The Arrhenius Equation
Lajoy Tucker
Teacher

Contents
The Arrhenius Equation Explained
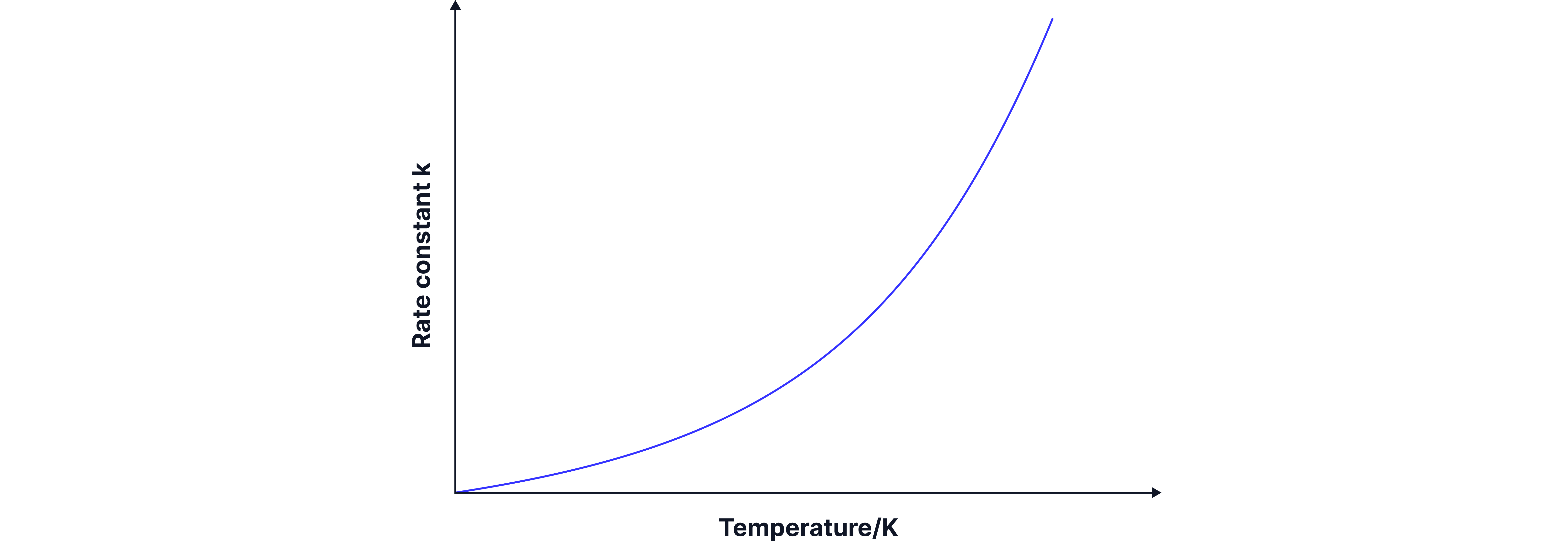
The Arrhenius equation describes how reaction rate is dependent on temperature. An increase in temperature leads to an increase in the rate constant , meaning that the rate of reaction increases. Roughly each rise in T doubles the rate constant Rate = .
The is given as:
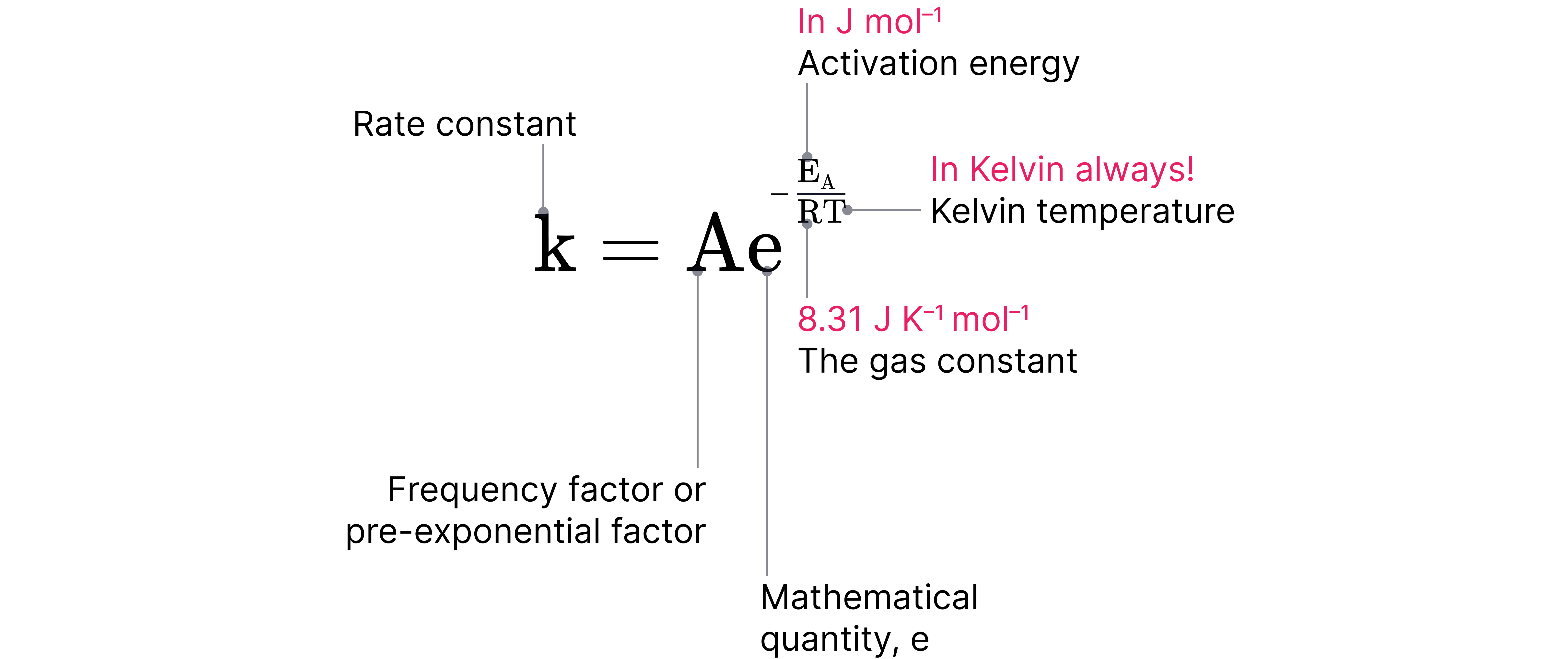
