RP3 - Factors Affecting the Rate of a Reaction
Lajoy Tucker
Teacher

Contents
Key Terms:
Rate of Reaction: Change in concentration of a reactant or product per unit time.
Initial Rate: The rate at the very start of the reaction .
Activation Energy : Minimum energy required for particles to react.
Collision Theory: Reactions occur when particles collide with sufficient energy and correct orientation.
Basic Principles
As temperature increases, more particles have energy , so more frequent successful collisions occur.
A small temperature rise can cause a large increase in rate due to the exponential nature of the Maxwell–Boltzmann distribution.
Measured via initial rate using a visible change, e.g., precipitate formation or gas production.
Aim:
To determine how increasing temperature affects the rate of a reaction.
Example reaction:
Sodium thiosulfate + Hydrochloric acid
Solid sulfur forms a yellow precipitate – rate measured by time taken for cross to disappear.
Experimental details
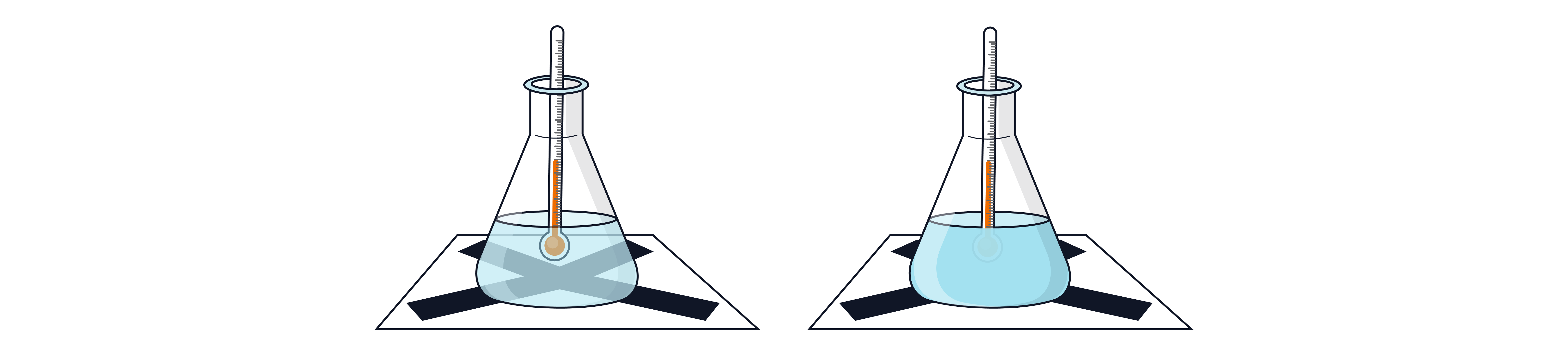
Known volumes and concentrations of sodium thiosulfate and hydrochloric acid are mixed together and placed on white paper with a black cross.
A timer is started as soon as the solutions are mixed.
The timer is stopped once the cross disappears.
This reaction is then repeated at a minimum of 5 temperatures using water baths to achieve each temperature.
The same concentrations and volumes of both solutions must be used for each test to ensure validity.
Note - The temperature of the solutions will not be the same in the water bath as when the reaction is actually taking place. Therefore, the temperature is taken before and after each reaction so a mean temperature can be calculated and used in analysis.
Processing data
It can be assumed that at each temperature, the cross disappears when the same amount of sulfur has formed.
