RP7 - Iodine Clock Reaction
Lajoy Tucker
Teacher

Contents
Introduction to Iodine Clock Reaction
What is an initial rates method?
The rate of reaction changes over time. Reactions are fastest at the start, then slow down as reactant concentration decreases over time.
The initial rate is the rate right at the beginning (t = 0).
Measuring the initial rate is useful because:
All concentrations are known exactly at the start.
It avoids complications from changing concentrations or side reactions later.
The rate is highest, so differences are easier to detect.
How can we measure an initial rate?
There are two main approaches:
1. Continuous monitoring and tangents
Follow the reaction over time (e.g. gas syringe, colorimeter).
Plot a concentration–time graph.
Draw a tangent at t = 0 to find the initial gradient.
2. Clock reactions
Instead of tracking the whole reaction, measure the time taken for one clear, visible event (e.g. a sudden colour change).
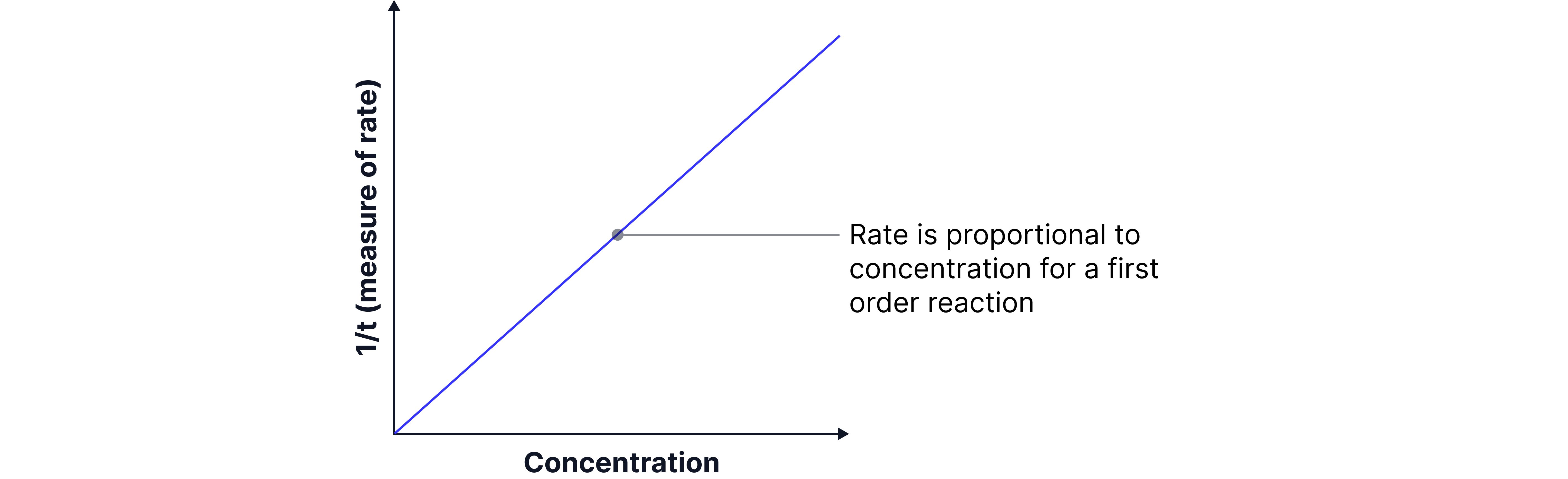
If the event always happens after the same small, fixed amount of product has formed, then we can say that: Initial rate ∝ 1/time .
The iodine clock reaction – an example of a clock method
The reactions
1. Slow step (rate-determining):
2. Fast step (mopping up iodine):
Sodium thiosulfate is the clock reagent.
It removes iodine as soon as it forms, so the solution stays colourless at first.
Once the fixed amount of thiosulfate is used up, iodine builds up and reacts with starch → sudden blue–black colour.
