name: 2-methylphenol (not 2-hydroxytoluene)
Example 2: 3-nitrobenzoic acid
Parent compound: benzoic acid (COOH has higher priority than )
Numbering: COOH at position 1, at position 3
Name: 3-nitrobenzoic acid
Example 3: 2-bromo-4-methylphenol
Parent compound: phenol (OH has highest priority)
Numbering: OH at position 1, gives Br at 2 and at 4
Alphabetical order: bromo comes before methyl
Name: 2-bromo-4-methylphenol
Positional terms for disubstituted benzenes
1,2- or ortho- (): adjacent positions
1,3- or meta- (): one position apart
1,4- or para- (): opposite positions
Key Differences From Alkenes
Why benzene doesn't behave like a triene?
Alkenes readily undergo addition reactions
Benzene resists addition reactions and prefers substitution reactions
Addition would destroy the delocalised π system and lose the stabilisation energy
Substitution preserves the aromatic system and maintains stability
Evidence Summary
Property | Kekulé Prediction | Actual Benzene | Conclusion |
|---|---|---|---|
Bond lengths | Alternating and | All bonds | Delocalised |
Reactivity | Addition reactions readily | Substitution preferred | Aromatic stability |
Stability | Same as triene | extra stable | Delocalisation energy |
Shape | Hexagonal | Hexagonal | Correct prediction |
Summary
Key Facts about Benzene structure
Formula:
Geometry: Planar hexagon, bond angles
Bond lengths: All C-C bonds (identical)
Hybridisation: carbons with unhybridised p orbitals
system: 6 delocalised electrons
Stability: delocalisation energy
Evidence for Delocalisation
1. Equal bond lengths ()
2. Unreactive to addition (doesn't decolourise water)
3. Exceptional stability ( stabilisation energy)
4. NMR evidence (all H equivalent, all C equivalent)
5. X-ray crystallography (perfect hexagonal symmetry)
Chemical Consequences
Substitution reactions preferred over addition
Electrophilic aromatic substitution is the main reaction type
Catalyst required for addition reactions (destroy aromaticity)
Stable to oxidation compared to alkenes