RP1 - Making a Volumetric Solution
Dr. Davinder Bhachu
Teacher

Introduction and Definitions
Standard solution: A solution of known concentration.
Titration: A technique used to determine the concentration of a solution by reacting it with another solution of known concentration.
Basic Principles
Titrations and many quantitative analytical techniques rely on accurately prepared solutions. This practical develops precision and care in solution preparation, which are skills frequently assessed in Paper 3.
Step-by-Step Method
Step 1. Calculate the Required Mass
Use the formula:
Mass = Concentration x Volume x Mr
Ensure:
Volume in
accounts for the hydrated form if present (e.g., )
Step 2. Weighing the Solid
There are multiple appropriate methods to obtain an accurate mass of a solid
Option a – Weighing by difference (Weigh- Reweigh)
1. Place the weighing boat on the pan of a 2 d.p. digital balance and zero the balance.
2. Place the approximate calculated mass of the solid into the boat.
3. Remove from the balance.
The steps above ensure you have the approximate mass of the desired solid. The following steps allow an accurate mass to be recorded.
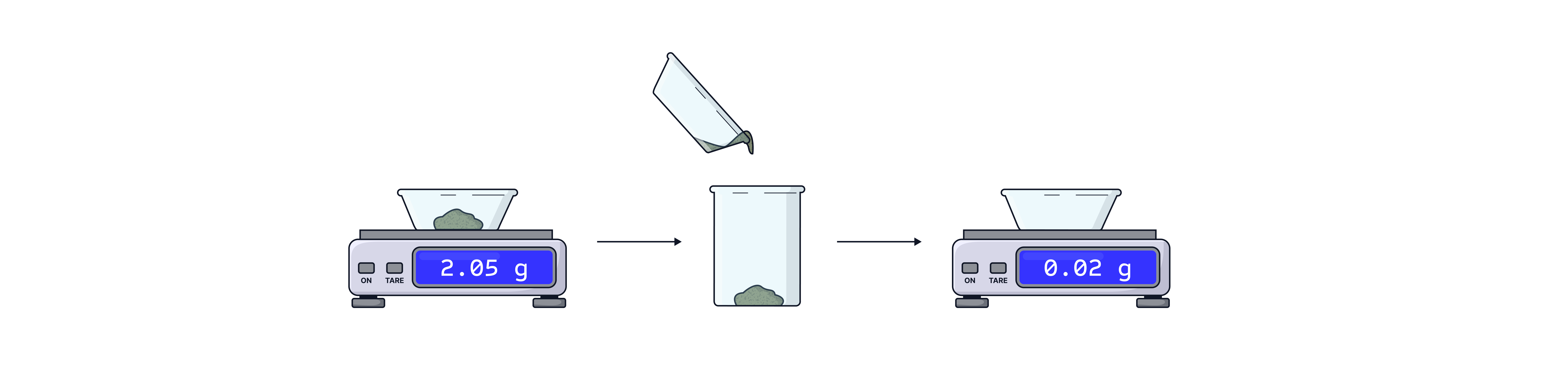
4. Set the balance to zero and weigh the weighing boat and its contents record this value.
5. Tip the contents of the weighing boat into a beaker
6. Set the balance to zero and reweigh the empty weighing bottle. Record this value
7. Calculate the accurate mass in the beaker by subtracting the second recorded mass from the first (step 4 – step 6)
This method accounts for any solid residue remaining in the weighing boat. It is important that the weighing boat is NOT then washed into the beaker as this would add a greater mass than was recorded.
