RP6 - Testing for Organic Functional Groups
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Functional group testing involves using specific reagents to detect the presence of key organic functional groups based on observable chemical changes.
The groups tested in this required practical are:
Alcohols
Aldehydes
Alkenes
Carboxylic acids
Haloalkanes
Each test provides a visible observation, making it suitable for qualitative identification.
Basic Principles
Each organic compound has a distinctive functional group that reacts with certain reagents.
Colour change, gas evolution, or precipitate formation provides evidence for the presence of a functional group.
Functional Group Tests
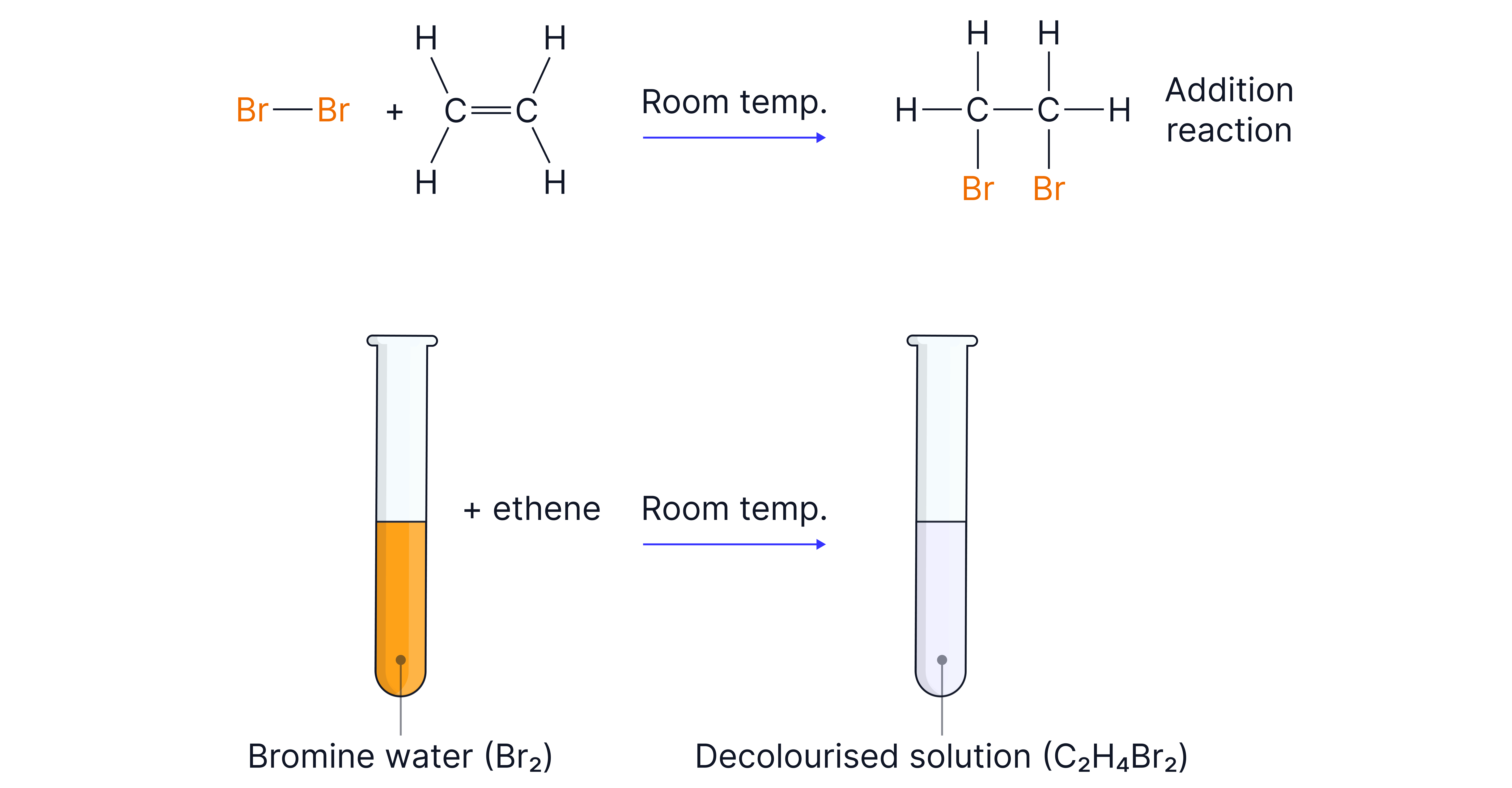
1. Test for Alkenes – Bromine Water
Reagent: Orange bromine water
Observation: Bromine water decolourises (orange → colourless)
Reaction: Electrophilic addition to the double bond
2.Test for Aldehydes – Tollens’ Reagent or Fehling’s Solution
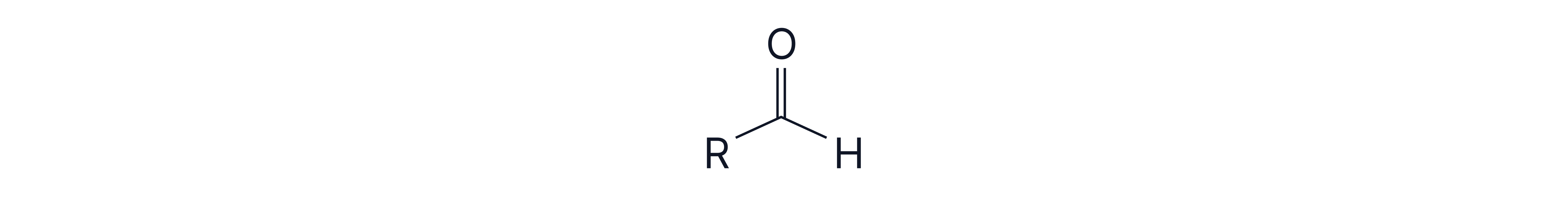
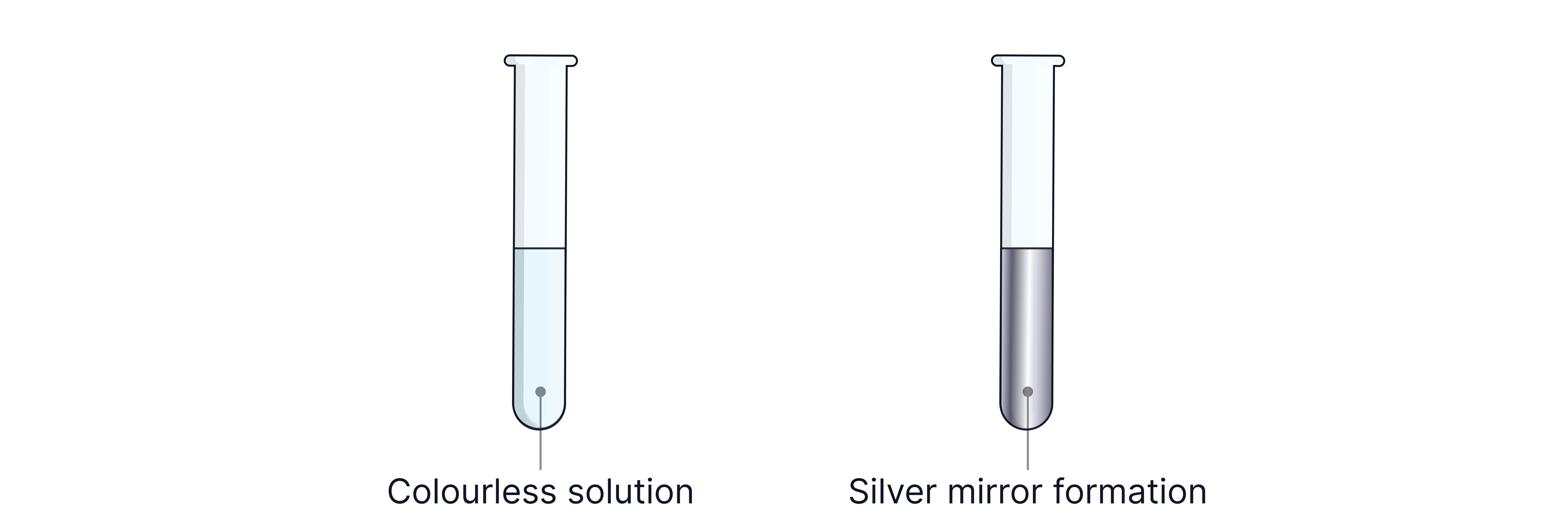
a) Tollens' Reagent
Reagent: Ammoniacal silver nitrate
Observation: Silver mirror forms
Reaction: Aldehyde is oxidised to a carboxylic acid; is reduced to
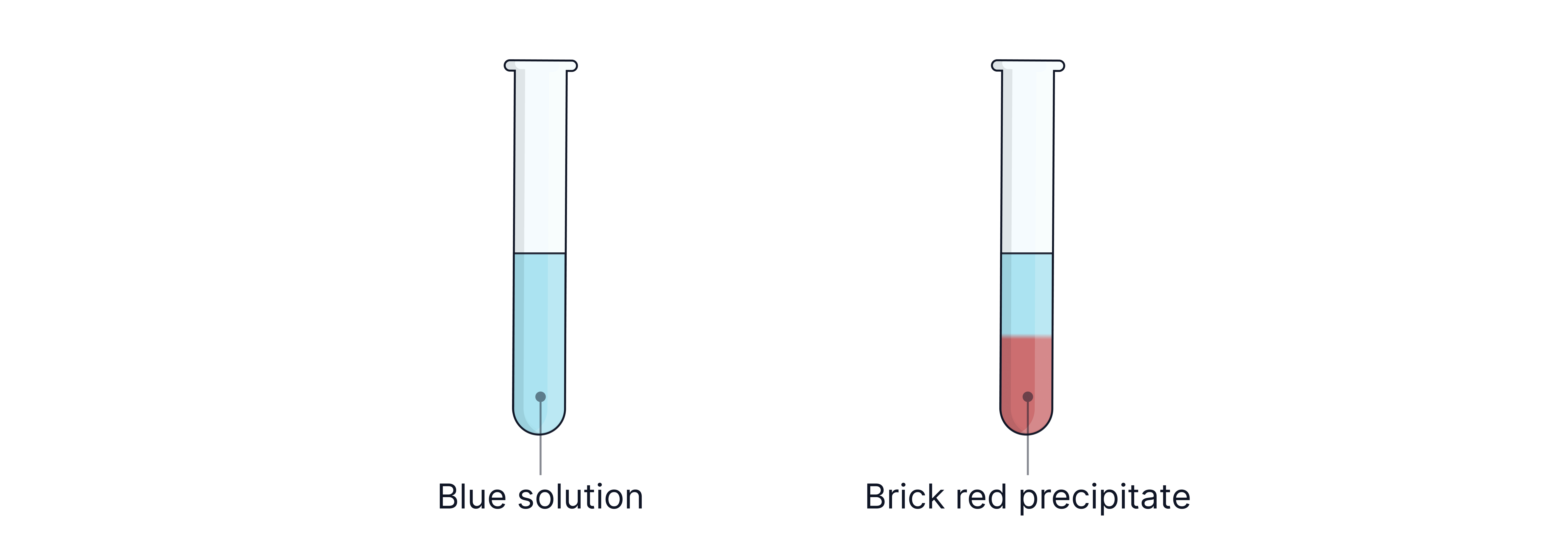
b) Fehling’s Solution
Reagent: Copper(II) solution + alkali (heat)
Observation: Blue solution → brick-red precipitate (change in colour AND state both required)
