RP5 - Distillation of a Product from a Reaction
Lajoy Tucker
Teacher

Contents
Introduction and Definitions
Distillation is a separation technique used to isolate a liquid product from a reaction mixture based on differences in boiling points.
Reflux is a method used to heat a reaction mixture for a prolonged time without loss of volatile substances, by condensing and returning vapours back to the flask.
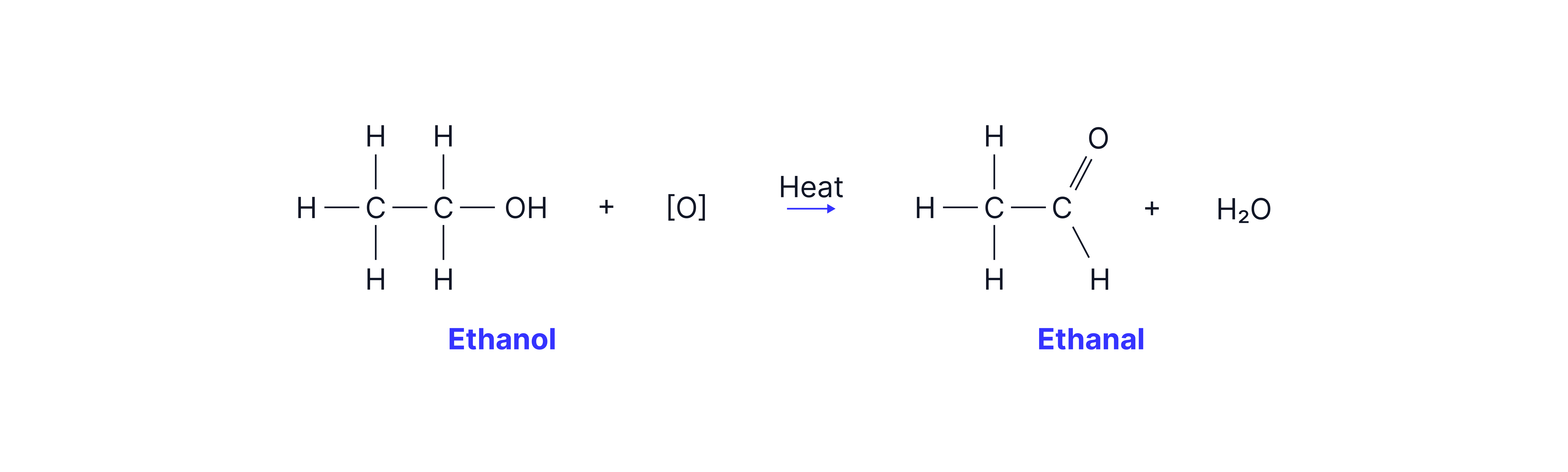
Oxidation of a Primary Alcohol (e.g. Ethanol → Ethanal)
Partial oxidation of ethanol using acidified potassium dichromate (K₂Cr₂O₇/H₂SO₄).
Distillation used to remove ethanal as it forms to prevent further oxidation.
