Reactions of Halides
Lajoy Tucker
Teacher

Introduction & Definitions
Halide ions : Anions formed when halogens gain an electron (e.g. ).
Reducing agent: A species that donates electrons and is itself oxidised.
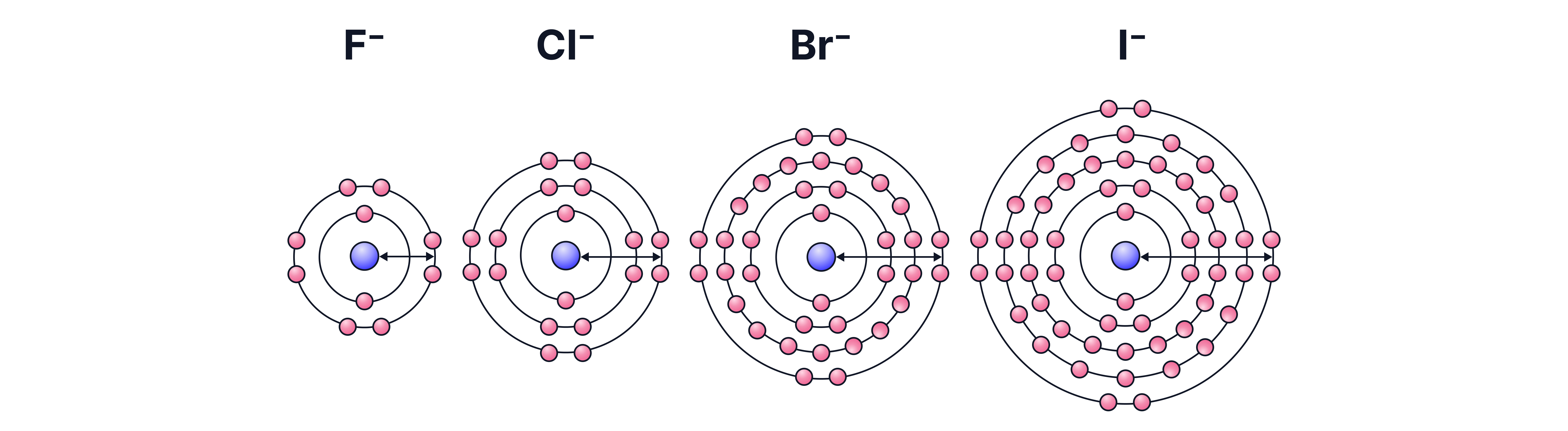
Reducing Power of Halide Ions
Increases down the group:
Reason: Ionic radius and shielding increase → weaker attraction to outer electron → easier to donate electrons (i.e. reduce something else).
Reactions of Solid with Concentrated
The trend in reducing ability can be demonstrated experimentally through reaction of the solid sodium halides with concentrated sulfuric acid.
The more the oxidation state of the in is decreased, the stronger the reducing ability of the halide ion must be.
(solid sodium chloride)
The chloride ion is a very weak reducing agent. The oxidation state of the sulfur has not changed. This is an acid-base reaction where the acts as an acid ( donor) and the acts as a base ( acceptor).
Steamy fumes of are observed.
(solid sodium bromide)
The first reaction is the same as with .
As is a stronger reducing agent, the reacts with further to produce sulfur dioxide, .
Half equations:
Overall:
Note: Do not just learn these equations by heart. Practice constructing half equations and overall redox equations. (link to redox equations note)
The has been reduced from +6 in to +4 in
Steamy fumes of , the orange/brown vapour , and a choking gas are all observed.
(solid sodium iodide)
The first reaction is the same as with and
As is a stronger reducing agent, the reacts with further to produce sulfur dioxide, , elemental sulfur, , and hydrogen sulfide,
Example half equations:
Overall:
Steamy fumes of and the purple vapour, are observed.
The third observation depends on which sulfur-containing product is formed.
| Sulfur-containing product | Observation |
| Choking gas | |
| Yellow solid | |
| Rotten egg smelling gas |
Iodide ions can reduce the in from +6 down to -2 in demonstrating its position as the strongest reducing agent.
Halide | Sulfur containing product | Oxidation state of S in product |
|---|---|---|
+6 | ||
+4 | ||
+4, 0, -2 |
Weakest reducing agent
Strongest reducing agent
In general:
Reaction 1 – Acid-base
Reaction 2 – Redox ( and only)
__ __ __ sulfur-containing product
Note – can also be written as
Note – You will not be expected to include multiple sulfur-containing products in the same equation. In exam questions, the observations are often used to communicate which one to include in the equation. For example, if a rotten smelling gas is mentioned, the equation must have .
Practice Question
Write the half equations for the reaction between sodium iodide and concentrated sulfuric acid to produce a yellow solid.
Answer
Oxidation of :
Reduction of to ;
Summary and key tips
Halide | Reaction Type | Observations |
|---|---|---|
Acid-base only | Steamy fumes | |
Acid-base + redox | Steamy fumes , brown gas , choking gas | |
Acid-base + redox | Steamy fumes , purple vapour/black solid , yellow solid , bad egg smell |
Reducing ability of halide ions increases down the group. Oxidising ability of halogens decreases down the group
Be able to write ionic half-equations for all redox steps.
Link the sulfur-containing product to the oxidising ability of the halide by identifying the change in oxidation state of the