RP8 - Making Simple Cells
Dr. Davinder Bhachu
Teacher

Measuring the EMF of an Electrochemical Cell
What is EMF?
Electromotive force (Ecell) is the maximum potential difference between two half-cells when no current flows.
It measures how readily electrons flow from one electrode to the other.
Standard conditions:
solutions,
,
pressure (for gases).
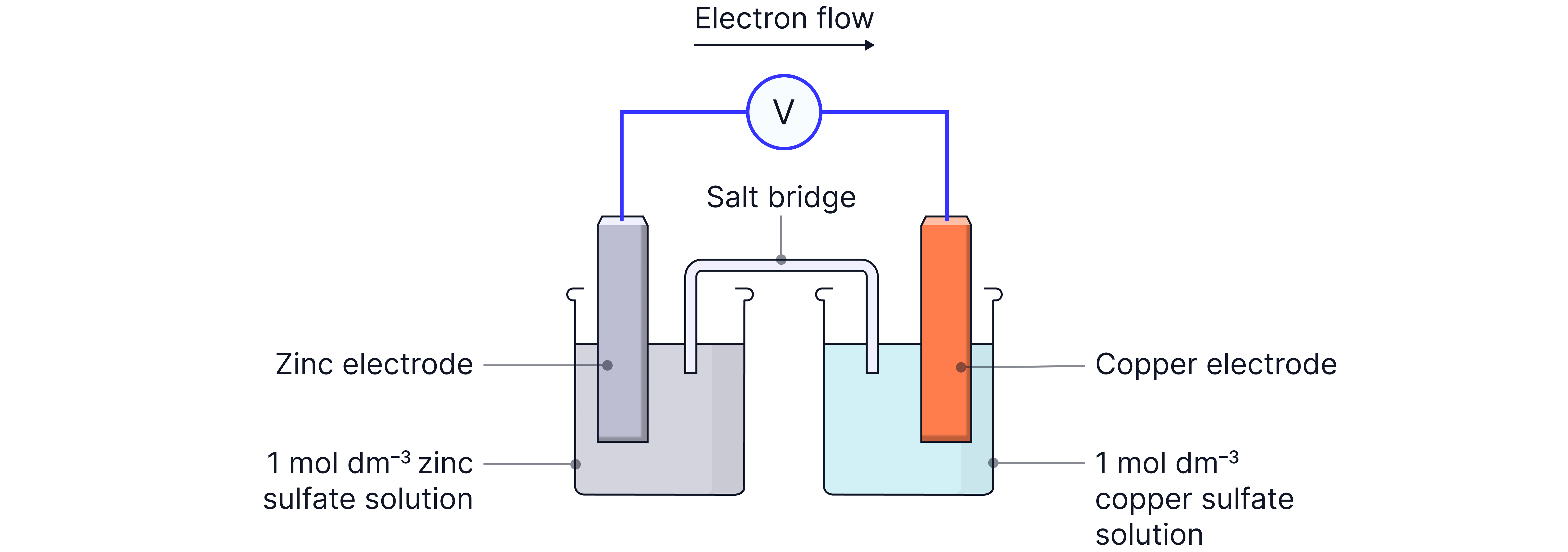
Method (metal/metal ion half-cells) – e.g. Zn/Cu cell
1. Prepare the electrodes
Clean zinc and copper strips with emery paper.
Degrease with propanone and cotton wool.
This ensures good electrical contact.
2. Set up the half-cells
Place zinc into of solution.
Place copper into of solution.
3. Connect with a salt bridge
Soak filter paper in saturated potassium nitrate solution (KNO₃).
Place strip across the two beakers.
The salt bridge provides mobile ions to complete the circuit without interfering chemically.
4. Connect electrodes to voltmeter
Use crocodile clips and wires.
Record the EMF.
Role of the salt bridge
Contains free ions which allow charge balance between the half-cells.
Must be inert (e.g. ) so it does not react with electrode ions.
Potassium chloride is unsuitable with copper – can form complexes with .
A wire is not used – a metal wire would introduce its own electrode system.
