Uses & Reactions of Chlorine
Lajoy Tucker & Dr. Davinder Bhachu
Teachers


Contents
Definition
Chlorine () is a pale green toxic gas used industrially and domestically, particularly in water treatment.
Reactions of Chlorine
- With cold water (disproportionation)
Equation:
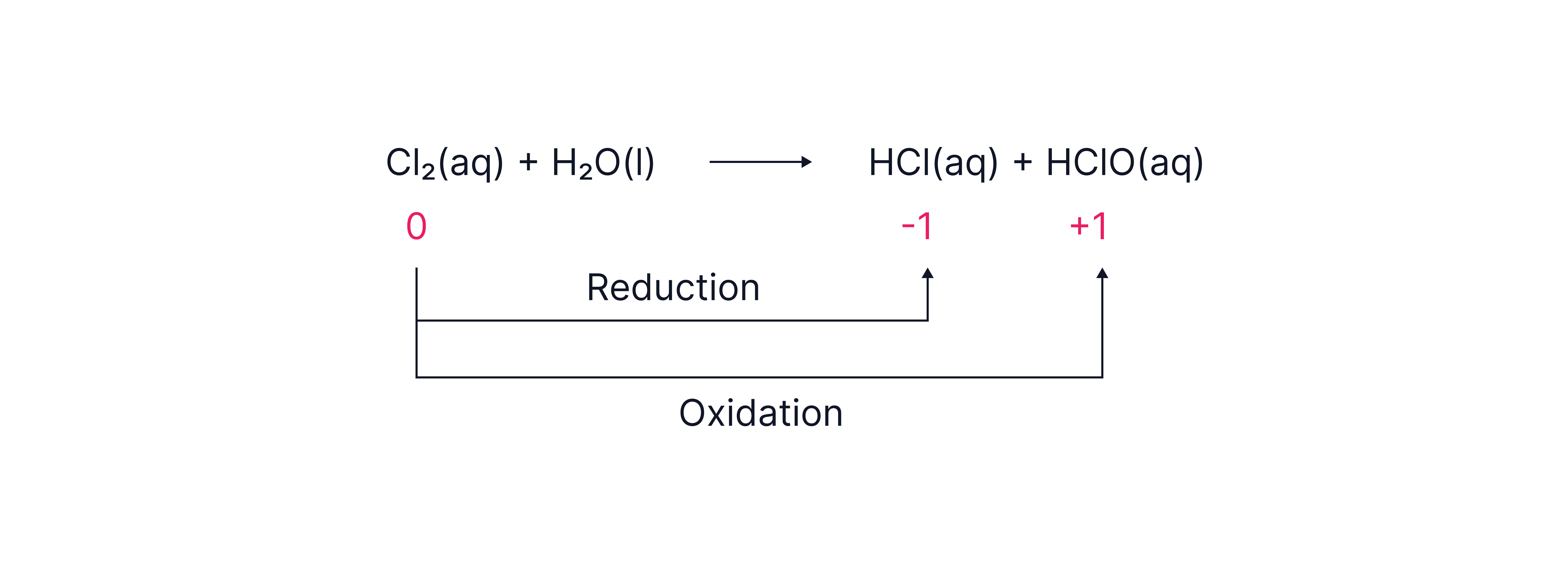
HCl: Chlorine is reduced from 0 to -1.
HClO (chloric(I) acid): Chlorine is oxidised from 0 to +1.
This is a disproportionation reaction, a reaction in which the same element in a single species is simultaneously oxidised and reduced.
Chloric(I) acid acts as a weak bleach and kills bacteria in water supplies.
- With water in the presence of sunlight
The same reaction as above takes place, however, in the presence of sunlight the chloric (I) acid (HClO) decomposes to hydrochloric acid (HCl) and oxygen gas ().
Equation:
Reaction of Chlorine with cold, dilute sodium hydroxide
Equation:
Sodium chlorate(I), NaClO, is the active ingredient in household bleach. Again, this is a disproportionation reaction.
NaCl: Chlorine is reduced from 0 to -1.
NaClO: Chlorine is oxidised from 0 to +1.
Benefits and Risks of Chlorine in Water Treatment
Benefits:
Destroys pathogenic microorganisms.
Prevents waterborne diseases (e.g. cholera, typhoid).
Maintains residual protection in water supply.
Risks:
Chlorine is toxic and corrosive.
Can react with organic compounds in water to form chlorinated hydrocarbons, some of which are carcinogenic (e.g. trihalomethanes).
Overall:
Despite the risks outlined above, chlorine is added to water in low enough concentrations that the benefit of disease prevention outweighs the risks associated with its toxicity.
Worked Example
What is the oxidation state of chlorine in each of the products of the following reaction?
Answer:
In NaCl: Cl = -1
In NaClO: Cl = +1
Questions
Question 1
Write the half-equations (in acidic conditions) to show the oxidation and reduction of chlorine that occurs when chlorine reacts with cold water.
Half equations:
Oxidation:
Reduction:
Question 2
Explain why the reaction in Q1 is a disproportionation reaction
The same element (chlorine) is oxidised from 0 in to +1 in and reduced from 0 in to -1 in .
Key Tips
In reactions with both cold water and sodium hydroxide solution, chlorine undergoes disproportionation
Always state oxidation states when explaining redox/disproportionation.
Memorise both equations for use in water and bleach production.