Reacting Solutions (including Titration Calculations)
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
This topic focuses on calculating the amount of substance in a solution and how to use this in chemical reactions.
Concentration = quantity/volume
Mass concentration, = mass dissolved per of solvent
Concentration = mass /volume
Molar concentration, , number of moles dissolved per of solvent
Concentration = moles /volume
This is often rearranged to calculate the number of moles dissolved in solution:
Moles = concentration × volume
moles (mol)
concentration
volume

Important conversions:
to : divide by 1000
Example
of were dissolved in of water. Give the concentration in .
Answer
Volume
Example
Calculate the number of moles of in of a solution
Answer
Volume
Dilutions
Adding water to a solution increases the volume of the solvent and decreases the concentration.
The number of moles of the solute remains unchanged
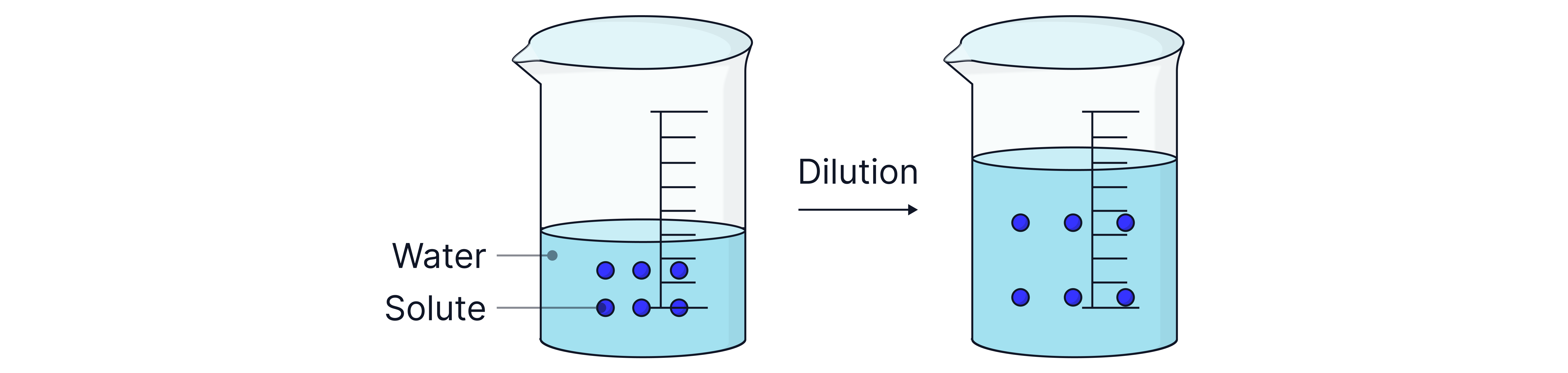
moles of solute in original solution
moles of solute in diluted solution
Therefore,
concentration of original solution
volume of original solution
concentration of diluted solution
volume of diluted solution
To calculate the new concentration of the solution after dilution, the expression above can be rearranged to .
Example
What is the concentration of the solution formed when of a glucose solution is diluted to a volume of .
Answer
You may also just spot the volume has increased by a factor of 6 and therefore, the concentration will decrease by a factor of 6.
Steps for Reacting Solutions
When calculating amount of substance for reacting solutions, the same principle is applied as for reacting masses
Write the balanced chemical equation.
Calculate moles of the known solution (using ).
Use mole ratios to find moles of unknown reactant or product.
Use moles to calculate the unknown quantity as required.
Example
What volume of is required to react with of ?
Equation:
Answer
Moles of
Ratio
Moles of
Volume of
Titrations
Titrations are often used to determine an unknown quantity (e.g. concentration, Mr, percentage purity).
A standard solution, where the concentration is known, reacts with a solution of which a quantity is unknown. By determining the exact volumes of each solution that react with each other unknowns can be calculated.
Practical details for making up standard solutions and carrying out a titration are covered in the RP1 revision notes.
