Millikan’s oil drop
Brook Edgar & Hannah Shuter
Teacher


Contents
Explainer Video
Stokes’ law (drag on a small sphere moving through air)
Millikan measured the charge on tiny oil droplets and found the charges always came in integer multiples of a smallest value. That smallest value is the elementary charge, . This means the electron is a “quantized” particle. i.e you can’t have half of on electron.
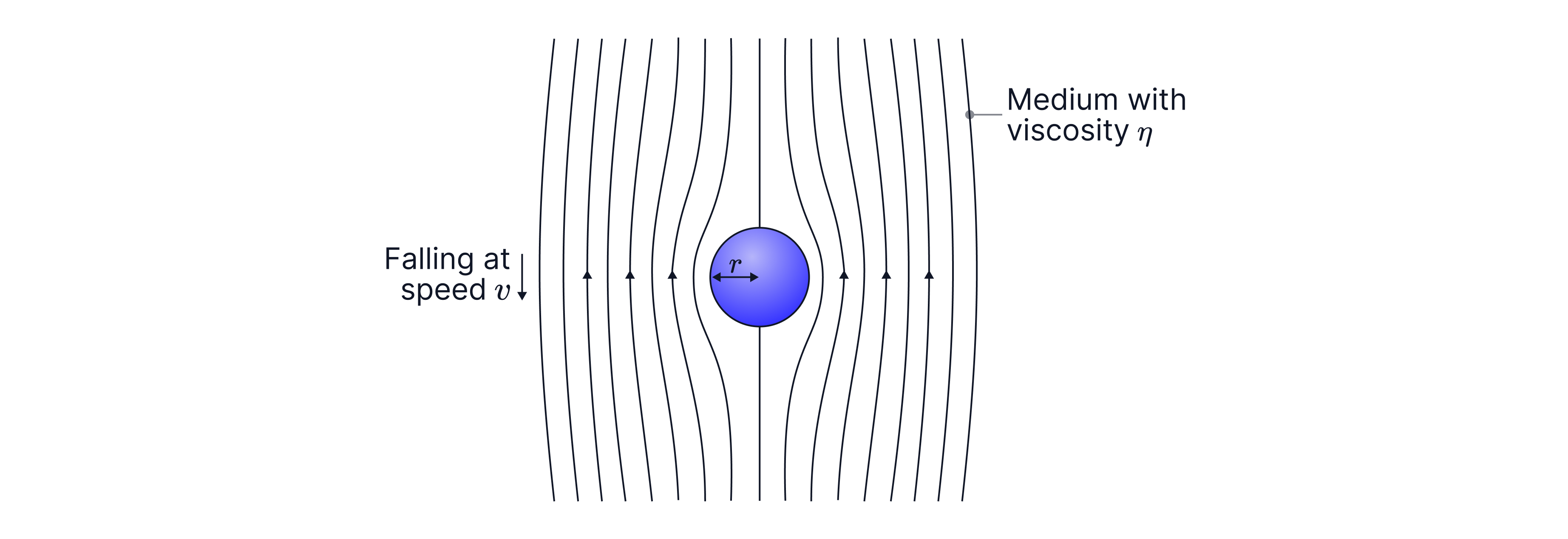
Imagine a tiny spherical droplet of radius r moving through air at speed .
For very small spheres moving slowly through a fluid (laminar flow), the viscous drag force is:
Formula:
This is sometimes known as Stoke's Law.