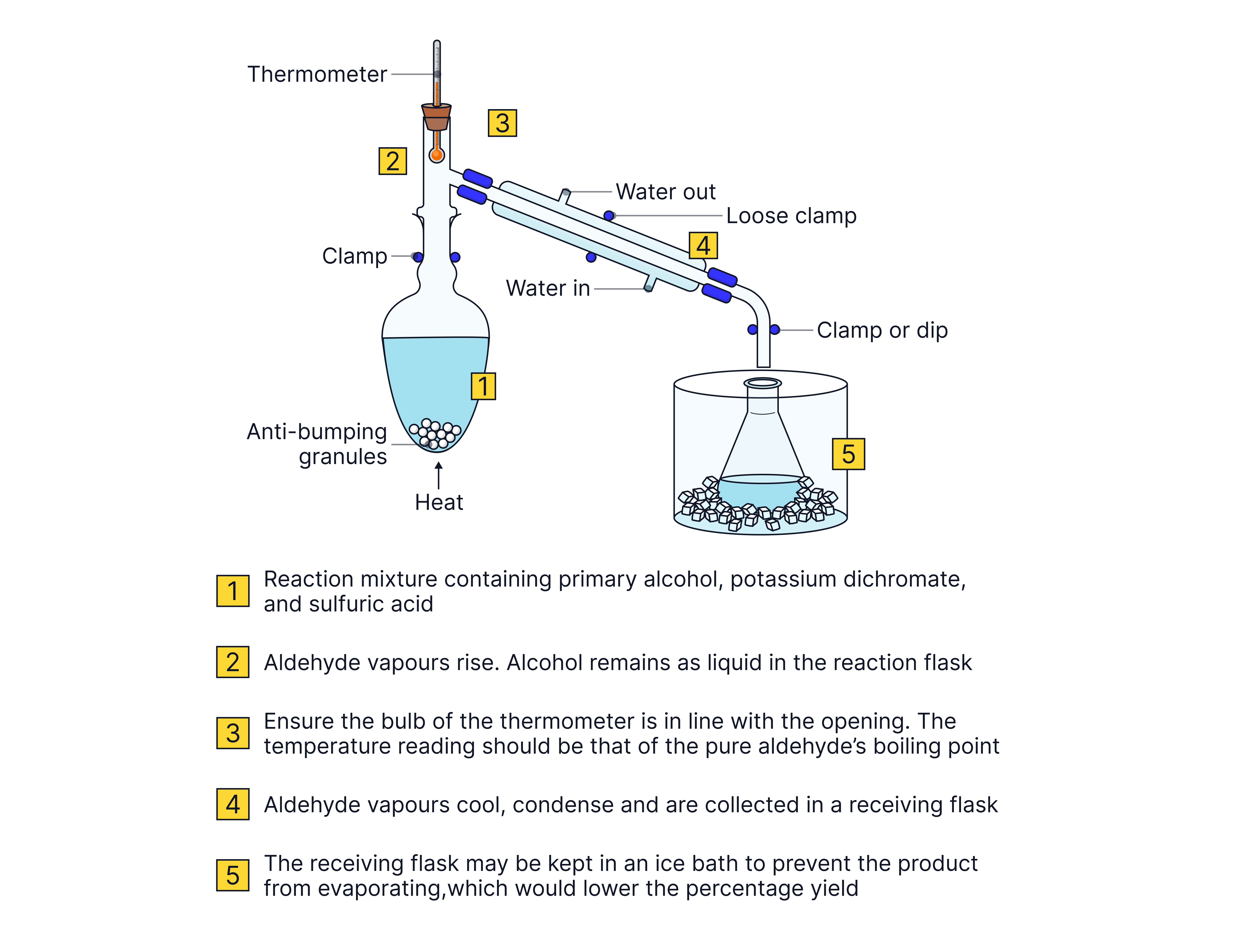
Quickfit apparatus is used to avoid substances leaking from connections between glassware.
The reaction mixture is gently heated the mixture using a water bath or heating mantle. This allows control of the temperature and the safe heating of flammable liquids.
Collect the distillate which boils at the boiling point of the desired product
The aldehyde has a lower boiling point than the alcohol reactant due to the absence of hydrogen bonds between molecules.
Collection of the distillate should stop if the temperature rises more than 2oC above the boiling point of the aldehyde.