Option b – Weigh directly into vessel
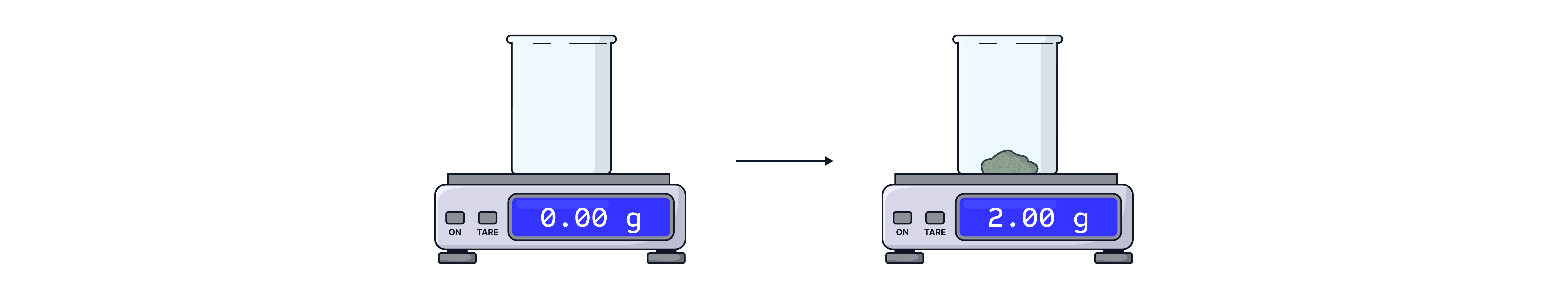
Place an empty beaker onto a 2 d.p digital balance and zero the balance.
Add the desired mass to the beaker. This is the accurate mass to record
Option c – Wash the weighing boat
1. Place an empty weighing boat onto a 2 d.p. digital balance and zero the balance
2. Add the desired mass to the weighing boat. This is the accurate mass to record.
3. Tip the contents of the weighing boat into a beaker
4. Wash any remaining solid from the weighing boat directly into the beaker with distilled water
Step 3. Dissolving the solid
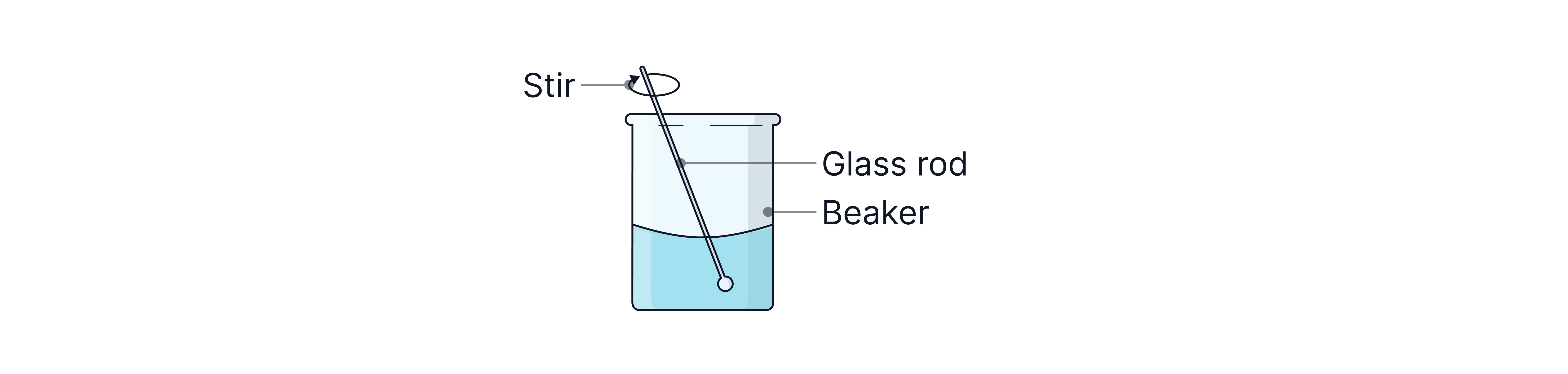
Dissolve the solid in the beaker with distilled water.
Stir with a glass rod to help dissolve the solid
Use a volume of water that is less than the volume of your final solution.
