RP10 - Preparing an Organic Solid
Dr. Davinder Bhachu
Teacher

Contents
Learning Goals
Make an organic solid (e.g. aspirin).
Purify it by recrystallisation.
Isolate it by vacuum filtration.
Dry the product properly.
Test purity by melting point (and, optionally, TLC).
Calculate percentage yield and comment on purity.
Choosing a Recrystallisation Solvent
Pick a single solvent or solvent pair that:
Dissolves the product when hot but not when cold.
Leaves impurities either always soluble (stay in filtrate) or always insoluble (removed by hot filtration).
Is volatile enough to dry off easily and is safe to heat.
Step | What to Do | Why It’s Done |
|---|---|---|
1 | Dissolve the impure solid in a minimum volume of hot solvent (near boiling). | A hot, saturated solution maximises recovery on cooling; “minimum volume” reduces losses to solubility. |
2 | Hot gravity filter through fluted paper and a pre-warmed funnel. Optionally treat with a pinch of activated charcoal, then refilter hot. | Removes insoluble particles; keeping solution hot prevents premature crystallisation; charcoal adsorbs dyes. |
3 | Cool slowly to room temperature undisturbed, then place in an ice bath to complete crystallisation. | Slow cooling gives fewer, larger crystals with fewer trapped impurities; the ice bath increases yield. |
4 | Vacuum filtration (Büchner funnel + side-arm flask + water pump). | Rapid filtration and partial drying of crystals. |
5 | Wash crystals with a small amount of cold solvent. | Removes mother liquor and soluble impurities without redissolving product. |
6 | Dry crystals: suction for a few minutes, then on absorbent paper, in a desiccator, or a low-temperature drying oven. | Prevents false high mass and poor melting point; ensures “dry to constant mass.” |
Actual Buchner flask
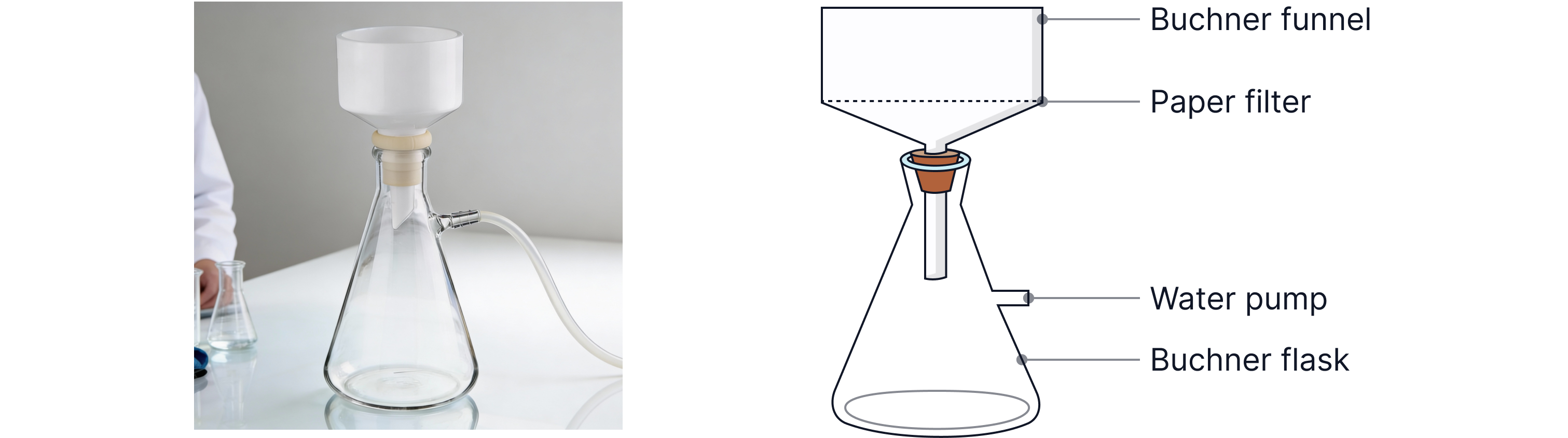
Common losses / how to minimise
Product stays dissolved in hot filtrate → use minimum hot solvent; cool fully.
Crystals lost during transfers/filtration → rinse glassware with tiny portions of cold solvent.
Oiling out (product oils instead of crystallising) → use different solvent or seed the solution.