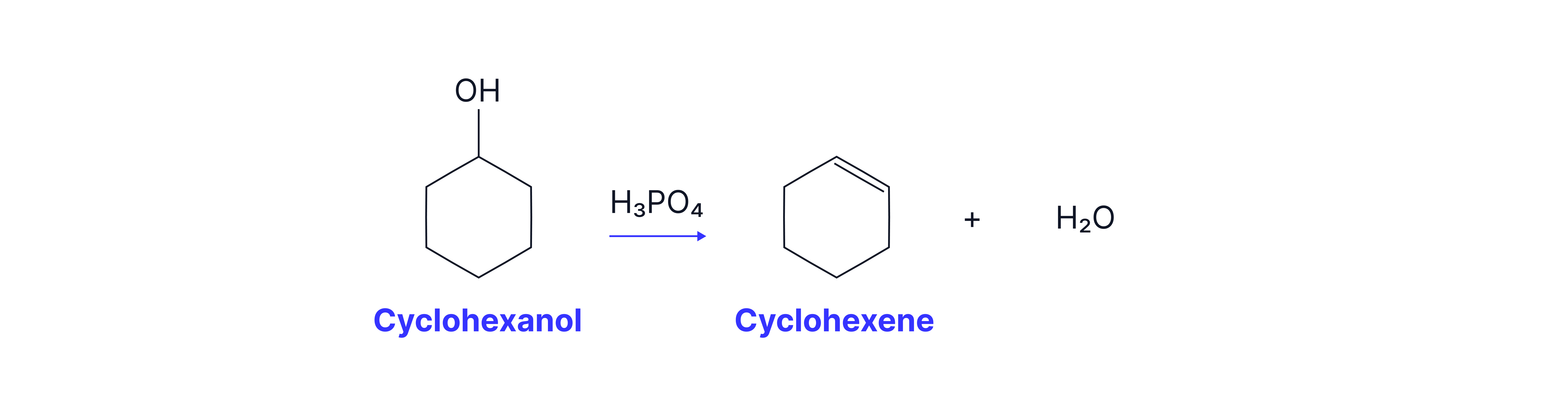
Cyclohexanol → Cyclohexene
Dehydration (elimination of H₂O) using concentrated H₃PO₄ or H₂SO₄ catalyst.
Distillation
Cyclohexene has a lower boiling point than the reactants/by-products and so can be collected as the distillate.
Purification
The cyclohexene product will also contain water and the phosphoric acid catalyst and so requires purification.
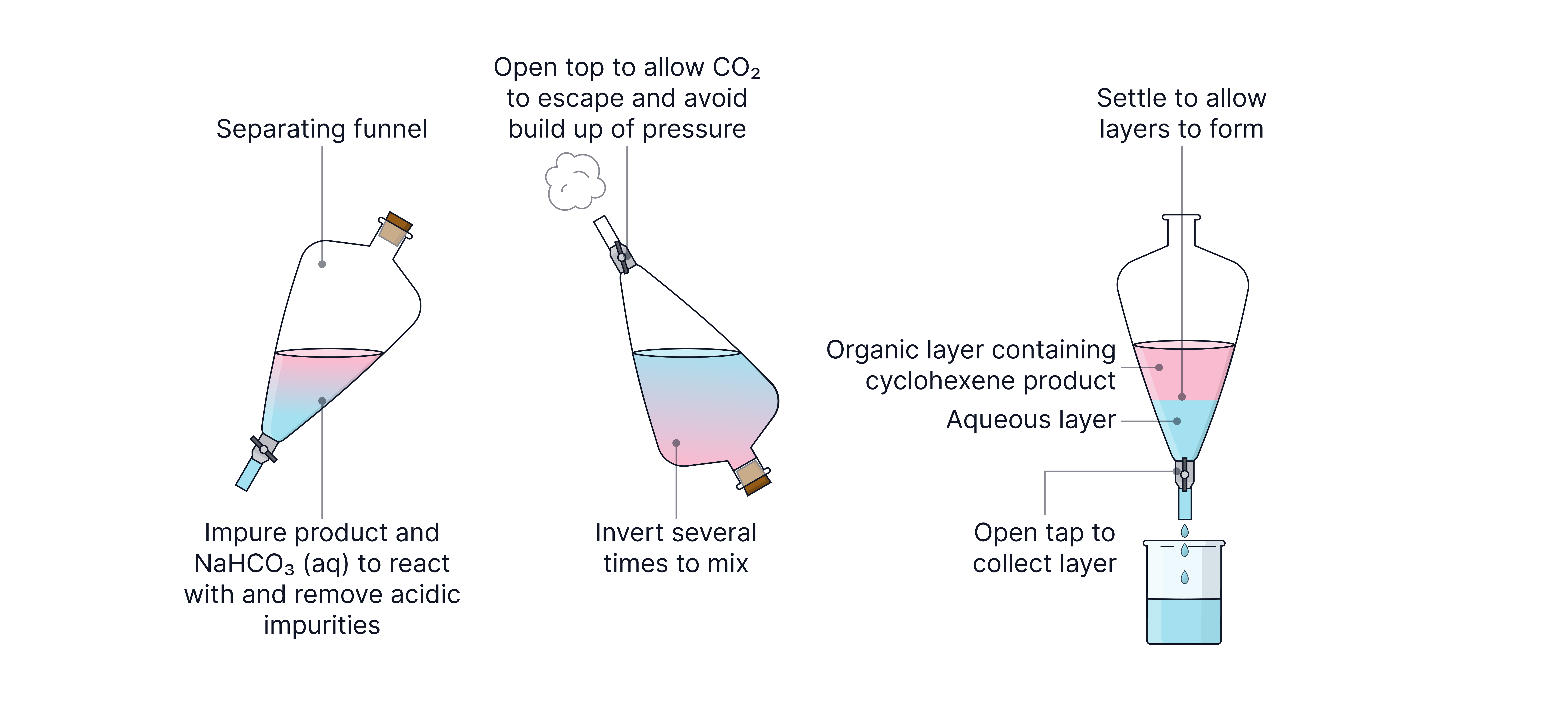
The acid is removed by washing the product mixture with aqueous sodium carbonate in a separating funnel.
