The Ideal Gas Equation
Lajoy Tucker
Teacher

Introduction & Definitions
The Ideal Gas Equation is:
Where:
pressure
volume
number of moles
ideal gas constant
temperature
Note: Students are not required to recall the value of in exams—it is always given.
Basic Principles
This equation links pressure, volume, temperature and amount of gas in a sample.
It applies to ideal gases—theoretical gases that obey all gas laws under all conditions.
Useful for calculating moles of a gas, or finding volume, pressure or temperature under specified conditions.
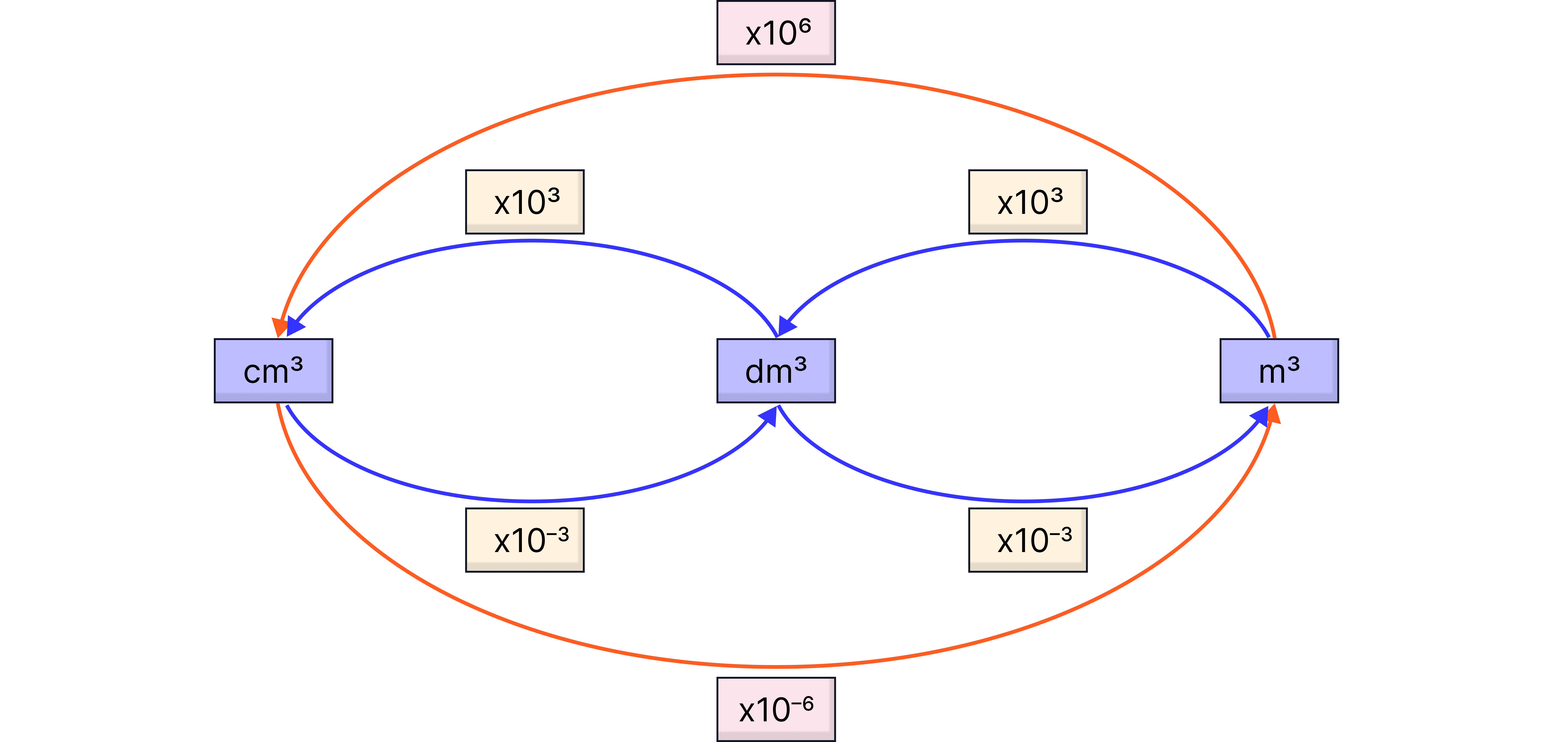
SI Units Required:
Quantity | Symbol | Unit | Conversion |
Pressure | |||
Volume | |||
Temperature | |||
Gas Constant | Given in exam: 8.31 |
The most commonly confused unit is volume
Using and Rearranging the Ideal Gas Equation
The formula can be rearranged depending on the variable required:
To find moles:
To find volume:
To find temperature:
Worked Examples
Calculating Moles of a Gas
A sample of nitrogen gas has a volume of at and . Calculate the number of moles.