Using terminal velocity to find the droplet radius
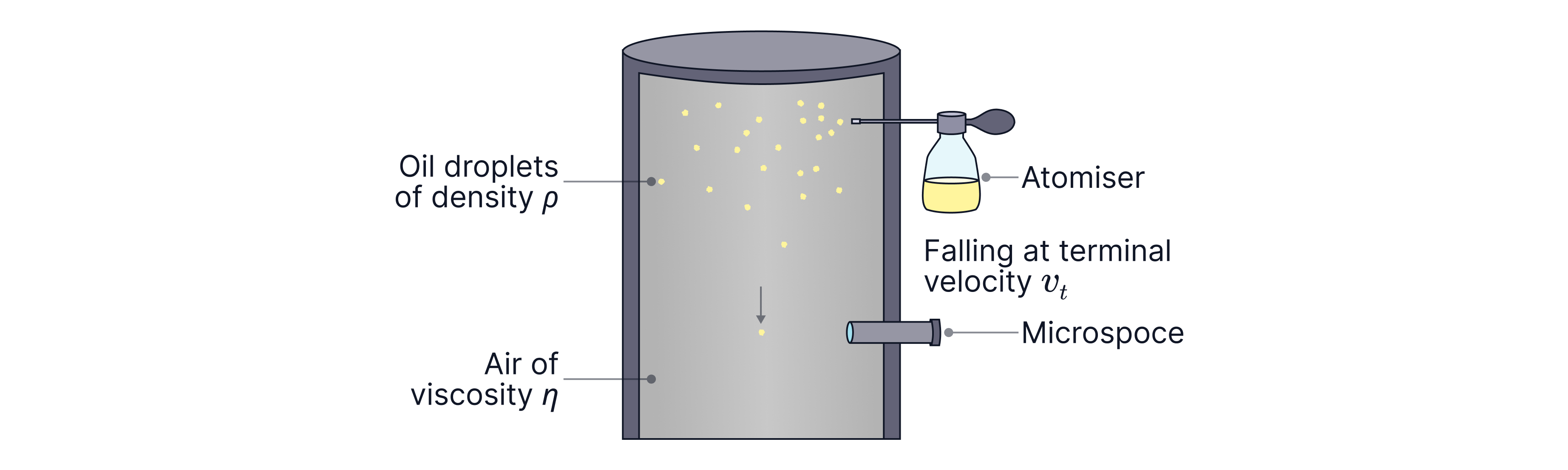
A droplet falls through air. After a short time it reaches terminal velocity (constant speed).
How terminal velocity was determined (method):
The droplet is viewed through a microscope with a calibrated scale. Millikan measures the time for the droplet to move a known distance (e.g. between two scale marks). At first the droplet speeds up, so the time per division changes. Once terminal velocity is reached, the time to travel equal distances becomes constant, so .
Forces on the droplet (falling):
• Weight (downward)
• Drag (upward)
At terminal velocity:
Resultant force = 0, so:
The oil droplet is a sphere so it’s mass is:
Joining this together can find the radius of the droplet.
Formula:
Charging the droplets and finding droplet charge
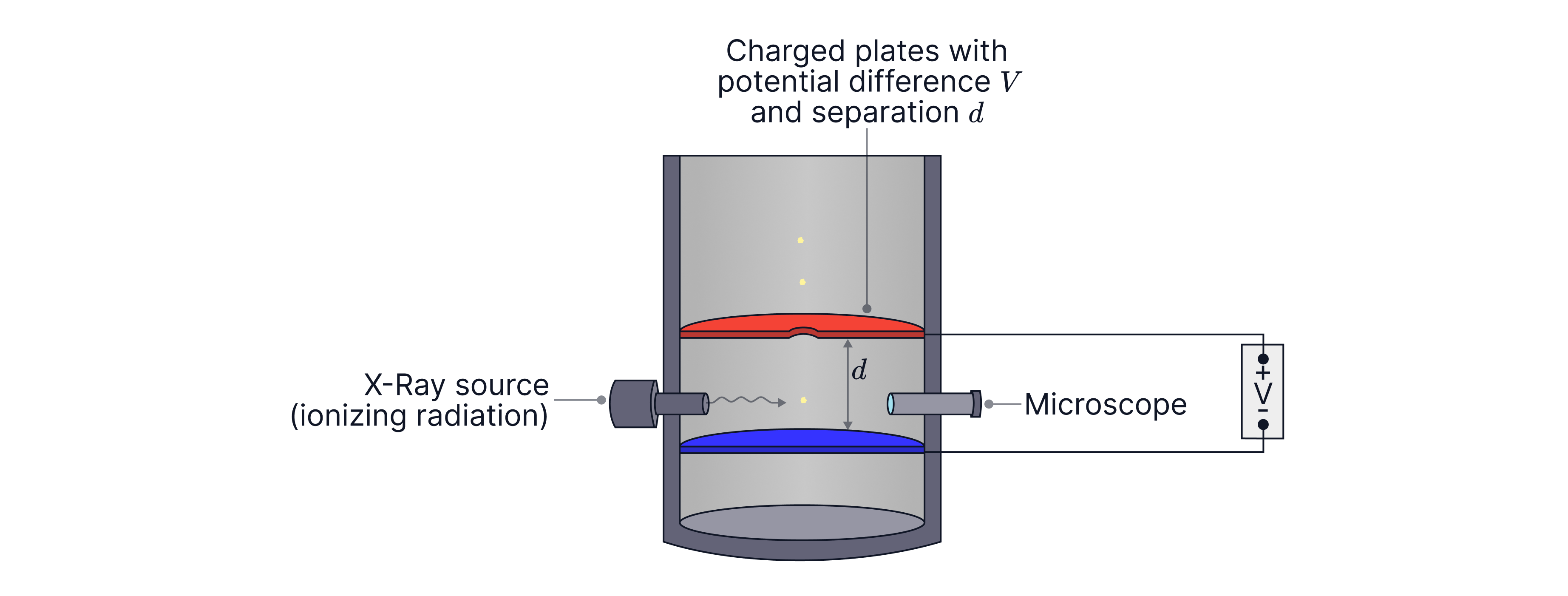
How Millikan produced charged droplets:
• Oil is sprayed from an atomiser. During this process droplets can become charged due to friction/contact effects as they pass through the nozzle.