This separates the mixture into two layers. The organic layer containing the cyclohexene product forms at the top due to lower density than water (the aqueous layer).
Any remaining water can then be removed from the organic layer by adding solid anhydrous calcium chloride which is a drying agent.
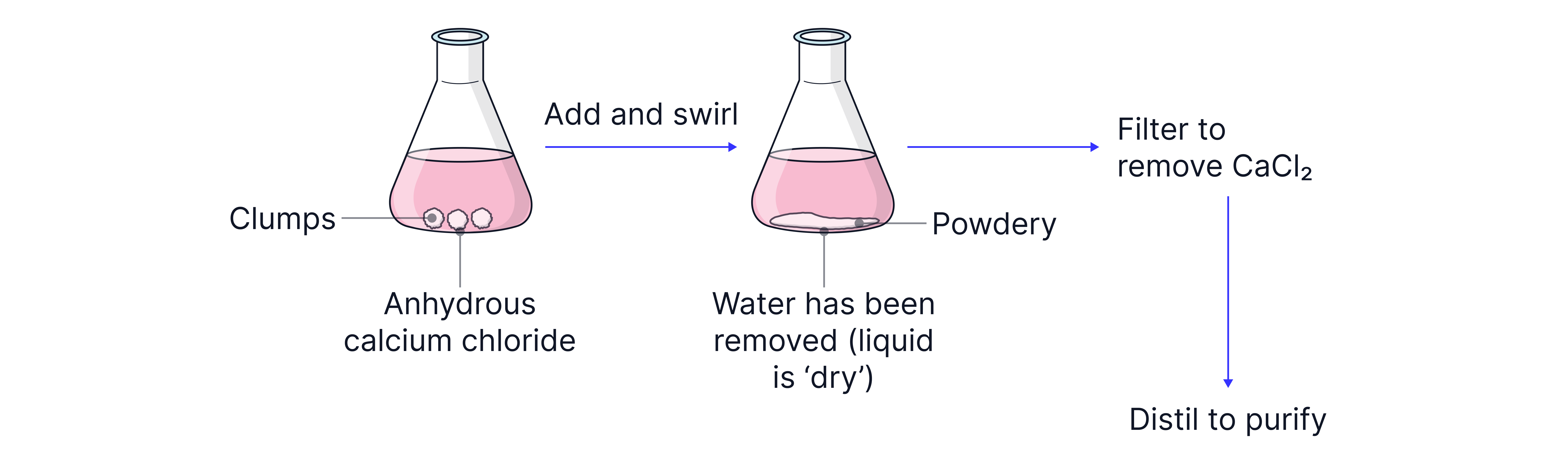
Practice Question
Question 1
Why is ethanol distilled immediately after adding acidified dichromate?
Answer:
To prevent further oxidation of aldehyde to carboxylic acid.
Question 2
Name the mechanism for the dehydration of cyclohexanol.
Answer:
Elimination
Key Tips & Reminders
For Ethanal:
Use limited oxidant + gentle heating.
Distillation, not reflux.
Add anti-bumping granules.
Cr₂O₇²⁻ turns orange → green.
For Cyclohexene:
Conc. acid catalyst + distillation.
Collect between 80–85°C.
Wash with NaHCO₃ and dry with CaCl₂.
Final product decolourises bromine water.