The Arrhenius constant accounts for the frequency of collisions and the orientation of molecules during a reaction and has the same units as the rate constant .
Temperature has an exponential influence on reaction rate because it appears in the denominator of the exponent in the equation.
Arrhenius Equation Explainer Video
Worked example:
Find the value of .
Given:
Answer
Logarithmic form of the equation
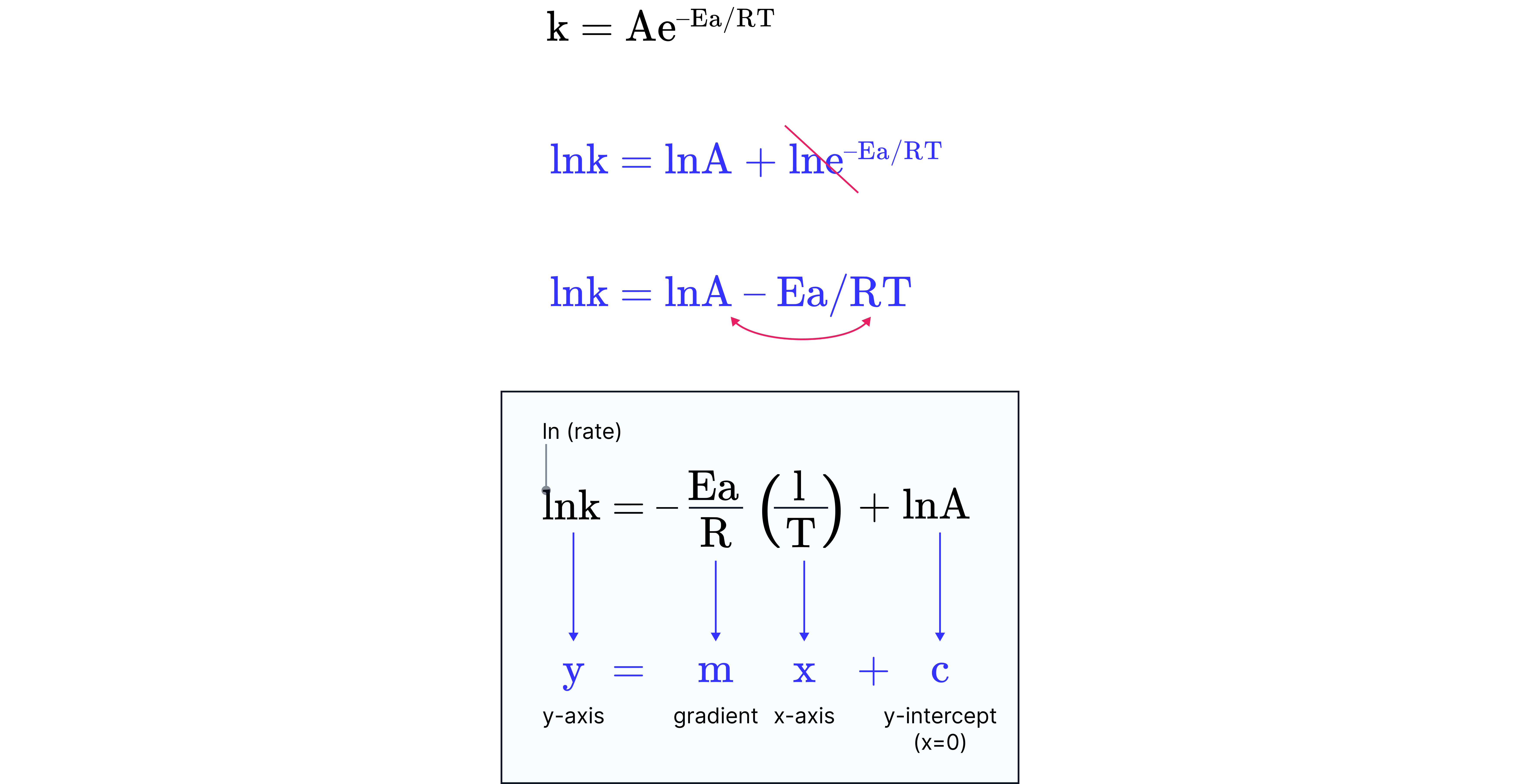
By taking the natural logarithm of both sides of the equation, the equation becomes:
This equation takes the form of a straight line, .
