Trends and Reactions of Halogens
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Group 7 elements (halogens): Fluorine, chlorine, bromine, iodine, and astatine – non-metals with similar chemical properties due to having seven outer electrons.
Displacement reaction: A more reactive halogen displaces a less reactive halide from its salt.
Halide ions: Negatively charged ions formed when halogens gain an electron (e.g., ).
Principles
Halogens exist as diatomic molecules .
They react via redox reactions, where the halogen is reduced to a halide ion.
Trends are observed down the group in:
Boiling points
Electronegativity
Oxidising power
Physical Properties of Halogens
Boiling points increase down the group due to:
Greater van der Waals forces (larger molecules with more electrons).
More energy required to separate molecules.
| Halogen | Standard state | Colour | Colour in aqueous solution |
| Flourine | Gas | Pale yellow | |
| Chlorine | Gas | Pale green | Pale green |
| Bromine | Liquid | Red-brown | Orange |
| Iodine | Solid | Grey | Brown |
Note that solid iodine easily vaporises to iodine gas which is a purple vapour.
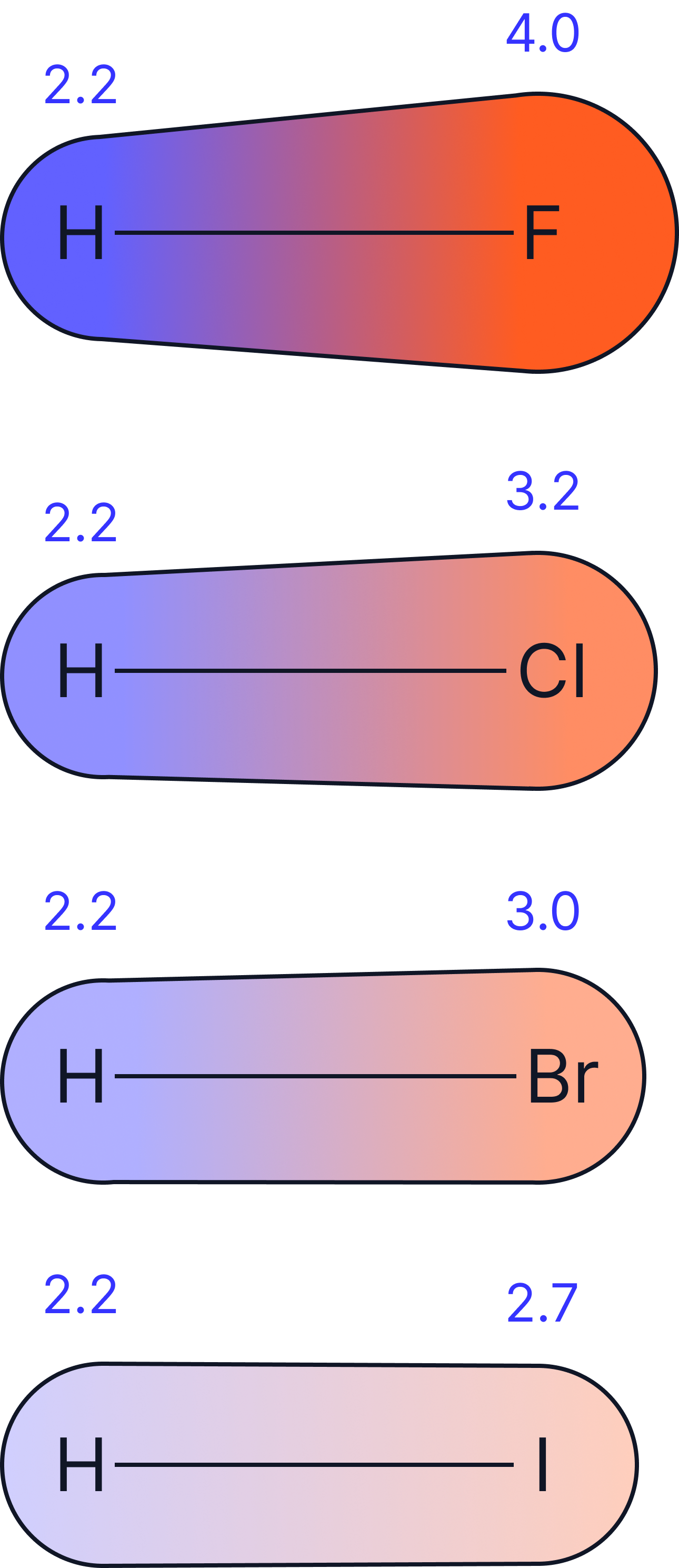
Electronegativity of Halogens
Electronegativity is the power of an atom to attract a pair of electrons towards itself while in a covalent bond.
Electronegativity decreases down group 7.
As atomic radius and shielding increases, the attraction between the nucleus and the shared electrons decreases.
Fluorine is the most electronegative element on the periodic table.
Oxidising Power of Halogens
Oxidising ability decreases down the group as the halogens are less likely to be reduced (gain electrons) themselves.
This means:
Fluorine is the strongest oxidising agent.
Iodine is the weakest.
Reason: Atomic radius and shielding increase down the group → harder to attract electrons.

