Wave particle duality
Brook Edgar & Hannah Shuter
Teachers


Contents
Explainer Video
De Broglie waves and electron diffraction
We’ve already seen that light shows both wave behaviour (diffraction/interference) and particle behaviour (photons). De Broglie suggested the reverse might also be true: particles such as electrons can have wave properties.
It turns out that electrons can diffract through tiny gaps/regular spacings in crystals (e.g. graphite). That diffraction is strong evidence that electrons behave as matter waves with a wavelength linked to their momentum.
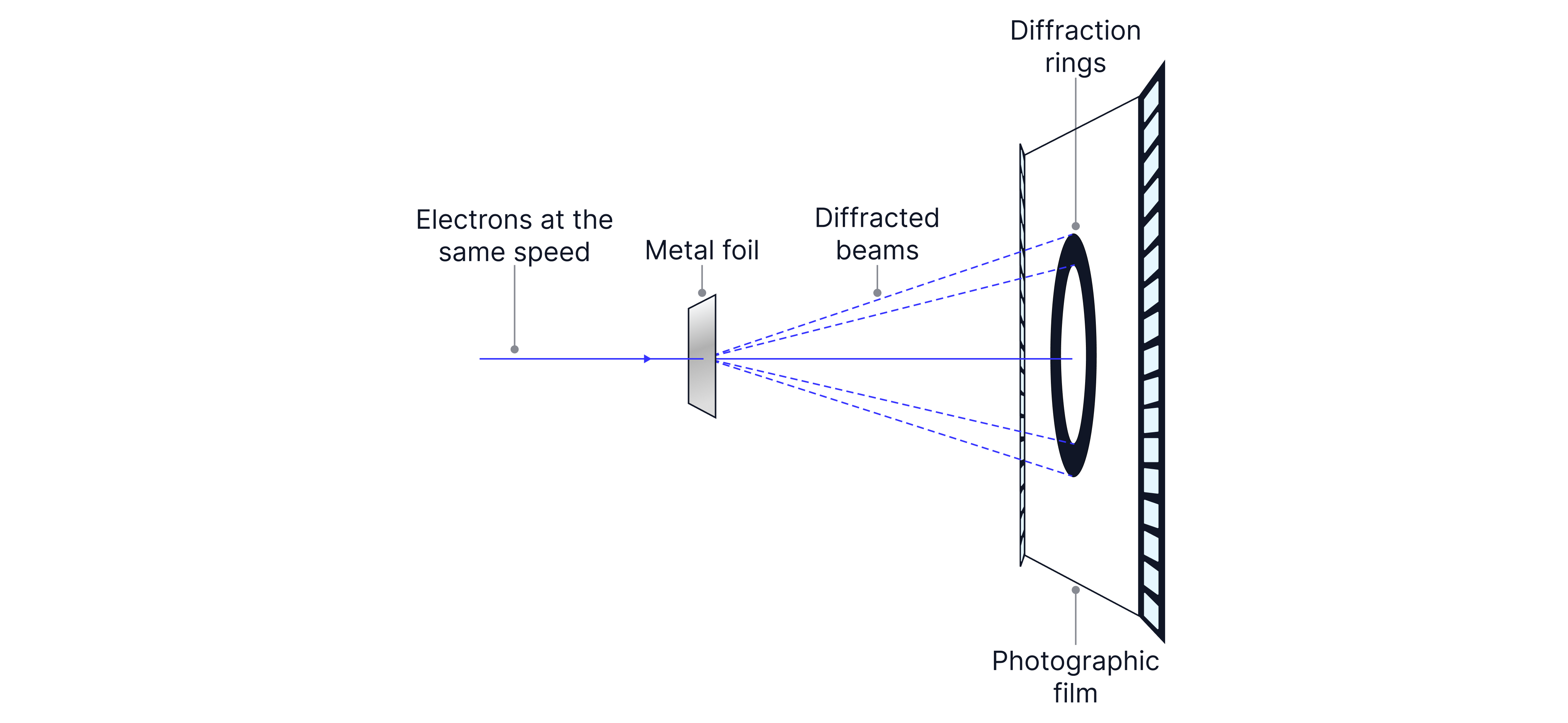
If electrons were only particles, you’d expect them to pass straight through. Instead, when a beam of electrons passes through a thin crystal (e.g. graphite), it produces diffraction patterns (often rings), just like waves diffracting through a grating.
Increasing the accelerating voltage makes the rings closer together, consistent with a smaller wavelength.
