RP9 - Titration Curves
Lajoy Tucker & Dr. Davinder Bhachu
Teachers


Contents
Introduction to Titration Curves
What is a titration curve?
A graph of pH against volume of base (or acid) added.
Shows how pH changes during neutralisation.
Helps us identify the equivalence point and select a suitable indicator.
Indicator | Colour at Low pH | pH Range of Colour Change | Colour at High pH |
|---|---|---|---|
Methyl orange | Red | 3.2 – 4.4 | Orange |
Phenolphthalein | Colourless | 8.2 – 10.0 | Purple |
Titration Curves Practice Question Explainer Video
Constructing a Titration Curve (Method)
1. Use a volumetric pipette to transfer of acid into a beaker.
2. Insert a calibrated pH probe and measure the initial pH.
3. Add base from a burette in portions, recording the total volume added each time.
4. Stir after each addition and record pH to 1 d.p.
5. Near the endpoint, switch to (or smaller) additions for accuracy.
6. Continue until base is in excess.
7. Plot pH vs volume of base added.
Accuracy Tips:
Calibrate pH probe using buffer solutions (pH 4, 7, and 9).
Maintain constant temperature (pH is temperature-dependent).
Using a Calibration Curve
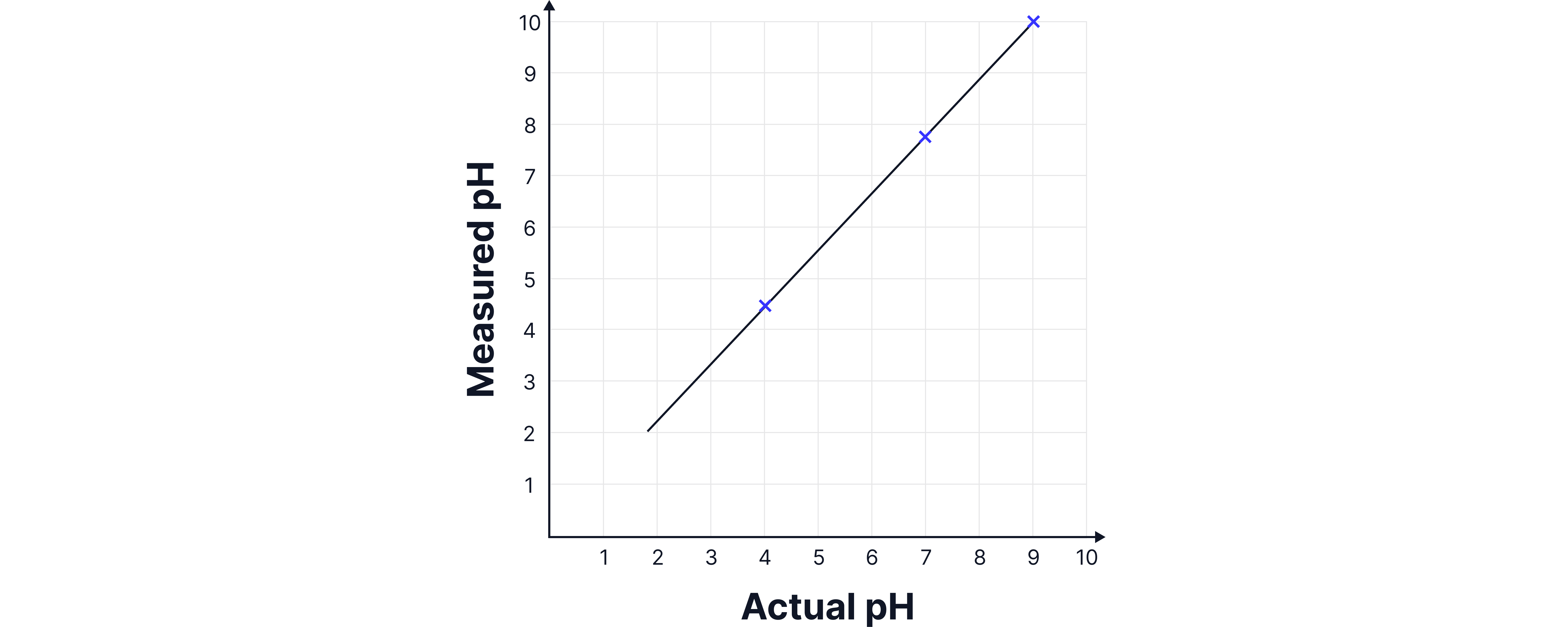
1. Constructing the calibration Curve
The probe is placed in buffer solutions of known pH (e.g. pH 4, 7, 10).
A graph of measured pH against known pH is plotted.
This calibration curve allows conversion of raw meter readings into accurate pH values.
Applying Calibration to a Titration
During the titration, the probe is placed in the solution and readings are taken as titrant is added.
These raw readings are converted into pH values using the calibration curve.
Plotting pH (y-axis) against volume of titrant added (x-axis) gives the titration curve.
Why this matters
Without calibration, the pH probe may give inaccurate readings (electrode drift, impurities, temperature effects).
Calibration ensures the shape of the titration curve is reliable – especially the steep change near equivalence.
How to read a titration curve – the four stages
Stage 1 – Before any base is added
Flask contains acid only.
Strong acid → very low pH (~1).
Weak acid → higher pH (~3–4).
Stage 2 – Adding base, before equivalence
Base reacts with acid.
Weak acid + strong base: mixture contains acid + salt → buffer region with gentle slope.
Stage 3 – Equivalence point
Acid has been completely neutralised by base.
Flask contains only salt (and water).
pH depends on type of salt:
Strong acid + strong base → pH ≈ 7.
Weak acid + strong base → pH > 7.
Strong acid + weak base → pH < 7.
Curve shows a steep vertical rise.
Stage 4 – After equivalence
Excess base is present.
pH levels off at ~13–14.
Why does the titration curve have a sharp vertical rise near the equivalence point?
1. Most of the acid has been neutralised
At the equivalence point, there is almost no free acid or base left in the flask – just salt and water.
The solution can’t “resist” changes in pH any more.
2. Small additions of base cause large pH changes
Before equivalence: adding a bit of base removes some acid, but there’s still lots of acid left → pH only rises a little.
At equivalence: only a tiny amount of acid is left. Adding the same volume of base now causes a much bigger swing in [H⁺], so pH shoots up.
3. It’s not gradual – it’s sudden
In just a drop or two of titrant, the solution goes from acidic to neutral to alkaline.
This sudden change creates the almost vertical part of the graph.
Key Exam Links
Indicators: must change colour within the steep vertical region.
Ka from titration curve: for weak acids, pH at half-neutralisation = pKa.
Choice of method: pH meter is essential if there’s no steep equivalence region (weak acid + weak base).