RP1 - Performing a Titration & Volumetric Analysis
Lajoy Tucker
Teacher

Contents
Introduction and Definition
Titration: A quantitative method used to determine the concentration of an unknown solution by reacting it with a standard solution of known concentration.
Volumetric analysis: Use of volumetric techniques (e.g. A burette) to determine quantities in chemical reactions.
End-point: An observable indication signalling the completion of a chemical reaction e.g. neutralisation.
Basic Principles
Use a pipette to measure a fixed volume of solution with unknown concentration.
Titrate against a standard solution in a burette.
Use a suitable indicator (e.g., phenolphthalein, methyl orange).
Perform repeat titres to ensure accuracy and concordant results.
Apparatus
Volumetric pipette and filler – to measure a fixed volume of solution into a conical flask
Burette – to gradually add measured volumes of a solution to the conical flask during the titration
Burette holder and stand
Conical flask – to hold a known volume of a solution transferred from the pipette
White tile – placed under the conical flask to better visualise the endpoint colour change
Rinsing Glassware
Burette
1. Wash with distilled water to remove and soluble impurities
2. Rinse with the solution that will be placed in the burette to ensure any remaining water does not dilute the solution and affect its concentration
Conical flask
Rinse with distilled water ONLY
(If rinsed with the solution, this would add additional moles to the flask and therefore result in a larger volume of solution from the burette.)
Diagram of titration set-up
Illustration of titration setup showing burette clamped vertically above a conical flask on a white tile.
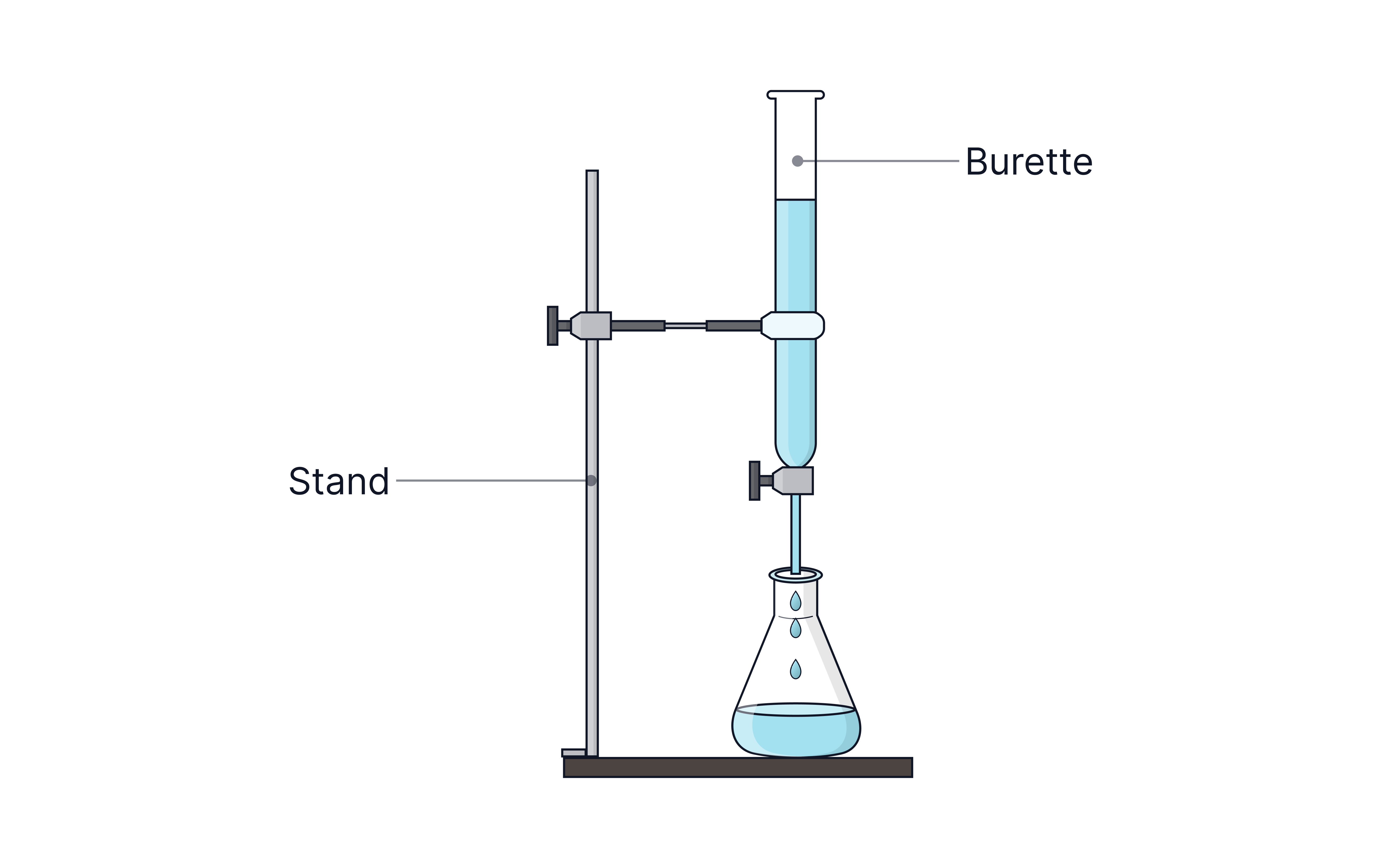
Titration Procedure
|
Step |
Explanation |
|
Fill burette with the standard solution using a funnel |
Minimises spillage. Bring the burette closer to the ground if required so as not to pour potentially harmful solutions above eye level |
|
Fill the burette tap and remove the funnel |
Ensures that all the solution leaving the burette during the titration is entering the conical flask and is not being used to fill the air in the tap |
|
Record the initial volume in the burette |