Reaction: reduced to
Note: Tollen’s Reagent & Fehling’s Solution test only for aldehydes, not
ketones as ketones can not be further oxidised
3. Test for Alcohols – Acidified Potassium Dichromate
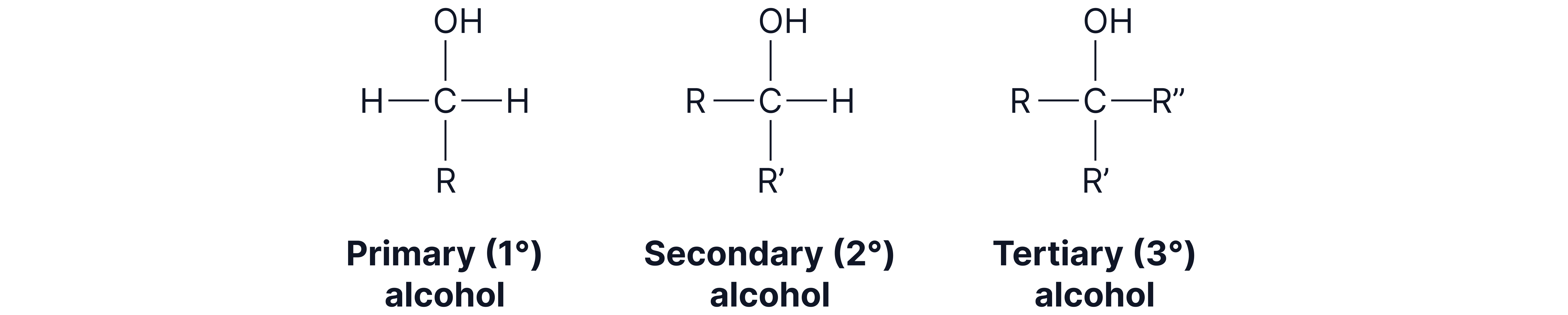
Reagent: Acidified (orange solution)
Observation:
Primary and secondary alcohols: Orange solution→ green solution( formed)
Tertiary alcohols: No observable change (tertiary alcohols are not oxidised)
Reaction: Primary and secondary alcohols oxidised, reduced to
Note: as aldehydes can also be oxidised, they will give a positive result with acidified potassium dichromate
4. Test for Carboxylic Acids – Acid + Carbonate
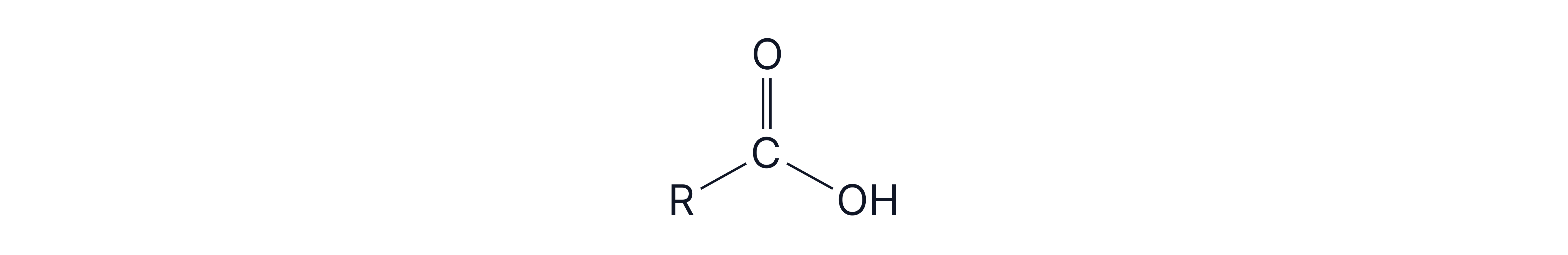
