RP7 - Continuous Monitoring Method
Lajoy Tucker & Dr. Davinder Bhachu
Teachers


Contents
Measuring the Rate of Reaction
What do we mean by “rate of reaction”?
The rate of a chemical reaction tells us how fast reactants are used up or products are formed. Mathematically, it can be expressed as:
Rate = change in concentration ÷ time
But in practice, we can’t usually measure instantaneous changes at every point. Instead, we use methods that either:
1. Track concentration changes over time (continuous monitoring).
2. Measure the time taken to reach a fixed point (initial rate/clock reactions).
Preparing Organic Solid Explained
Method 1 – Continuous Monitoring
Key Idea
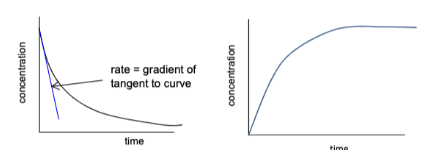
We follow one experiment over time, recording how the concentration of a reactant/product changes with respect to time. If we plot a graph of concentration vs time:
The gradient at any point = rate at that moment.
The reaction is fastest at the start (steepest gradient).
As reactants are used up, the gradient flattens.
Eventually, the line becomes horizontal – the reaction has stopped.
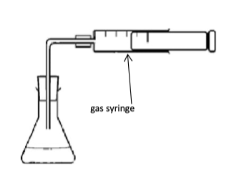
1. Measuring the Volume of Gas Produced
When it works:
Useful if there is a change in the number of moles of gas during the reaction.
Best method when a gaseous product is formed.
How it works:
Attach a gas syringe to the reaction vessel.
As the reaction proceeds, gas collects in the syringe.
The increase in gas volume is recorded at regular time intervals.
Example reactions:
2. Measuring Change in Mass
When it works:
If a gas is produced and allowed to escape, the mass of the reaction mixture decreases.
Works best with dense gases such as CO₂ (loss of mass is easier to measure accurately).
How it works:
The reaction is carried out in an open flask placed on a balance.
Mass is recorded at timed intervals.
Example reaction:

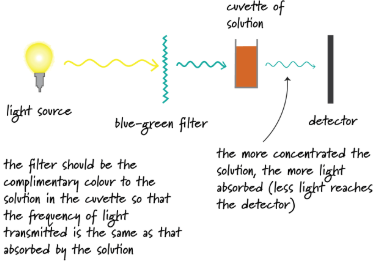
3. Colorimetry
When it works:
If one of the reactants or products is coloured.
A colorimeter can track the colour intensity, which is proportional to concentration.
Example reaction:
Iodine produced gives a brown colour.
Diagram Placeholder:
4. Measuring Change in Electrical Conductivity
When it works:
Useful if the number of ions in solution changes during the reaction.
The more ions present, the higher the conductivity.
Example reaction:
5. Measurement of Optical Activity
When it works:
Some reactions involve chiral molecules.
If optical activity changes (rotation of plane-polarised light), a polarimeter can be used to track it.
Example reaction:
Continuous Monitoring Explained
Non-continuous methods
Using Titration to Monitor Rates
Titration can be used to track reaction progress, but it is not a continuous method. Instead, it is a discontinuous sampling method – you only get concentration values at separate time points, not a real-time trace.
How it works
1. Remove a small sample of the reaction mixture at set time intervals.
2. Quench the sample (stop the reaction immediately) so no further reaction occurs before analysis. Quenching can be done by:
Diluting with cold water,
Cooling rapidly, or
Adding a reagent that reacts instantly with one reactant.
3. Titrate the quenched sample with a suitable standard solution to measure the concentration of a chosen reactant or product.
4. Repeat for several time points to build a concentration–time graph.
Example
At intervals, samples are withdrawn.
Each sample is quenched and titrated with sodium thiosulfate to find how much iodine is left.
Plot [I₂] against time.
Why it’s useful
Accurate for reactions involving acids, bases, or iodine.
Allows rate data to be collected even when no gas or obvious colour change is produced.
Limitations
Not continuous – relies on manual sampling and gives only “snapshots”.
Results depend on quenching being fast and effective.
Slower and more labour-intensive than gas syringes or colorimetry.