De Broglie proposed that a moving particle has an associated wavelength . For a non-relativistic particle, the momentum , where is the mass and is the speed:
Formula:
A charged particle accelerated through a potential difference gains kinetic energy equal to the electrical work done, , where is the charge on the particle:
Formula:
Rearranging gives the speed:
Substitute this into to write momentum in terms of , and :
Now substitute into to get the wavelength of a particle accelerated through a p.d.:
For an electron, and , so:
Electron microscopes Vs Light microscopes
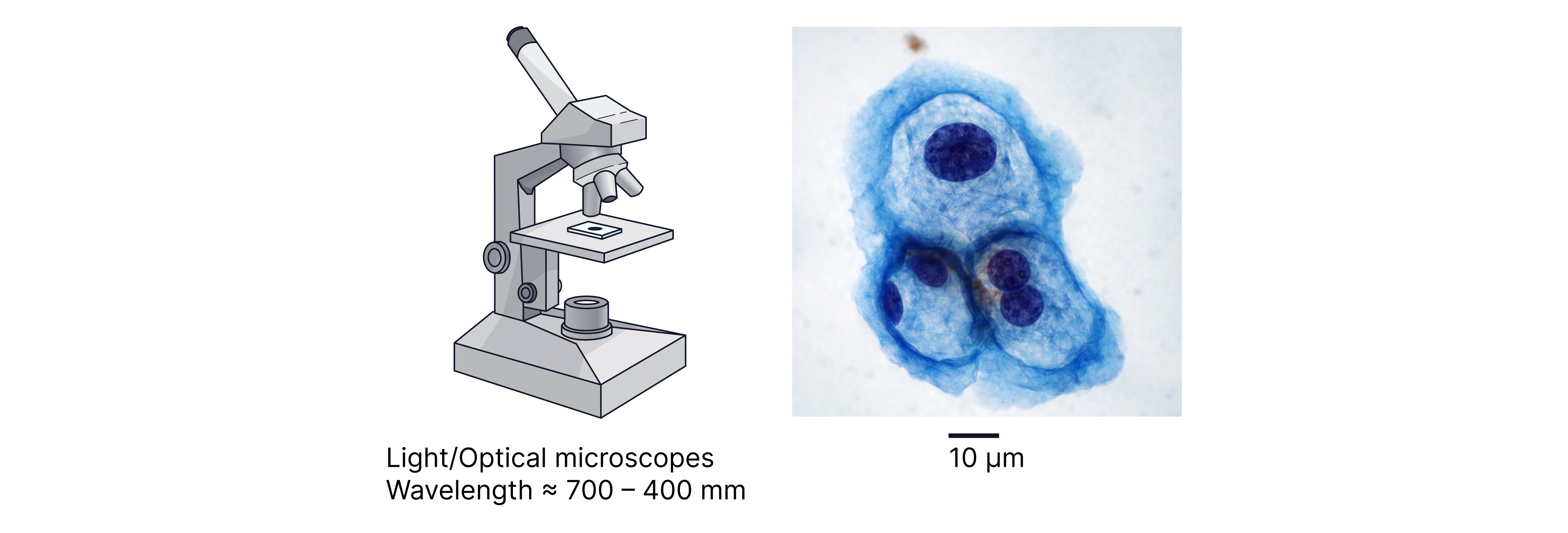
A microscope can only resolve details down to about the same order as the wavelength being used (shorter wavelength → better resolution). Visible light has wavelength roughly , so optical microscopes struggle to resolve features much smaller than this scale.