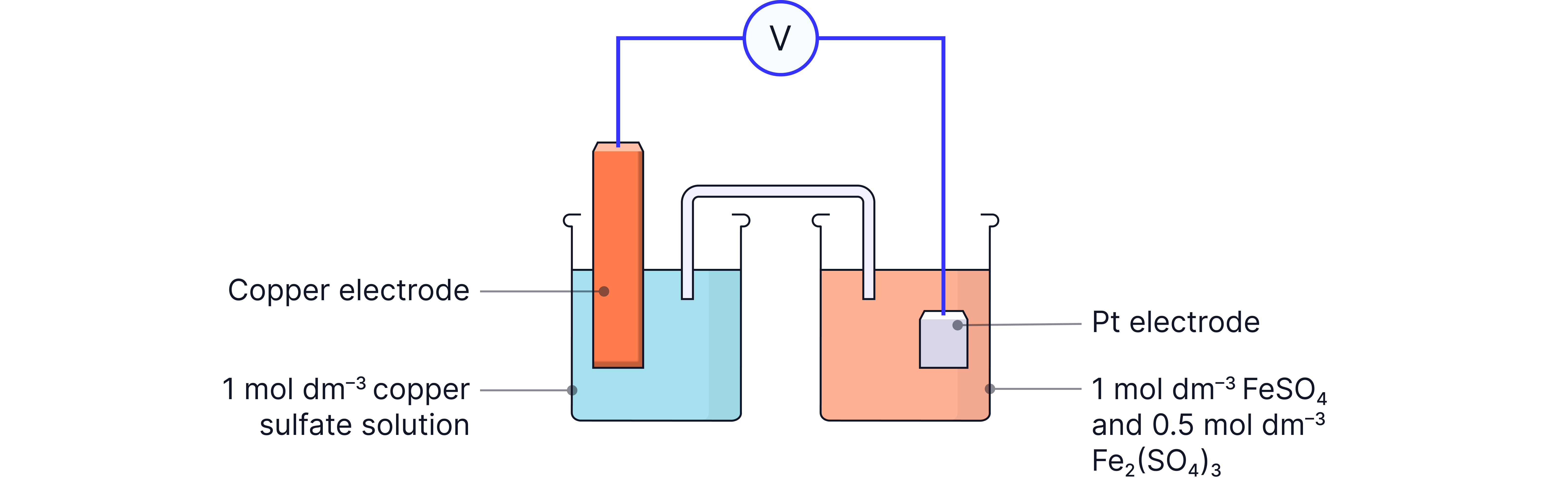
Method (inert electrode half-cell) – e.g. Fe²⁺/Fe³⁺ with Cu²⁺/Cu
1. Prepare the iron half-cell
Mix equal volumes of and , acidified with .
This gives overall.
2. Electrode
Use a platinum electrode electrode
Platinum is inert (won’t react) but conducts electricity.
3. Complete the cell
Combine with a half-cell.
Use a fresh salt bridge.
Connect via voltmeter and record EMF.
Why platinum?
Unreactive and provides a conducting surface for electron transfer.
Essential when no solid conducting electrode is available (e.g. ).
Making Cells Explainer Video
Key Exam Points
Direction of electron flow: electrons always flow from the more negative electrode (greater tendency to oxidise) to the more positive.
Salt bridge: prevents solutions mixing directly (which would set up side reactions), but allows ionic conduction.
Measuring EMF: use a high-resistance voltmeter so no current flows – ensures the measured value is the maximum potential difference.
Standard electrode potential (): compare each half-cell against the standard hydrogen electrode (SHE).
Common Pitfalls and Errors
Not cleaning electrodes properly → poor contact → inaccurate EMF.
Salt bridge drying out or not soaked enough → circuit incomplete.
Using incorrect salt bridge (e.g. with ) → side reactions.
Solutions not equimolar → not standard conditions.
Predicting feasibility: combine half-cell values to calculate overall . Positive suggests reaction is feasible.
Limitations: even if is positive, kinetics may be too slow (high activation energy) or non-standard conditions may alter outcomes.