Wet crystals → longer suction; spread thinly to dry.
Making aspirin – typical synthesis and work-up (example)
Reaction:
2-hydroxybenzoic acid (salicylic acid) + ethanoic anhydride → aspirin (acetylsalicylic acid) + ethanoic acid.
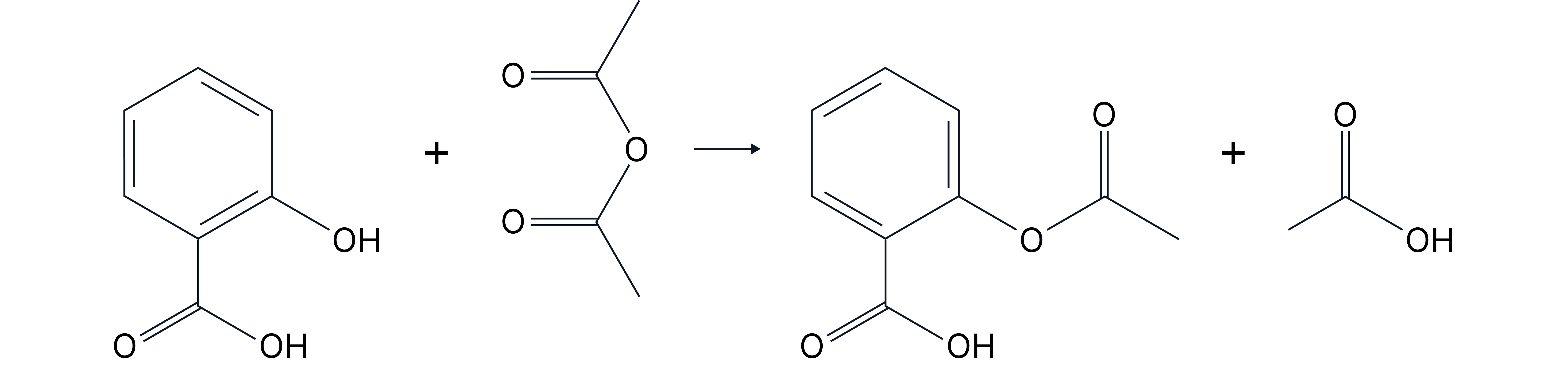
Use ethanoic anhydride (cheaper, safer, less moisture-sensitive than ethanoyl chloride).
Acid catalyst (few drops of or ).
Warm under reflux ~5–10 min.
Work-up
1. Pour hot mixture into cold water → aspirin precipitates; swirl to help nucleation.
2. Vacuum filter and wash with water to remove acids.
3. Recrystallise from ethanol–water (as above).
4. Dry to constant mass.
Why these choices?
Quench into cold water drives product out of solution and hydrolyses excess anhydride to harmless ethanoic acid.
Recrystallisation removes residual acids and by-products.
Avoid naked flames – ethanol is flammable.
Preparing an Organic Solid Explained
Testing purity – melting point (MP)
How
Pack a dry capillary tube with a tiny amount of dry sample.
Measure MP using a digital melting point apparatus or a simple oil-bath + thermometer.
Heat slowly near the expected MP (≈ 1–2 °C min⁻¹); record start (first signs of melt) and end (fully liquid).