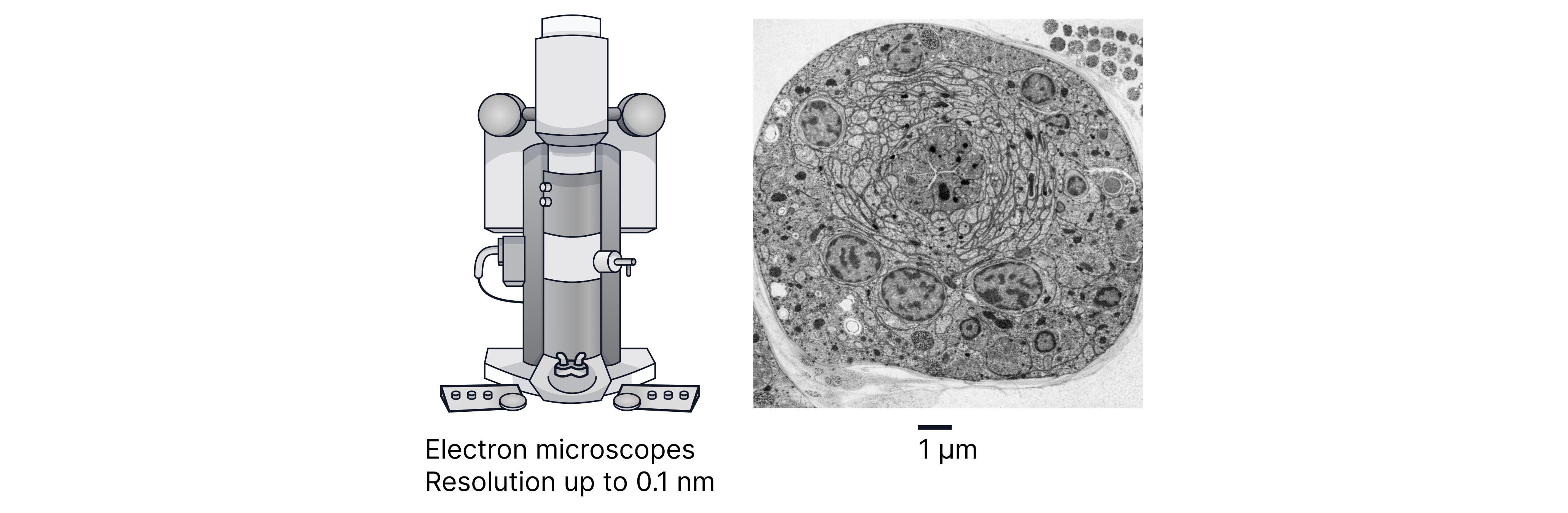
Electrons accelerated through large voltages can have wavelengths far smaller than visible light (and comparable to atomic spacings). That’s why electron microscopes can resolve much finer detail.
Transmission Electron Microscope (TEM)
Set-up:
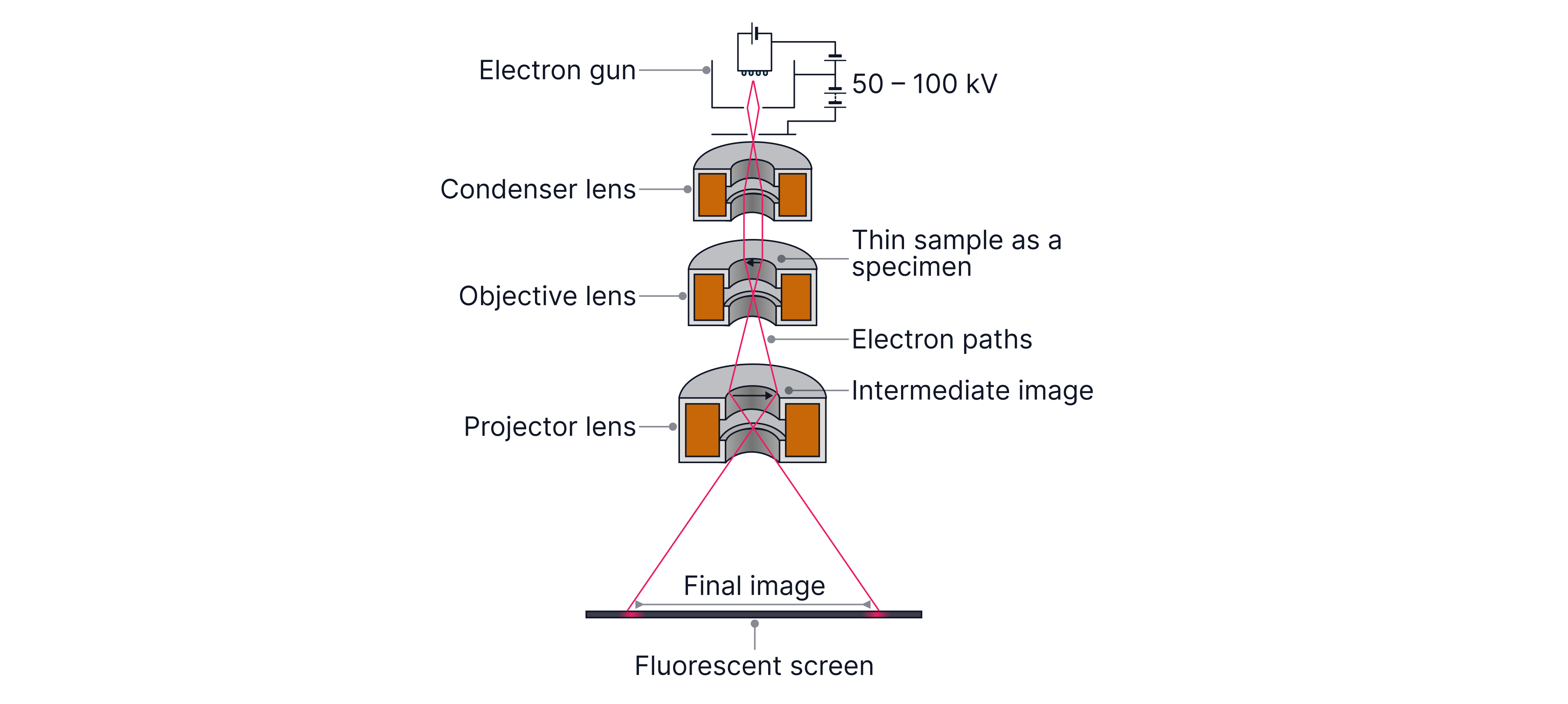
A transmission electron microscope (TEM) forms an image by sending a beam of fast electrons through an ultra-thin specimen in a vacuum.
Key components (what they do):
Electron gun: produces and accelerates electrons (typically tens to hundreds of ).
Condenser lens: forms a narrow parallel beams so the specimen is illuminated uniformly.
Objective lens: forms a magnified intermediate image of the specimen.
Projector lens: magnifies and focuses the image onto the screen/camera.