Reagent: Aqueous or
Observation: Effervescence (CO₂ gas evolved)
Confirmation: Bubble gas through limewater → turns cloudy
Reaction:
5. Test for halogenoalkanes – Silver Halide Precipitates
alkyl group
Process
1. Warm the sample with a mixture of sodium hydroxide solution and ethanol
Reaction: Nucleophilic substitution releasing X- ions
2. Add dilute nitric acid followed by silver nitrate solution
Reaction: Precipitation of silver halide
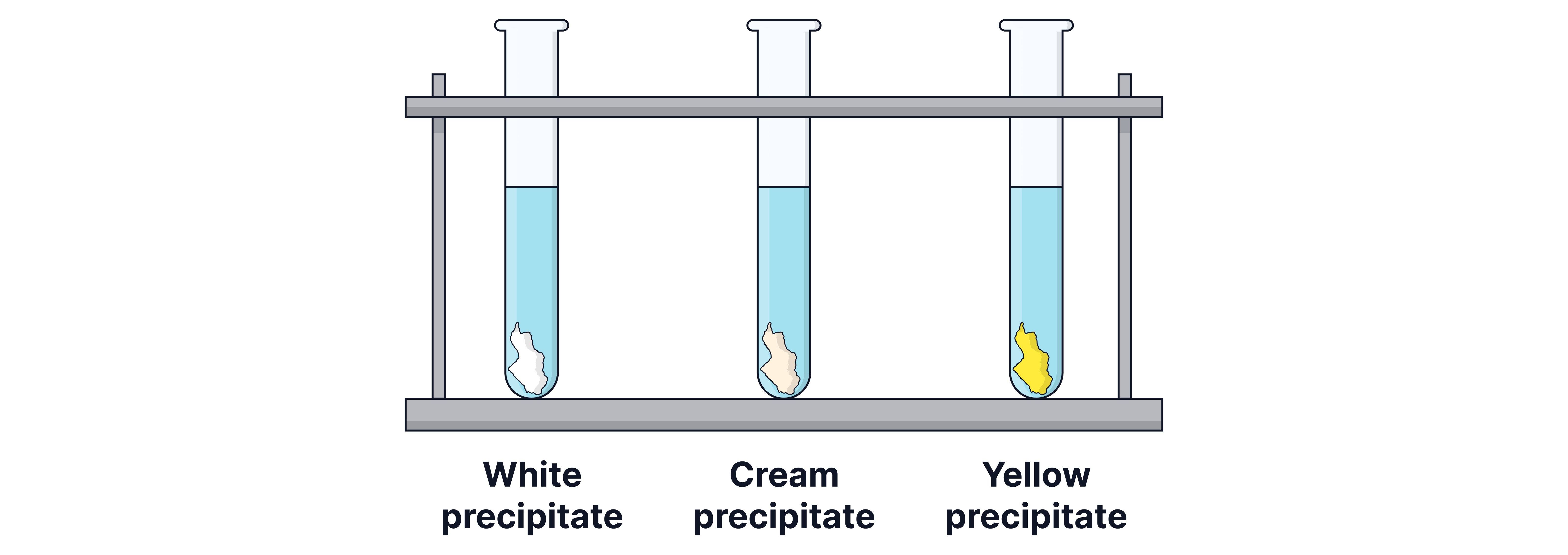
Observation: Precipitate colour determined by identity of X
White ppt , cream ppt = , yellow ppt =
Summary of tests
Functional group | Reagent(s) | Positive Result |
|---|---|---|
Alkene | Bromine water | Orange to colourless solution |
and alcohol | Acidified potassium dichromate | Orange to green solution |
Aldehyde | Tollens’ Fehling’s | Silver mirror forms Blue solution to brick red precipitate |
Carboxylic acid | Sodium carbonate or sodium hydrogencarbonate solution | Effervescence |
Halogenoalkane | in ethanol Acidified silver nitrate solution | white ppt cream ppt yellow ppt |
Key Tips & Reminders
Tollens’ and Fehling’s only work for aldehydes, not ketones.
Tertiary alcohols cannot be oxidised by — no colour change..
Examiner Tip: Many students lose marks for missing observations or failing to name the reagent precisely (e.g. "acidified potassium dichromate", not just "dichromate").