where:
is plotted against
Gradient is
The y-intercept is .
The activation energy, , in and A can be calculated by plotting a graph of vs .
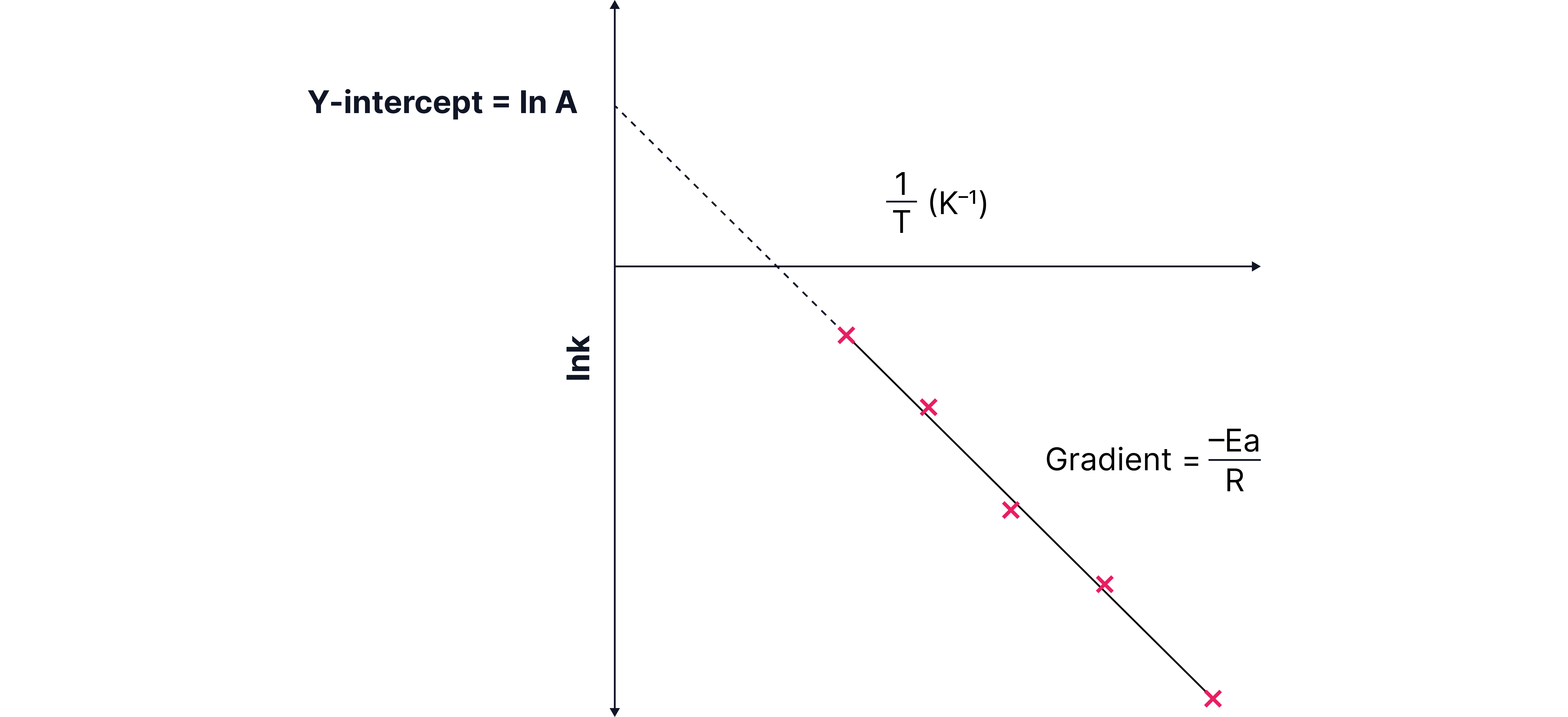
So,
Key tips:
Activation energies are positive therefore the gradient must be negative
Values on the axes do not have to start at 0. Choose an appropriate scale based on the values given.