The lenses are magnetic (not glass) because the electrons are charged, so their paths can be bent and focused by magnetic fields.
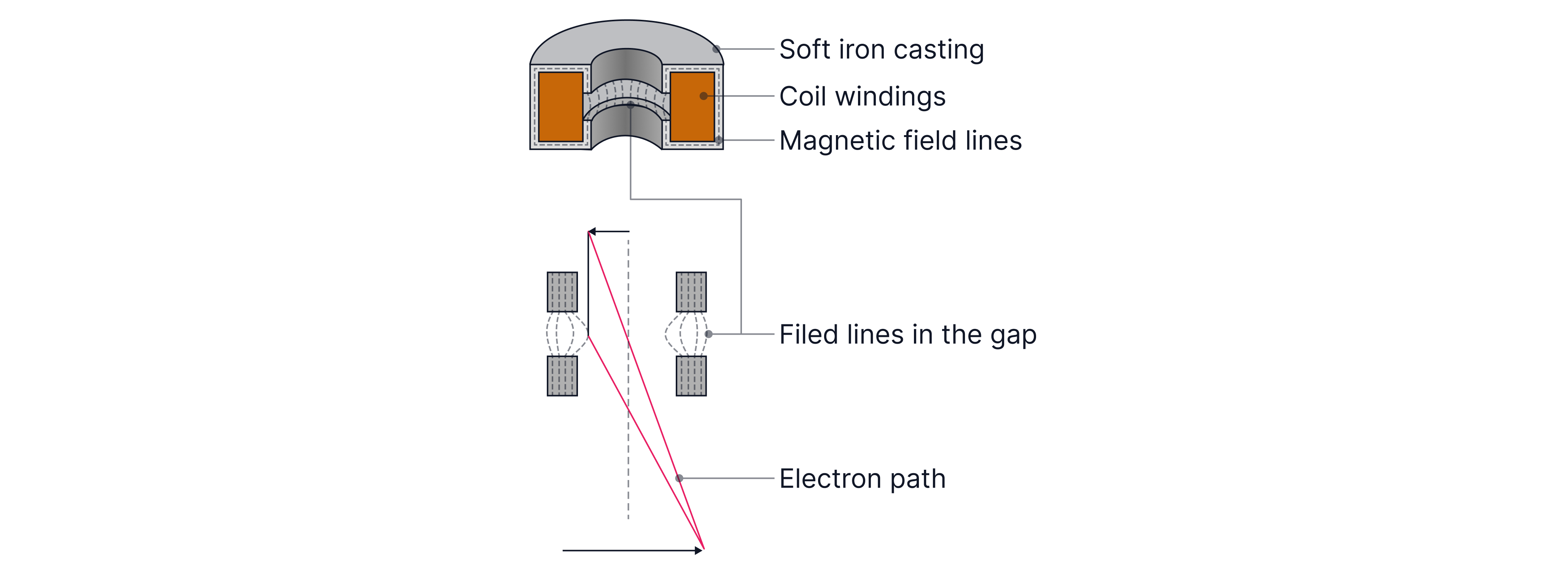
Image Resolution Limitation 1: Lens aberration
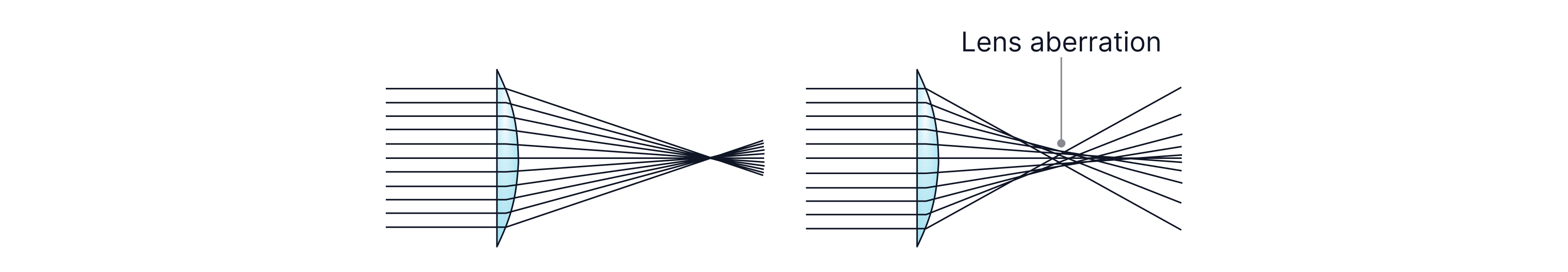
Lens aberration blurs the image because electrons originating from the same point on the sample aren’t always focused onto the same point in the image. This can be due to imperfections in the lens magnetic field or variations in the initial speeds of the electrons.
Image Resolution Limitation 2: Sample thickness
As electrons pass through a thicker specimen, they are more likely to lose energy / slow down. Slower electrons have larger de Broglie wavelength, reducing resolution. Also, the focusing becomes less consistent if electron speeds vary.
Worked Example:
An electron microscope accelerates electrons through a potential difference of .