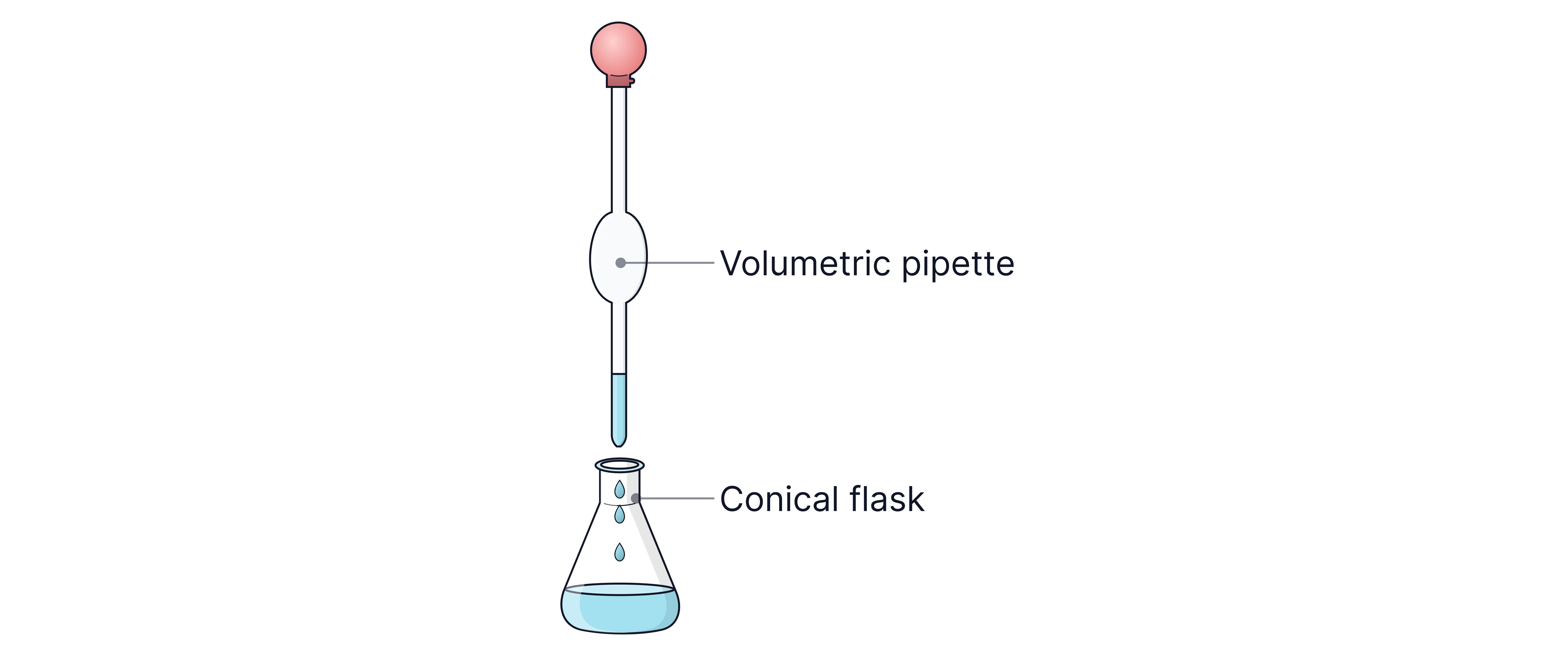
A known volume of a solution of unknown concentration is measured into a conical flask.