Read from the bottom of the meniscus at eye-level. Record all volumes to two decimal places* |
|
Use a pipette to transfer a fixed volume the unknown solution to the conical flask e.g. 25cm3. |
Use the pipette filler to fill the pipette to where the bottom of the meniscus is on the mark. Use a pipette of the volume required for the titration. |
|
Add 2–3 drops of suitable indicator to the conical flask |
The indicator will change colour permanently when the reaction is complete e.g. neutralisation |
|
Place the conical flask on a white tile |
This makes the colour change more clearly visible |
|
Slowly add the solution from the burette to the conical flask, swirling as added |
Swirling allows the solutions to mix and react completely |
|
Stop adding the solution when a permanent colour change is observed (endpoint) |
The indicator may change colour in spots before the reaction is complete due to not the solution from the burette being unevenly distributed. With swirling, when the solution changes colour permanently, the reaction is complete |
|
Record the final volume |
This allows calculation of the titre (volume added) as ‘final volume – initial volume’ |
|
Repeat above steps this time adding the solution dropwise as the endpoint is approached |
This allows an accurate titre to be determined |
|
Repeat until two concordant titres are achieved |
Titres within 0.10cm3 of eachother |
Selecting an Indicator
|
Indicator |
Colour in Acid |
Colour in Alkali |
Suitable for… |
|
Methyl Orange |
Red |
Yellow |
Strong acid/base |
|
Phenolphthalein |
Colourless |
Pink |
Weak acid/strong base |
Recording Volumes
A suitable results table is set out below
Notice that all volumes are recorded to two decimal places. When reading the volume from a burette with intervals of 0.1 cm3, volume where the bottom of the meniscus is on the mark should end with a ‘0;. Volumes where the bottom of the meniscus is between two marks should end in ‘5’.
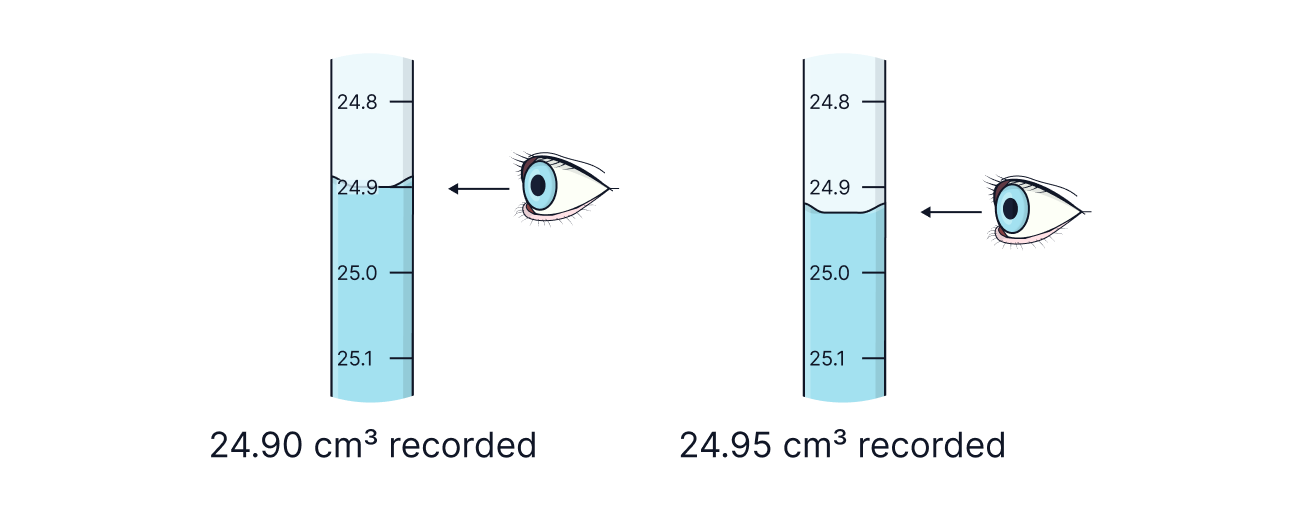
|
Rough |
1st Trial |
2nd Trial |
3rd Trial |
|
|
Final volume (cm3) |
24.95 |
24.60 |
49.40 |
24.50 |
|
Initial volume (cm3) |
0.00 |
0.00 |
24.60 |
0.00 |
|
Titre (cm3) |
24.95 |
24.60* |
24.80 |
24.50* |
Mean Titre
The ‘mean’ or ‘average’ titre is calculated from the concordant results only. These are titres within of eachother.