Step 4. Transferring to volumetric flask
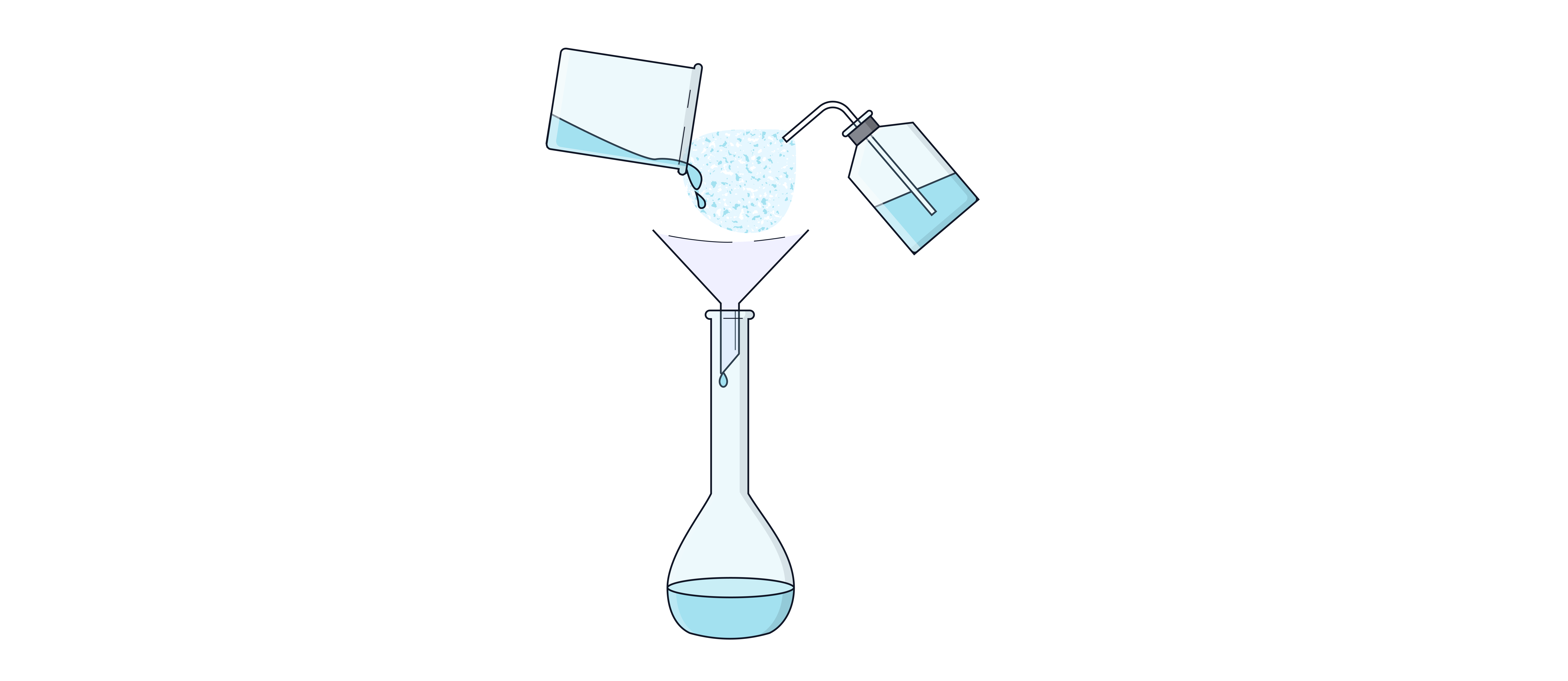
Use a funnel to pour the solution into a volumetric flask of the desired volume e.g.
Rinse the beaker, stirring rod, and funnel into the flask to ensure the mass recorded is the mass in the volumetric flask.
Note – do NOT rinse the weighing boat into the flask unless option 3 was used. The weighing by difference method already accounts for solid remaining in the weighing boat so this can not now be transferred to the flask.