Calculate the de Broglie wavelength of the electrons. Take , , .
Explain two reasons why the actual resolution is worse than the wavelength you calculate.
Answer:
Lens aberration: magnetic lenses do not focus all electrons from one point perfectly to the same image point → blurring.
Sample thickness / energy spread: electrons can lose energy and slow down, increasing wavelength and making focusing less consistent.
Scanning Tunnelling Microscope (STM)
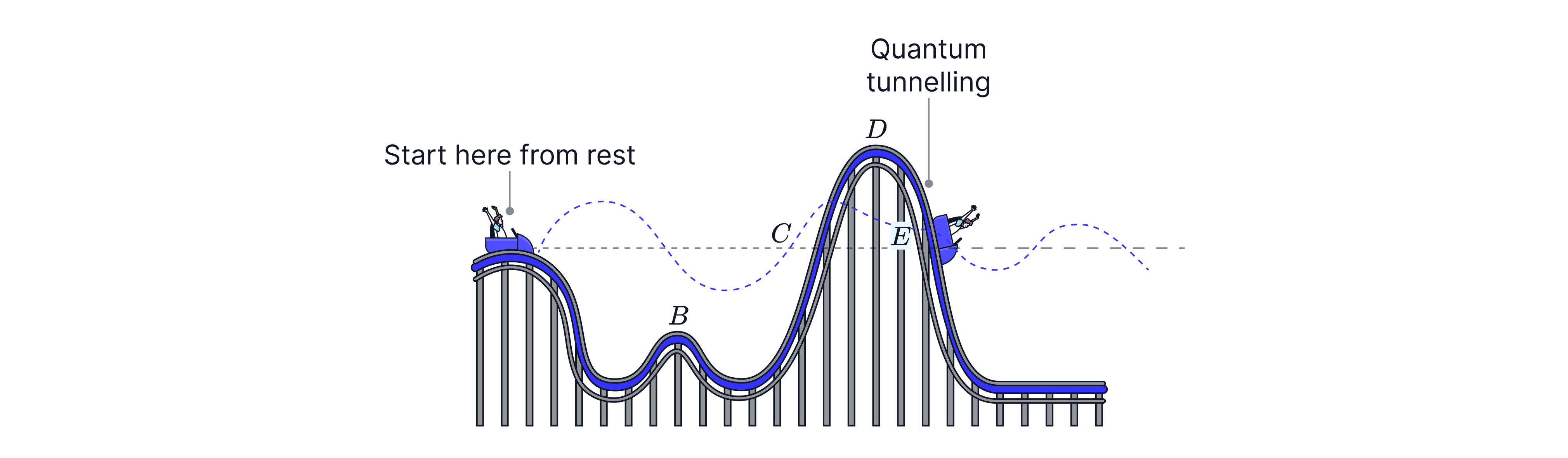
Classically, a particle without enough energy can’t get over a barrier. In quantum physics, electrons have wave properties, and there can be a small probability of appearing on the other side of a thin barrier — this is quantum tunnelling and this effect is used in Scanning Tunnelling Microscopes (STM).
