RP4 - Identifying Anions & Cations
Lajoy Tucker
Teacher

Contents
Introduction
Inorganic ions: Charged species formed by elements or compounds in aqueous solution (e.g. , , , ).
Analytical tests allow identification of specific ions via characteristic reactions and their accompanying observations (precipitation, gas evolution, etc.).
In solution, soluble ionic compounds dissociate into positive ions (cations) and negative ions (anions). One or both of these can be identified through a series of tests.
Test for Halide Ions (Cl⁻, Br⁻, I⁻)
Step 1: Add dilute nitric acid ()
Step 2: Add silver nitrate solution ()
A silver halide () precipitate forms, and its colour will allow identification of the halide ion that was present in solution.
Ionic Equation:
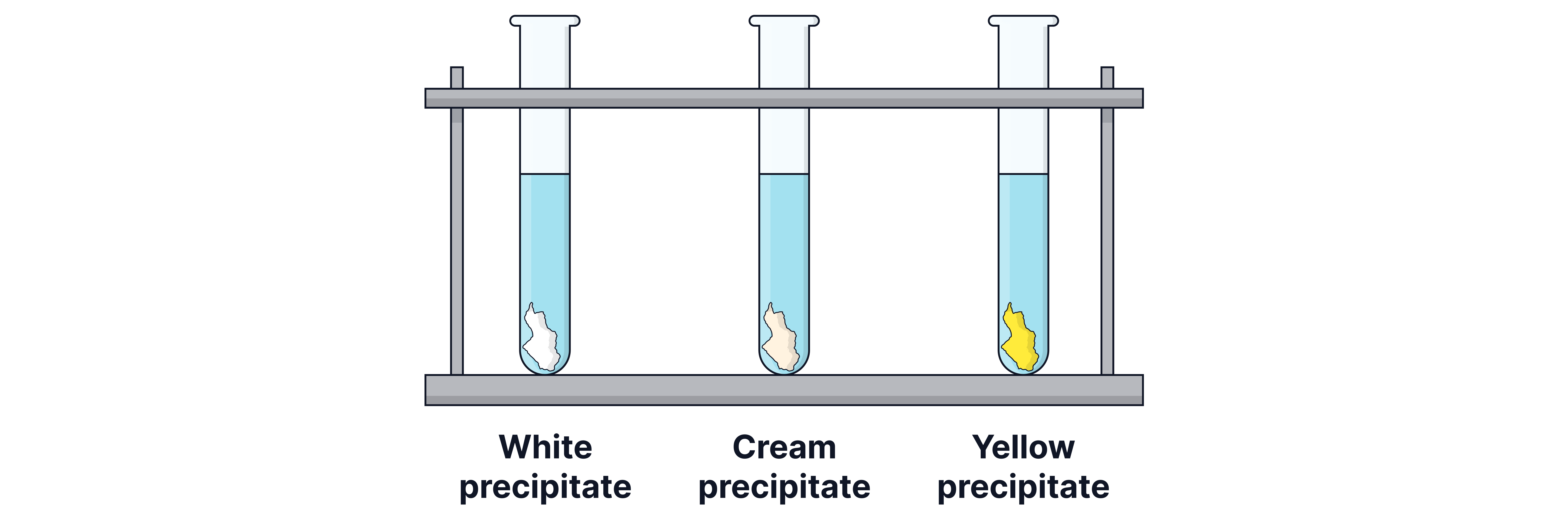
These precipitates can look very similar, so ammonia solution is added to distinguish between them. The silver chloride precipitate () is soluble in dilute ammonia, and the silver bromide precipitate () is soluble in concentrated ammonia.
Step 3: Add dilute ammonia solution. If the precipitate dissolves, it was .