A few drops of indicator are added
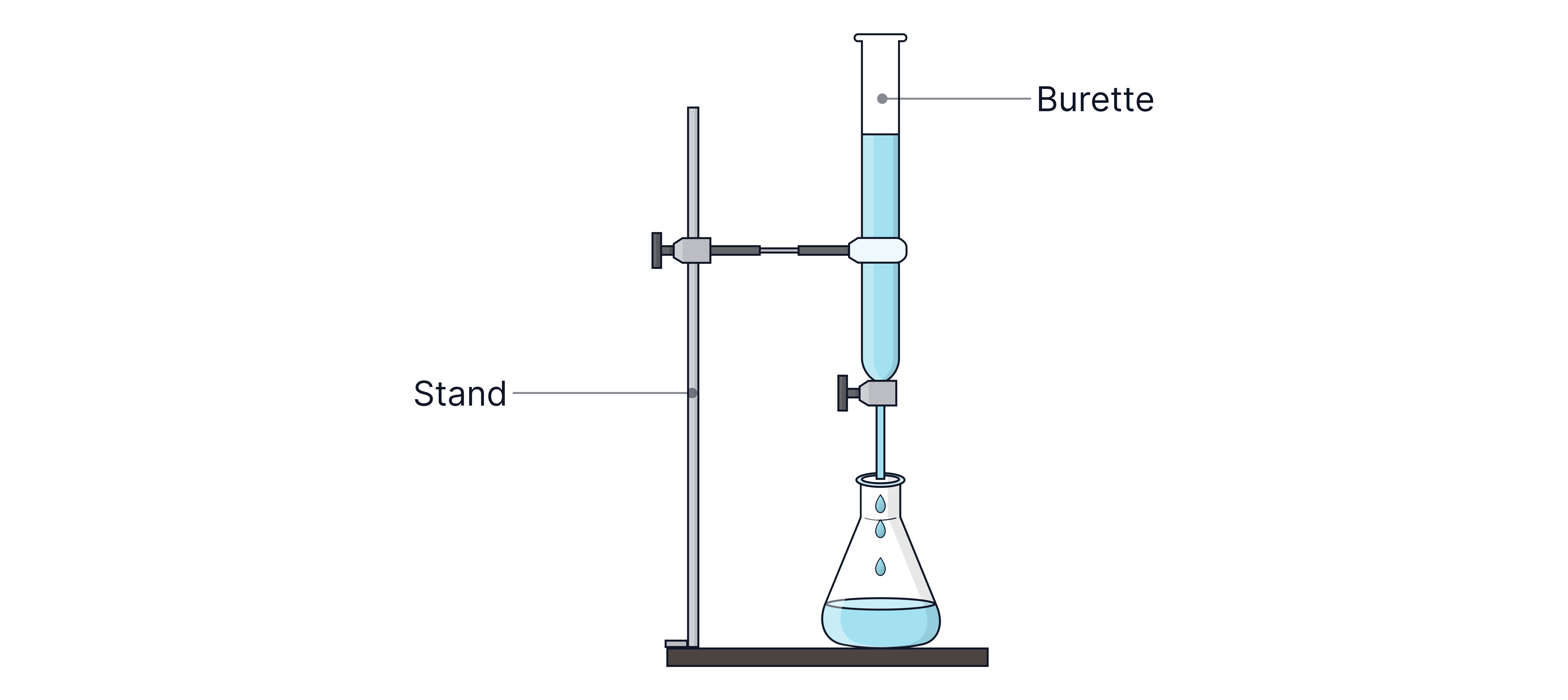
A solution of known concentration in the burette is added to the flask until the indicator changes colour permanently
Example: Simple titration
of is required to react completely with , Calculate the concentration of the solution.
Equation:
Answer
Moles of
Moles of (1:1 ratio)
Concentration of
Example: Water of crystallisation
A sample of hydrated sodium carbonate is dissolved in water and made up to in a volumetric flask.
A sample of this solution required of hydrochloric acid for complete neutralisation.