As rate is proportional to the rate constant k, sometimes ln(rate) is plotted on the y axis. All following calculation steps are the same.
Worked Example
Given below is initial rate data collected at five different temperatures.
Complete the table calculating and ln(rate).
Plot a graph a use it to calculate the activation energy using ln(rate) .
Answer
| ||||
| ||||
| ||||
|
Plot against
Draw a line of best fit
Calculate the gradient
Activation energy in
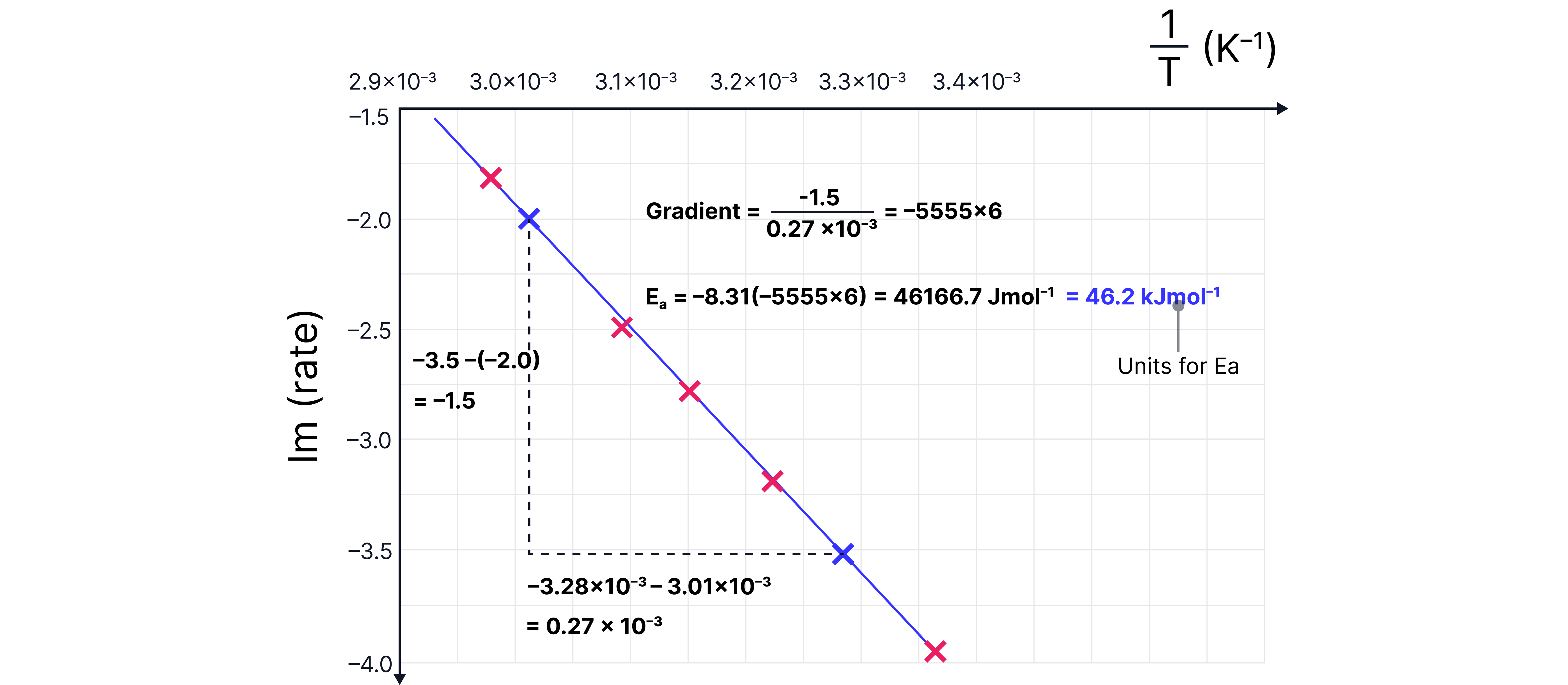
Reminders:
Always convert temperature from Celsius to Kelvin
Activation energy in the Arrhenius equation is in . If asked in , the divide by 1000