For example, above, the concordant results are those from trials 1 and 3.
The mean titre is calculated as
Key Equations:
-
Moles = c × V (c in mol dm⁻³, V in dm³)
-
When the indicator changes colour, n(acid) = n(base)
Example 1: Titration Calculation
25.00 cm³ of HCl is titrated with 0.100 mol dm⁻³ NaOH. The mean titre is 26.40 cm³.
Calculate the concentration of HCl.
Answer:
Step 1: Moles of NaOH = 0.100 × 26.40/1000 = 2.64 × 10-3 mol
Step 2: Reaction:
HCl + NaOH → NaCl + H2O
1:1 molar ratio → so moles HCl = 2.64 × 10⁻³ mol
Step 3:
C = n/V = (2.64×10-3) /(25.00×10-5) = 0.1056 mol dm-3
More titration calculation examples can be found in the ‘Reacting Solutions’ note
Percentage Uncertainty
Percentage uncertainty = [(absolute uncertainty x number of readings)/measure value] x 100
Absolute uncertainty is inherent to the measuring equipment itself e.g. a burette’s absolute uncertainty ±0.05cm3.
Number of readings = 2 (initial volume and final volume)
Measured value = titre
Example 1
The uncertainty in each burette reading is ± 0.05cm3. Calculate the percentage uncertainty in a titre of 24.65cm3.
Answer:
Percentage uncertainty = [(0.05 x 2)/24.65cm3] x 100 = 0.41%
Note - A titre requires two burette readings (initial and final volumes).
Example 2
The total uncertainty of a 25.0cm3 pipette is ± 0.06 cm3. Calculate the percentage uncertainty in the volume measured.
Answer:
Percentage uncertainty = [(0.06 x 1)/25.0cm3] x 100 = 0.24%
Reducing Percentage Uncertainty
Using the same apparatus, percentage uncertainty can only be reduced by increasing the measured value (the denominator).
To reduce the percentage uncertainty for a titre, the measured value would have to be greater. This means requiring a larger volume of solution to be added from the burette. This can be achieved by:
Using a larger volume of the solution in the conical flask – this increases the number of moles in the conical flask therefore resulting a greater volume of solution from the burette to react with it
Making up a standard solution of lower concentration for the burette – this means a greater volume of the solution will be needed from the burette to equal the number of moles of the solution in the conical flask
Using a more concentrated solution in the conical flask* – this increases the number of moles in the conical flask therefore resulting a greater volume of solution from the burette to react with it. This may not be possible depending on what is being analysed*
Question 1
Why is it important to rinse the burette with the titrant and not water before filling it?
Answer:
To avoid dilution of the titrant which would lead to an inaccurately low concentration, affecting the titre value.
Question 2
Suggest the effect of this procedural error: The student doesn’t swirl the flask while titrating.
Answer:
Acid/base may not fully mix; colour change delayed, overshooting the end point → titre too high.
Key Tips & Reminders
Rinse all glassware with appropriate solutions, not water.
Take burette readings to 2 decimal places, ending in .00 or .05.
Swirl constantly to ensure complete mixing.
Use only concordant results for calculating mean titre (within 0.10 cm3 of each other).
Avoid parallax error – eye level at bottom of meniscus.