Answer
Finding the Volume of a Gas
Calculate the volume occupied by of at and .
Answer
Finding Temperature
A gas occupies at . If there are of the gas, find the temperature.
Answer
Using molar ratios
of calcium nitrate is heated until it fully decomposes. Calculate the total volume of gas collected in at and .
Answer
Moles of calcium nitrate
Moles of gas formed
From the equation,
So gas per 1 mol Ca(NO3)2 = 5/2 = 2.5 mol
n(gas)
Volume of gas (ideal gas equation)
Use
Practical Applications
Determining the molar mass of a volatile liquid by measuring mass, temperature, and volume of vapour produced.
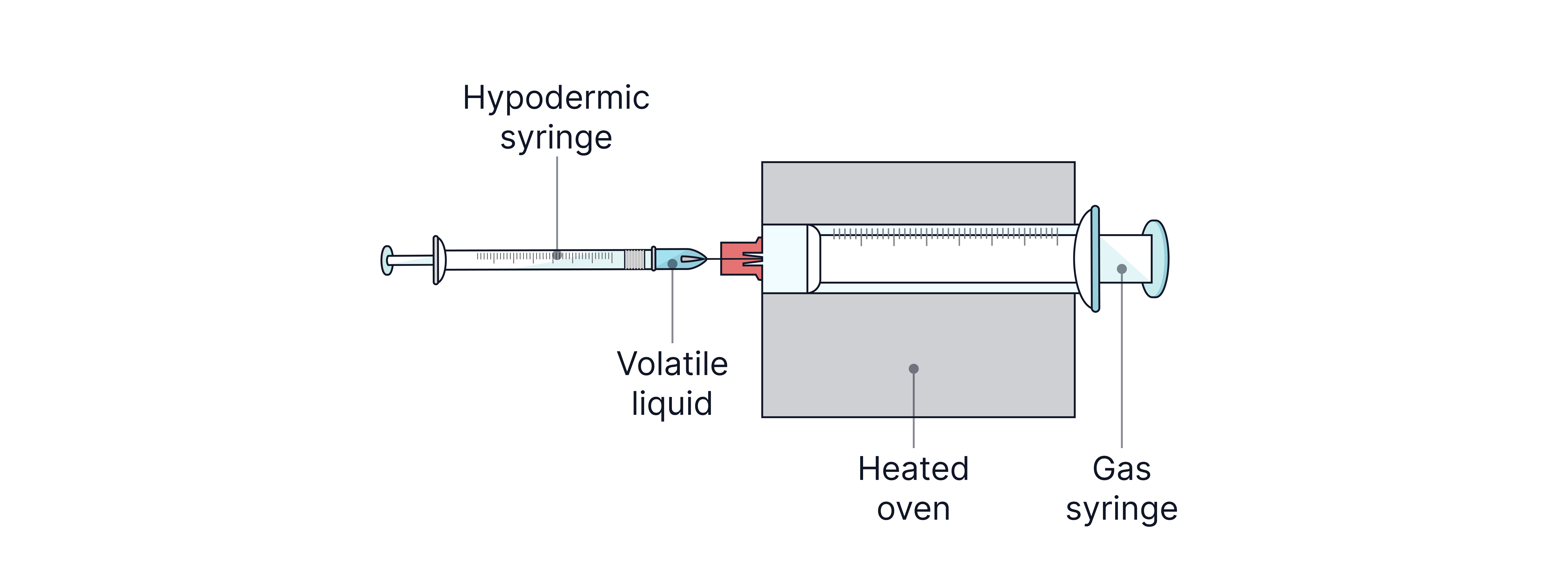
To determine the relative molecular mass, , of an unknown volatile liquid, a hypodermic syringe is used to inject a sample of the liquid into a gas syringe in an oven. The liquid vaporises adds to the volume being occupied in the gas syringe.
The data collected can be used to calculate the .
Initial mass of hypodermic syringe and liquid | |
Final mass of hypodermic syringe and liquid | |
Temperature of oven | |
Pressure | |
Increase in volume in gas syringe |
Answer
= mass / moles
Use
Use = mass / moles
Mass
Key Tips & Reminders
Convert:
Volume to
Temperature to
Pressure to
Final answers should match the least number of significant figures from the data.
Use molar ratios if the species in question is different to the one you have values for (e.g. Example 4 above)