Step 5. Make up to the mark with distilled water
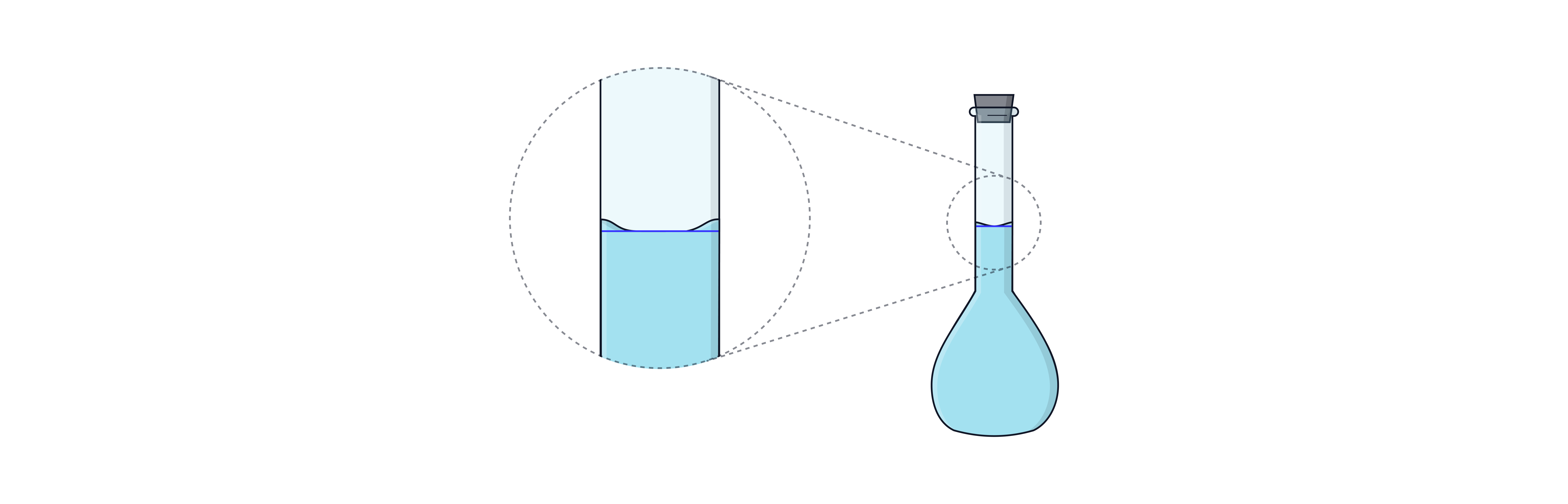
Use a dropping pipette to reach the line exactly if necessary.
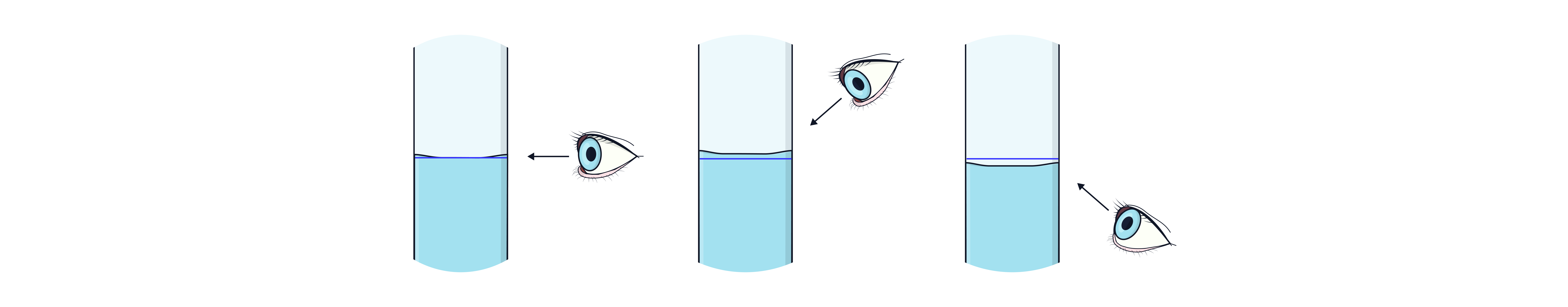
Ensure bottom of meniscus is on the line at eye level.
Step 6. Inversion
Stopper and invert the flask multiple times to mix. And ensure a uniform solution (same concentration throughout).
Worked Examples
Example 1: Calculating Mass
You are asked to make of solution. Calculate the mass of required.
Answer:
Molar mass =
Volume =
Moles needed =
Mass =
Example 2: Common Error
A student prepares a solution but forgets to rinse the funnel into the flask. How might this effect the concentration calculated in the titration?
Answer:
Effect: The actual concentration will be lower than expected as not all solute entered the solution.
Practice Question
Question 1
A student prepares of a solution of potassium hydrogen phthalate (). Calculate the required mass.
Answer:
Question 2
State why it's important to reweigh the weighing boat after transferring the solid.
Answer:
To determine the exact mass of solid transferred, accounting for any residue left in the weighing boat.
Key Tips & Reminders
Use deionised/distilled water only – tap water may contain ions that interfere.
Invert to mix – often missed but crucial for uniform concentration.
Avoid parallax error – view meniscus at eye level.