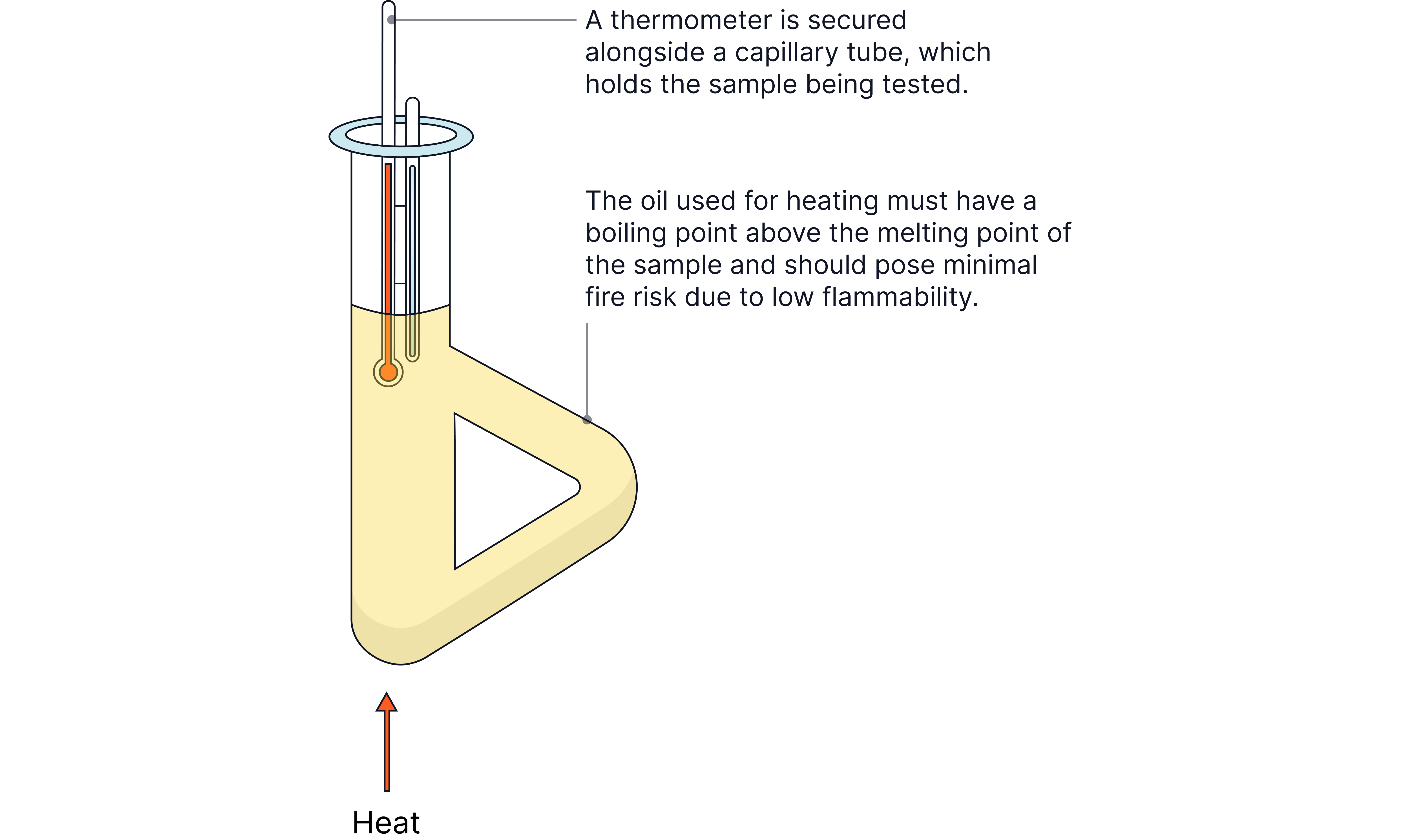
Interpreting
Pure solid → sharp MP, close to literature value (often within 1–2 °C).
Impure → MP lowered and broadened (range widened).
Calculations
Worked example – % purity of aspirin
salicylic acid (). The ethanoic anhydride is in excess.
n(salicylic acid) = .
Stoichiometry 1:1 → theoretical n(aspirin) = .
If you isolated 2.10 g: % yield = .
Practice Questions
Why “minimum hot solvent”?
Answer
To create a saturated hot solution – maximises crystallisation on cooling and minimises loss in filtrate.
Why cool slowly, then ice?
Answer
Slow cooling → larger, purer crystals; ice bath increases yield once crystals have formed.
Why hot gravity filtration before cooling?
Answer
Removes insoluble grit and charcoal while keeping product dissolved – avoids crystallising in the funnel.
Why wash with a little cold solvent?
Answer
Removes soluble impurities without dissolving the product.
Why vacuum (Büchner) rather than gravity filtration at the end?
Answer
Faster, and gives drier crystals ready for drying/MP.
Why might yield be < 100%?
Answer
Product remains in mother-liquor, losses on transfer/filtering, side reactions, incomplete drying.
> 100%? Wet crystals or trapped solvent
Quick checklist (for students)
Minimum hot solvent used – yes/no
Hot filtered through fluted paper – yes/no
Cooled slowly then iced – yes/no
Vacuum filtered and washed cold – yes/no
Dried to constant mass – yes/no
MP measured slowly – yes/no
Yield calculated and comments on purity made – yes/no