Step by Step Practical Method
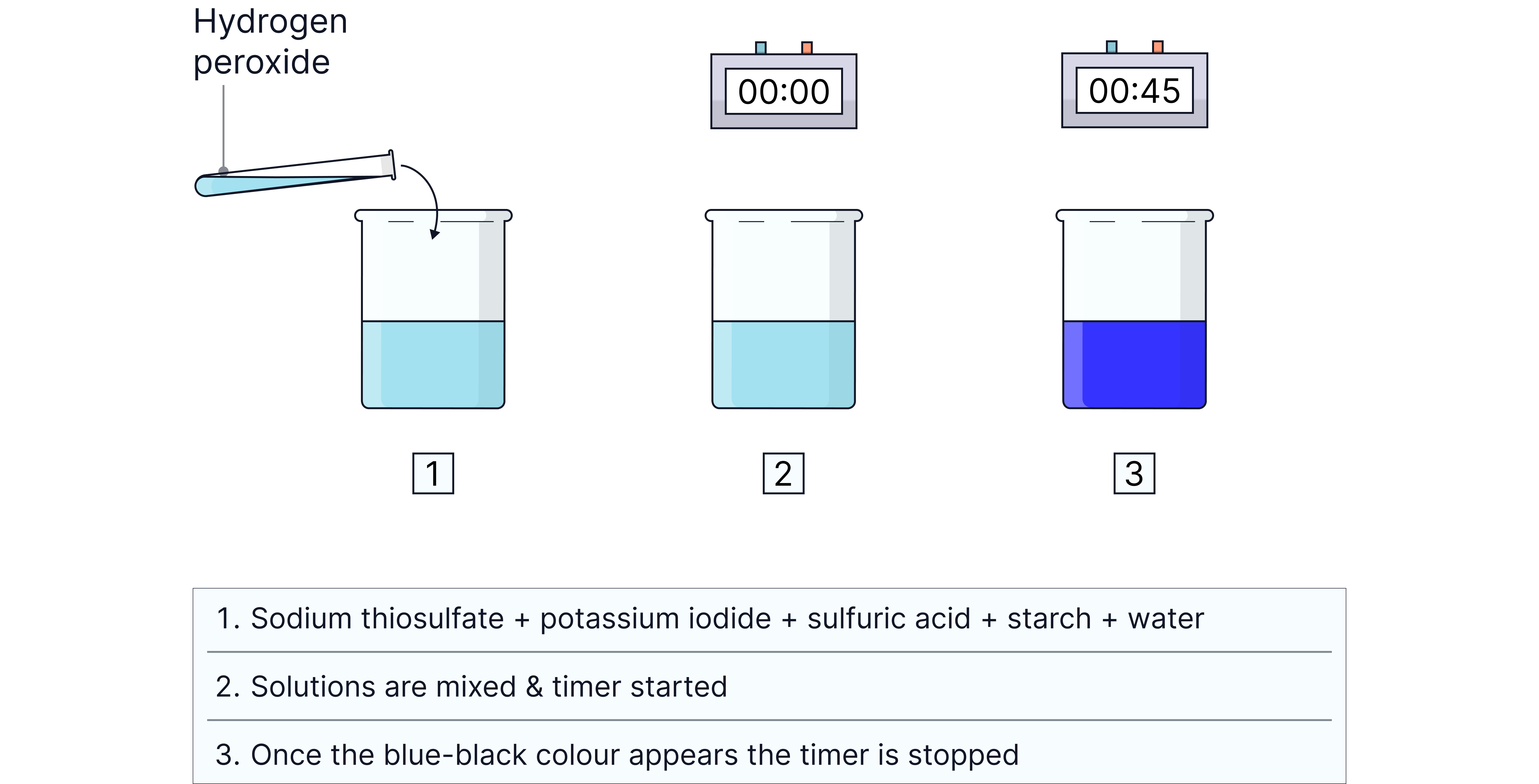
1. Set up the reaction mixture
Use burettes to measure out solutions of sulfuric acid, sodium thiosulfate, starch, and water into a conical flask.
Add a known volume of potassium iodide solution .
2. Start the reaction
Measure a fixed volume of hydrogen peroxide into a test tube.
Quickly pour it into the flask, swirl, and start the stopwatch.
3. Record the time
Stop the timer when the mixture first turns blue–black.
4. Repeat
Keep all other volumes constant, but change the volume of KI (and adjust the water so the total volume stays the same).
This means only changes – all other concentrations remain constant.
Why keep the total volume constant?
The concentration of each reagent in the conical flask depends on:
the volume of solution added, and
the final total volume in the flask.
If the total volume is always the same (e.g. 50 cm³), then the concentration of a reactant is directly proportional to the volume of that solution added.
Example: doubling the volume of KI solution added doubles the in the flask.
To achieve this, whenever we increase the volume of KI, we reduce the volume of water so the total volume is unchanged.
Why this matters:
If we didn’t control total volume, then every reactant concentration would change at once.
By keeping the volume constant, we can be confident that any change in rate is due only to the reactant we are investigating.
Processing results
Run |
|
| |
1 | 0.020 | 36.0 | 0.0278 |
2 | 0.040 | 18.0 | 0.0556 |
3 | 0.060 | 12.0 | 0.0833 |
Comparing runs: doubling halves the time, which doubles first order in .
Why this is an initial rates method
When the solutions are mixed, iodine is produced slowly by the reaction between hydrogen peroxide and iodide ions:
At the same time, any iodine formed is immediately removed by a fast reaction with thiosulfate ions:
As long as thiosulfate ions are present, iodine does not accumulate and no colour is seen. The colour change only occurs when all the thiosulfate has been used up, which means that a fixed amount of iodine must have been formed by the slow reaction. Because only a small amount of iodine is needed to use up the thiosulfate, only small amounts of hydrogen peroxide and iodide are consumed, so their concentrations remain close to their initial values and the rate has not slowed significantly. The measured rate is therefore approximately the initial rate.
The rate of reaction is defined as the amount of iodine formed divided by the time taken. In this experiment, the amount of iodine formed at the colour change is always the same; we can call this fixed amount . Therefore, rate , which can also be written as rate . Since C is a constant, this shows that the rate is .
By repeating the experiment while changing the concentration of just one reactant, such as iodide ions or hydrogen peroxide, and keeping all other conditions constant, any change in must be due to the change in that concentration. This allows the order of reaction with respect to that reactant to be determined.
Repeating for other reactants
The same process can be repeated for or .
Each time, vary just that one concentration while keeping all others constant (adjusting water to keep total volume fixed).
By analysing how changes, you can find the order with respect to each reactant.
Final outcome
Once the order with respect to each reactant is known, you can write the overall
rate equation:
You can then use data from one experiment to calculate the rate constant,