Determine the value of in . Give your answer as an integer.
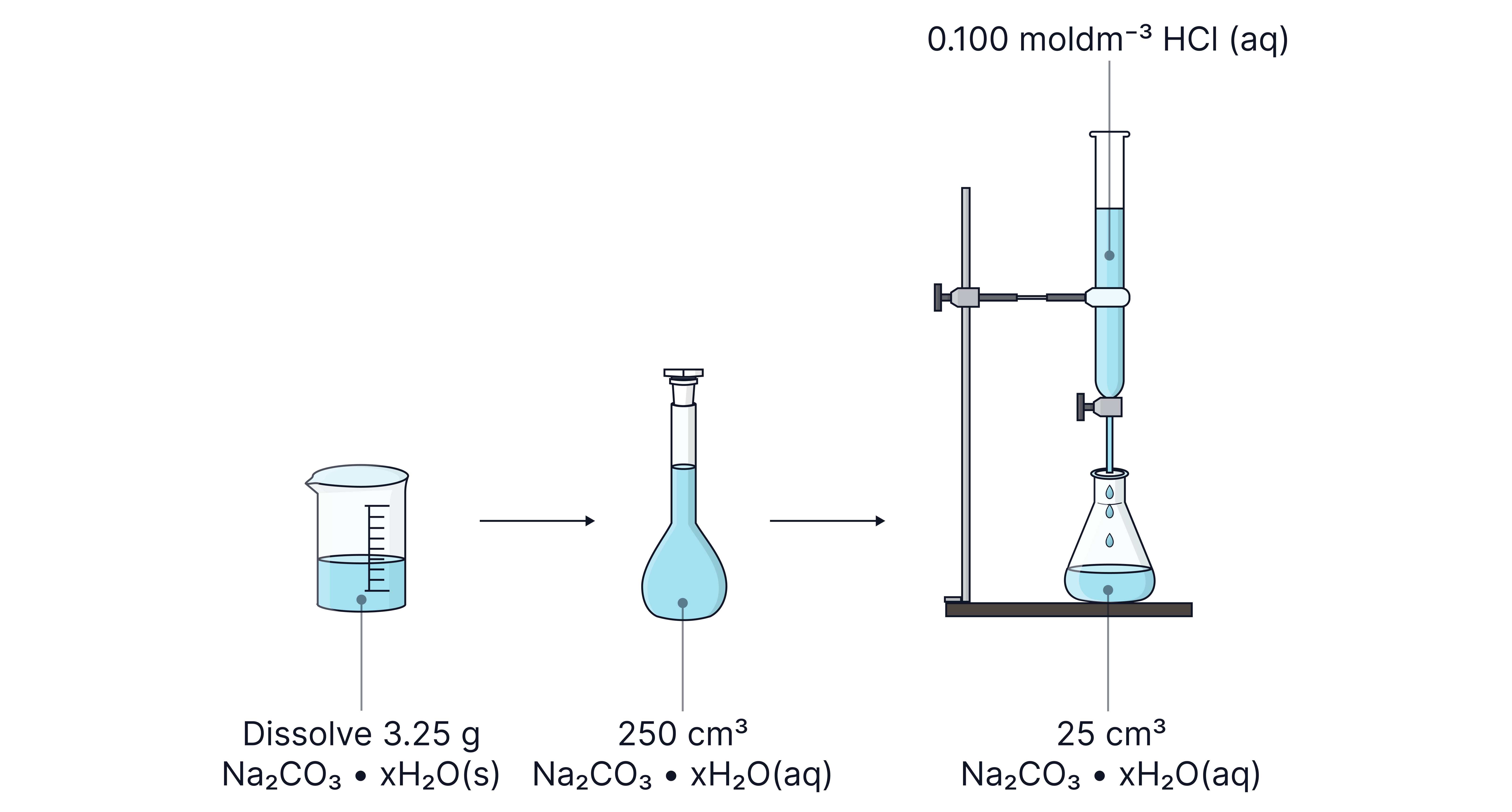
Answer
When determining , start by calculating the Mr of the hydrated salt as:
1. Calculate moles of from provided concentration and volume
2. Use molar ratio to determine moles of hydrated salt
hydrated salt in
3. Scale the moles to the number in the original solution
hydrated salt in
Note – only 10% of the solution (and therefore 10% of the moles) in the volumetric flask was transferred to the conical flask for the titration. To determine the number of moles of the salt in the original sample, the moles must therefore be multiplied by 10.