Step 4: Add concentrated ammonia solution. If the precipitate dissolves, it was . If the precipitate persists it was
Ionic Equation:
Reagent | Cl⁻ | Br⁻ | I⁻ |
|---|---|---|---|
+ AgNO₃ | White precipitate (AgCl) | Cream precipitate (AgBr) | Yellow precipitate (AgI) |
+ dilute NH₃ | Precipitate dissolves | No change | No change |
+ conc. NH₃ | Precipitate dissolves | Precipitate dissolves | No change |
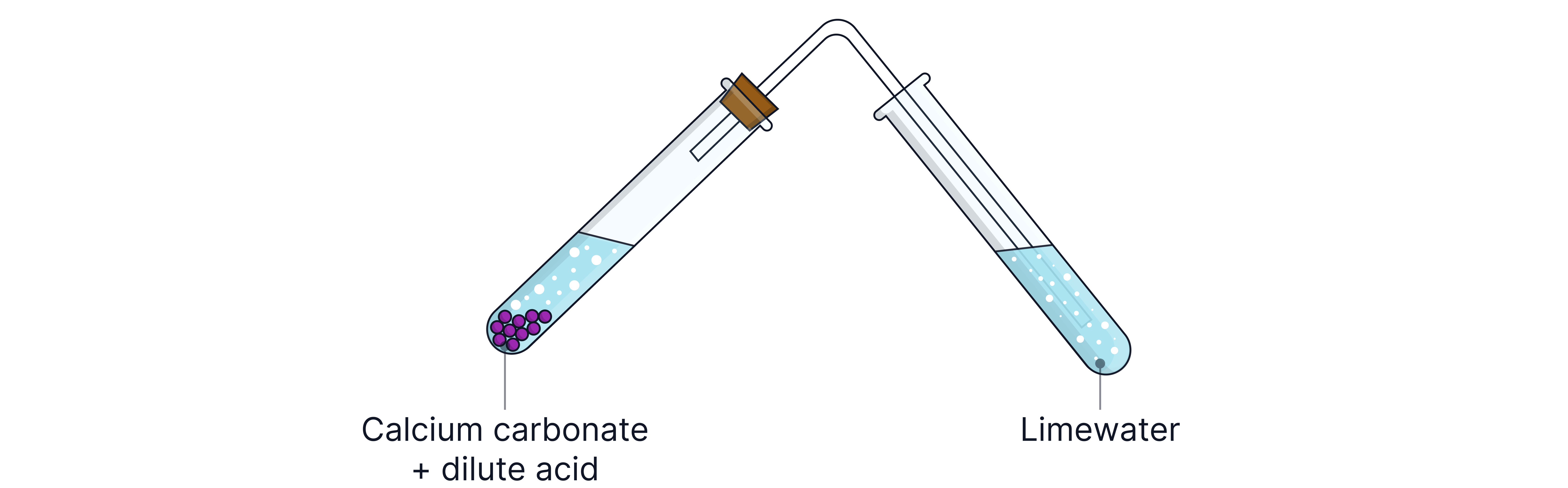
Test for Carbonate Ions ()
Reagents: Add dilute or dilute to the solution sample
Observation: Effervescence (bubbling) due to gas.
Ionic equation:
Confirmatory Test: Bubble gas through limewater turns milky (white precipitate of )
Equation:
Test for Sulfate Ions ()
Step 1: Add dilute to the solution sample
Step 2: Add barium chloride solution ()
Observation: White precipitate of barium sulfate () forms
Ionic equation:
Use of Acid
The tests for halide and sulfate ions require the addition of a dilute acid before the test reagent was added.
The acid removes any carbonate or hydroxide ions which may give a false positive with the test reagent.
, , and are all white solids. These would form as white precipitates in solution if the interfering and/or ions were not removed first by adding acid
The acid used should have the same anion as the test reagent so as not to add interfering ions e.g. silver nitrate used for halide test, therefore nitric acid used to acidify the solution.