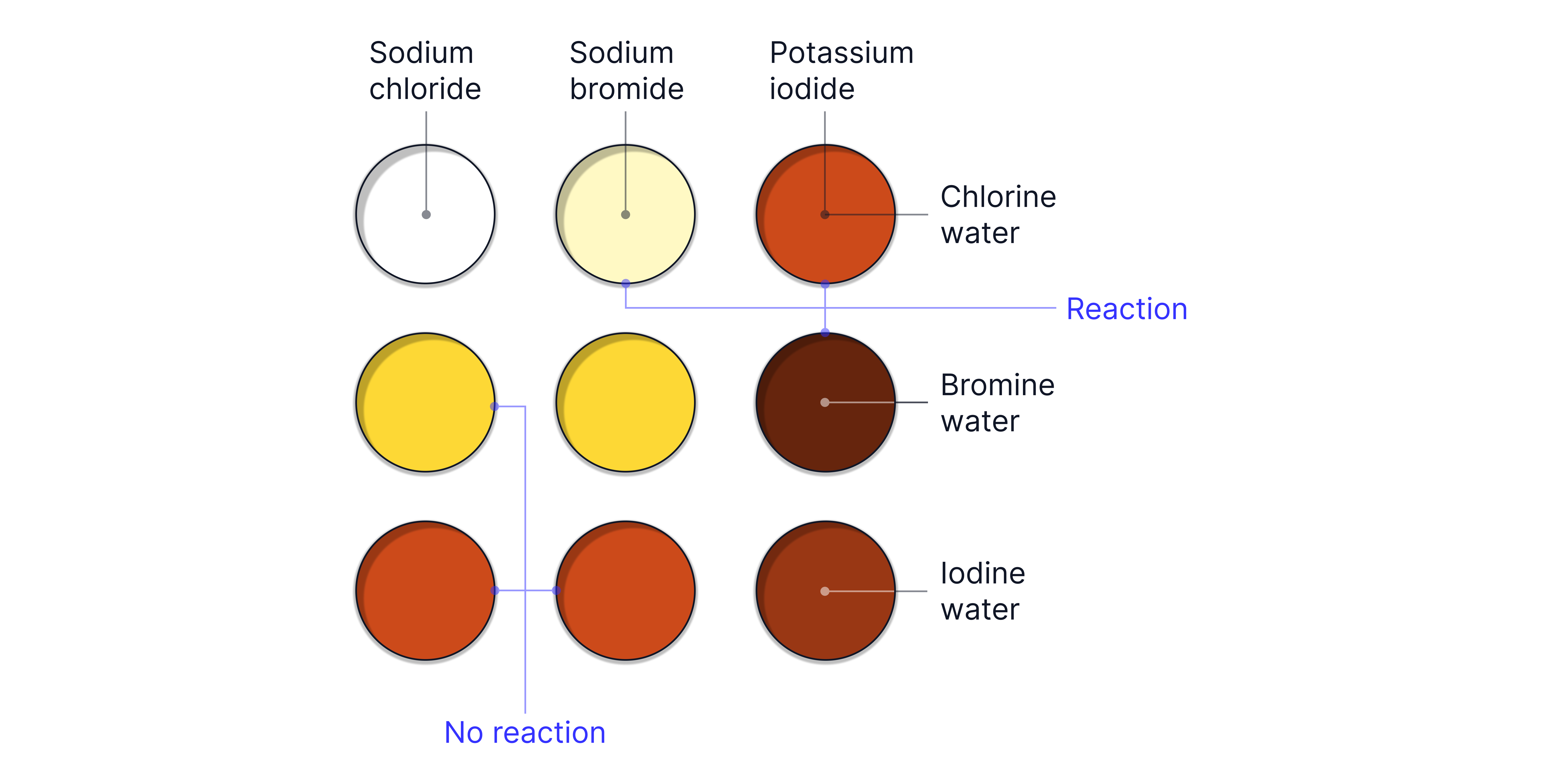
Displacement Reactions
The oxidising ability of the halogens can be demonstrated experimentally through displacement reactions.
A more reactive (more strongly oxidising) halogen displaces a less reactive halide from its compound.
Example
This is a redox reaction as the is reduced from 0 to -1 and the is oxidised from -1 to 0.
A colour change is observed if a reaction takes place as the halogen in solution determines its colour.
Example ionic equations (without spectator ions):
Reaction | Observation | |
|---|---|---|
Pale green to orange solution | ||
Pale green to brown solution | ||
Orange to brown solution | ||
Solution stays brown ( is less oxidising than ) |
Practice Questions
Write the ionic equation for the reaction between chlorine water and potassium bromide.
Answer
Chlorine displaces bromide. Chlorine is reduced, bromide oxidised.
Explain why the boiling point of iodine is higher than that of chlorine. (2 marks)
Answer
Iodine has more electrons than chlorine
So stronger van der Waals forces
More energy needed to break intermolecular forces
Key Tips
Use the correct language. The halogens (fluorine, chlorine, bromine, iodine) vs the halide ions (fluoride, chloride, bromide, iodide)
When describing colour changes make sure to include the initial and the final colour.