4. Calculate Mr of hydrated salt
5. Calculate by subtracting Mr of
6. Calculate by divding by the Mr of
Back Titrations
We use back titrations if:
The analyte is volatile or an insoluble salt
The direct reaction is too slow for a practical direct titration
Weak acid – weak base reactions are involved (no suitable indicators)
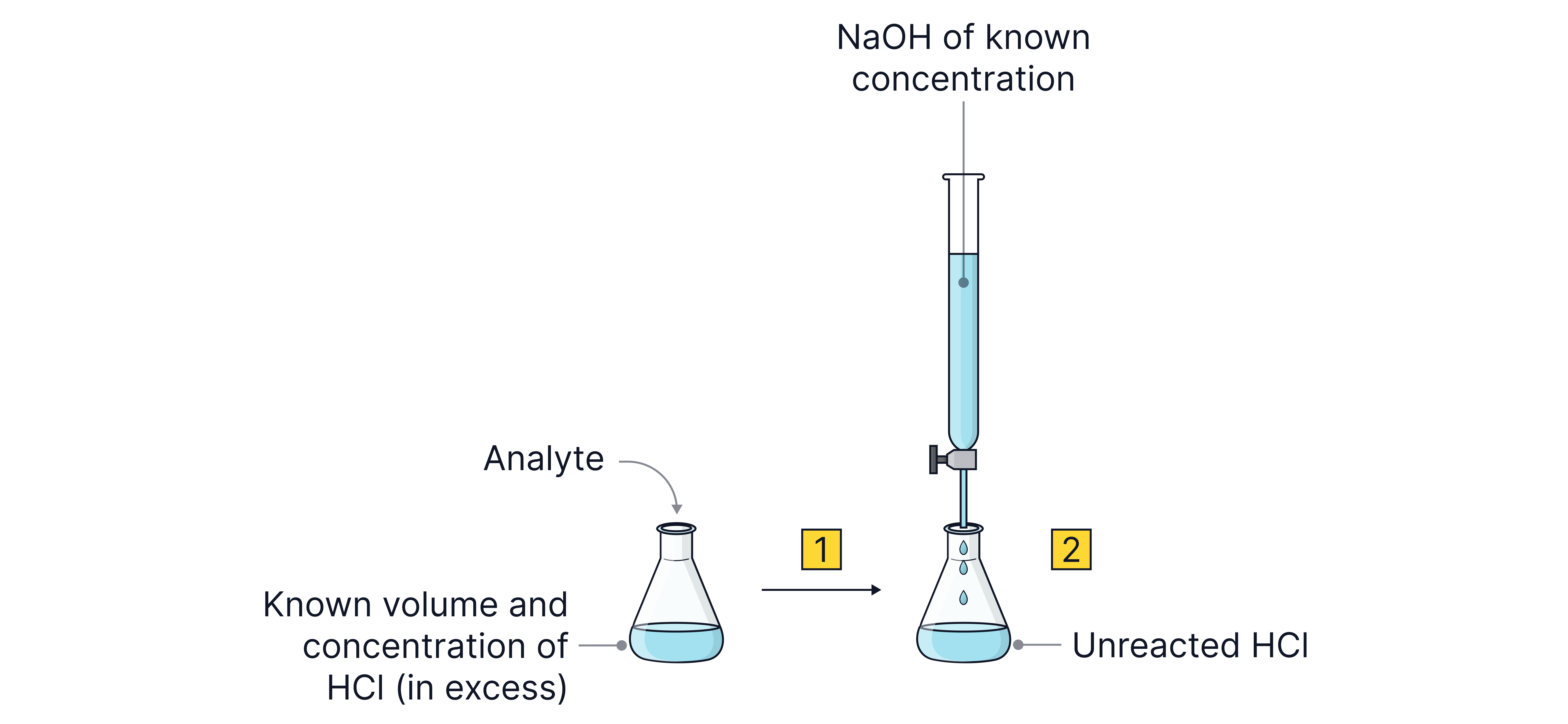
1. React the analyte with a known excess of an acid or base e.g. .
2. Titrate the leftover acid or base to determine the number of unreacted moles.
3. Subtract the leftover moles from the added moles to determine the number of moles that reacted.
4. Use the molar ratio in the balanced symbol equation determine the number of moles of analyte that reacted.
5. Use the moles to calculate the final quantity (mass, Mr, water of crystallisation, purity etc.
Worked Example
An impure sample of barium hydroxide, of mass was added to of hydrochloric acid.
All the barium hydroxide reacted. The excess of acid needed of sodium hydroxide for neutralisation.
Calculate the percentage purity of the sample of barium hydroxide.
Answer
1. React the analyte with a known excess of an acid or base e.g.
Known excess of added
2. Titrate the leftover acid or base to determine the number of unreacted moles
Moles of required to neutralise unreacted
Moles unreacted
3. Subtract the leftover moles from the added moles to determine the number of moles that reacted.
Moles of that reacted with
4. Use the molar ratio in the balanced symbol equation determine the number of moles of analyte that reacted.
Moles
5. Percentage purity = (mass /mass sample) x 100
Mass
Percentage purity
Key Tips & Reminders
Always convert to before using
Ensure the equation is balanced to get the correct mole ratio.
Use correct significant figures based on data provided.
Label units throughout your working — especially in answers.
Show your working out/calculations! If the final answer is wrong, you can still get some marks.
When in doubt… MOLES