Therefore, as rate is calculated as change in quantity of reactant or product/time
Where change in quantity = amount of sulfur produced = constant
Rate = constant/time
Rate
The initial rate of reaction is therefore approximated as
Example results table
Temperature | ||
|---|---|---|
Time taken for cross to disappear | ||
Rate |
A graph can then be plotted of rate against temperature
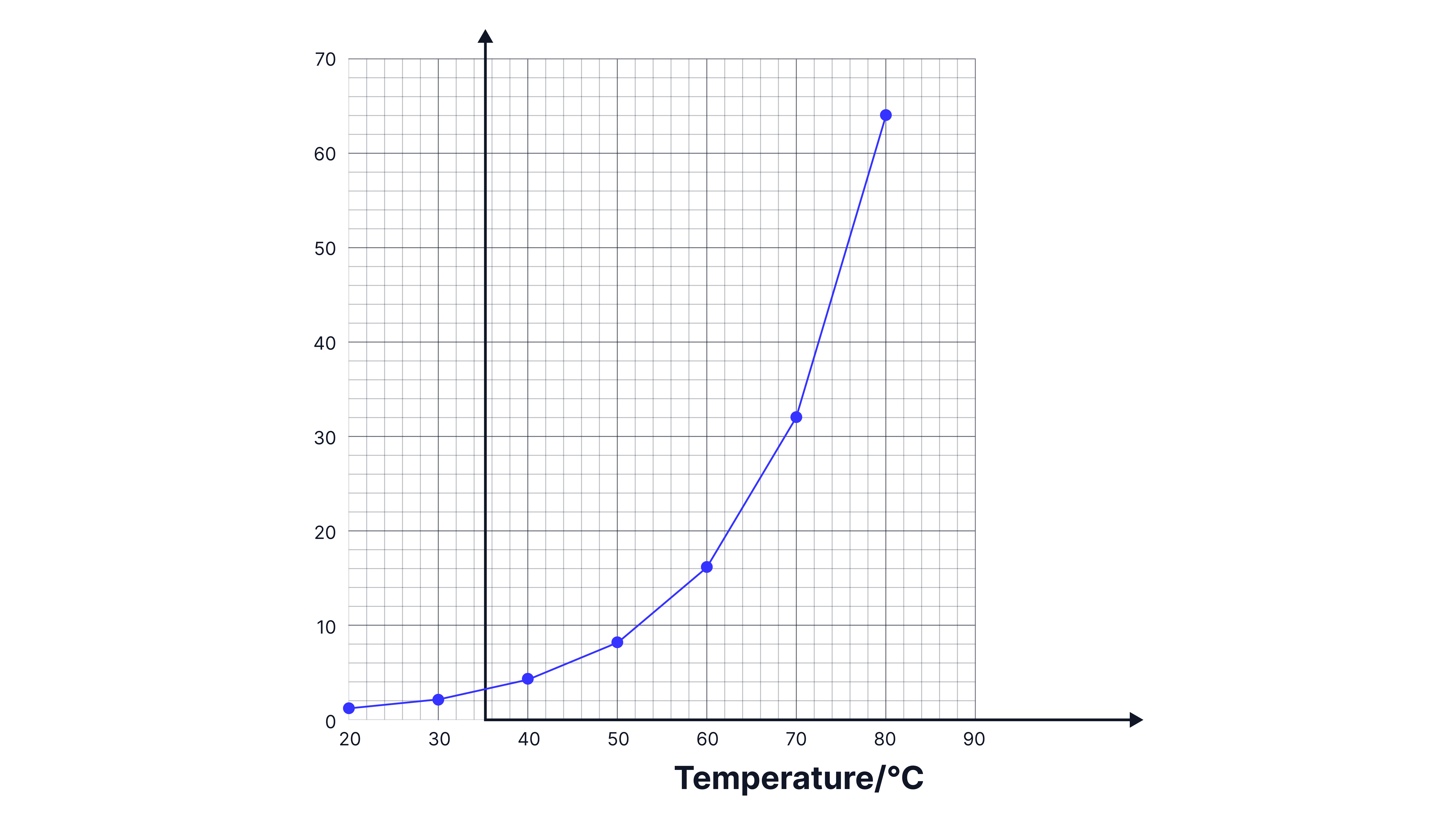
Source of error
The cross disappearing is subject to the observer’s judgement and their reaction time.
Accuracy could therefore be improved by using colorimetry or a light sensor along with a data-logger to determine when the reaction mixture turns opaque.
Practice Questions
Question 1
Explain why the rate of reaction increases with temperature (3 marks).
Answer
Particles have more kinetic energy
More particles have energy ≥ activation energy.
More frequent successful collisions.
Question 2
Suggest two ways to improve accuracy when using the disappearing cross method.
Answer
Use a light sensor and data logger instead of visual observation.
Ensure consistent viewing angle or use the same observer for all trials.