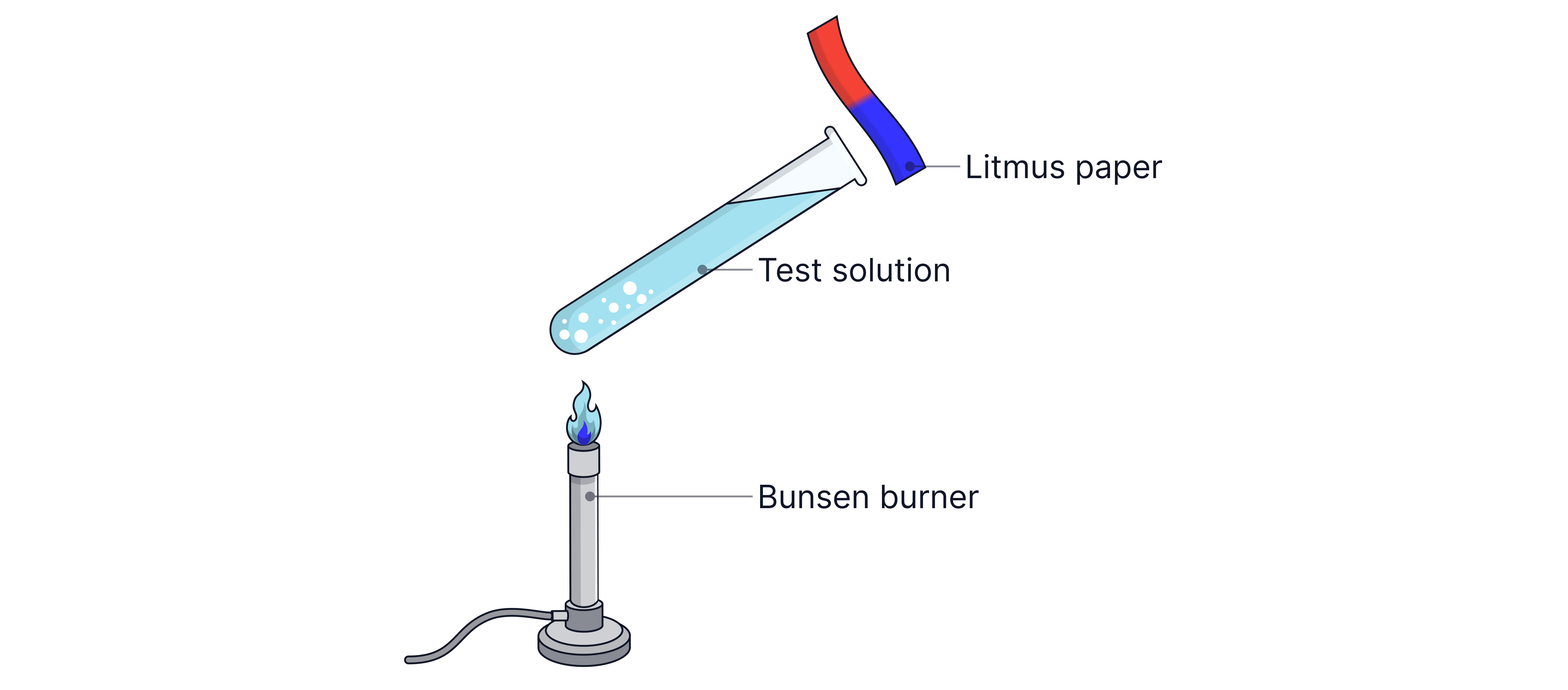
Test for Ammonium Ions (NH₄⁺)
Step 1: Add sodium hydroxide () and warm gently
Step 2: Hold damp red litmus paper over the mouth of the test-tube
Observation: Gas is produced that turns damp red litmus paper blue
Ionic equation:
Test for hydroxide ions ()
Hydroxide ions can be tested for using universal indicator paper or damp red litmus paper both of which would turn blue if present.
Group 2 ions ()
Group 2 ions can be identified through knowledge of the solubility of their sulfate and hydroxide compounds.

|
|
|
|
|
|
|
Excess NaOH (aq) added |
White precipitate |
White precipitate |
Slight white precipitate |
Colourless solution (no observable change) |
|
Excess |
Colourless solution (no observable change) |
Slight white precipitate |
White precipitate |
White precipitate |
Practice Questions
Question 1
A solution gives a cream precipitate with silver nitrate, which dissolves only in concentrated ammonia. Identify the ion.
Answer:
Cream ppt =
Soluble only in concentrated
Therefore, ion is
Question 2
Describe a test and observation to confirm the presence of ions in aqueous solution. (2 marks)
Answer:
Add followed by
White precipitate of forms
Key Tips
Always add acid (
or
) before precipitation tests to remove unwanted ions (e.g.
,
).
Include changes in colour AND state for observations
Include state symbols when writing equations for these tests to make observations clear.
If given a solid sample to test, be sure to dissolve it in water first to form a solution.