• Millikan also used ionisation of the air using X-rays. This creates free electrons and ions in the air, which can attach to droplets, changing their charge.
Why the charge can change during the experiment:
Droplets may gain or lose electrons by collisions with ions/electrons in the air (especially when ionised), so the droplet charge can change in steps.
Using the electric field to find the droplet charge
After finding , a potential difference is applied across the plates to create an electric field. The field is adjusted so the droplet is held stationary.
From the eq. sheet:
This makes sense as the droplet is stationary, force due to electric field = force due to gravity
so:
Formula:
Significance
Millikan measured droplet charges and obtained values such as , , , , .
At first glance they look like some random mixed (positive/negative) numbers, but the magnitude of each is a whole-number multiple of . This showed charge is quantised:
Worked Example:
Describe how Millikan’s experiment allows the charge on a droplet to be found. Your answer should reference terminal velocity, Stokes’ law, charging the droplet, the electric field between plates, and holding the droplet stationary.
Answer:
Spray oil from an atomiser to create tiny droplets; droplets can become charged by friction/contact effects.
Use X-rays to ionise the air so droplets can gain/lose electrons, changing charge in steps.
Measure terminal speed as a droplet falls by timing it between microscope scale marks.
At terminal speed forces balance, so weight equals Stokes’ drag: .
Use this to calculate the droplet radius , and hence its mass.
Apply a p.d. across plates to create an electric field .
Adjust until the droplet is stationary, so .
Calculate the droplet charge . Repeating for many droplets shows charges are multiples of .
Worked Example:
A tiny oil droplet (density ) falls through air (viscosity ) and reaches a terminal speed of . Using Stokes’ law and the idea of terminal velocity, show that the radius of the droplet is of order .
The droplet is then held stationary between two parallel plates separated by when a p.d. of is applied. Using your value of r from part (a), calculate the charge q on the droplet.
Comment on your value of in terms of .
Answers:
So the droplet’s charge is the third multiple of e.
(An integer multiple of the elementary charge, e is expected)
Practice Questions
Summarise how the charge on an electron was discovered using oil drops
-> Check out Hannah's video explanation for more help.
Answer:
A tiny oil droplet is observed falling and its terminal speed is measured by timing it between microscope scale marks.
At terminal speed the forces balance, so the droplet’s weight is balanced by viscous drag (resultant force ).
Using Stokes’ law for the drag force, , the droplet radius r can be found.
From , the droplet’s volume (and hence mass) can be calculated, allowing mg to be determined.
A potential difference is applied across parallel plates to create a uniform electric field .
The field is adjusted until the droplet is stationary, so the electric force balances the weight: .
Rearranging gives the droplet charge: .
Repeating for many droplets shows the charges are multiples of a smallest value, identified as the electron charge.
Deduce how Millikan used these results to find the charge on an electron:
, , , , .
-> Check out Hannah's video explanation for more help.
Answer:
Millikan measured droplet charges and obtained values such as , , , , .
These values are all exact multiples of .
There were no “in-between” values, showing the charges were not continuous.
This provides evidence that electric charge is quantised — it comes in discrete packets.
The smallest packet corresponds to the electron charge: .