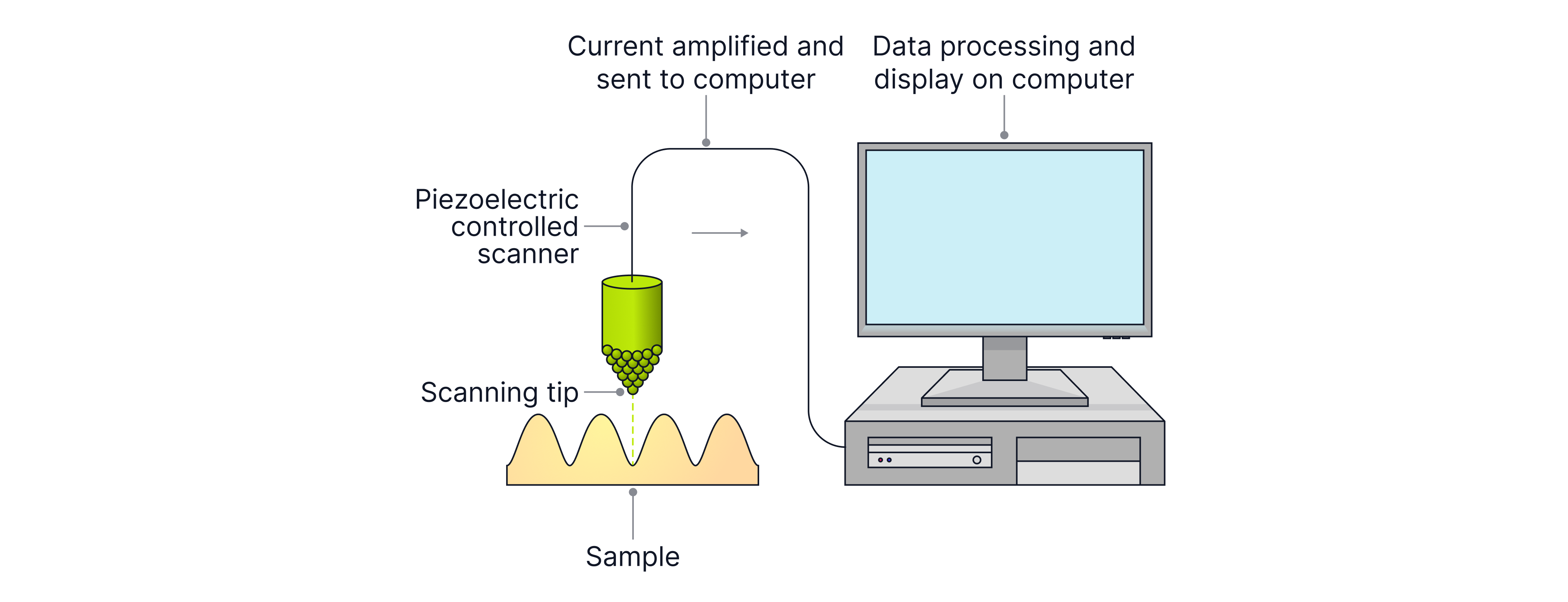
A scanning tunnelling microscope uses an extremely sharp conducting tip brought very close to the sample (around separation). A small p.d. between tip and sample allows electrons to tunnel, producing a tiny tunnelling current.
Key idea: tunnelling probability (and current) is extremely sensitive to the gap: smaller gap → larger current; larger gap → smaller current.
A piezoelectric transducer controls the tip position with very high precision.
Constant height mode
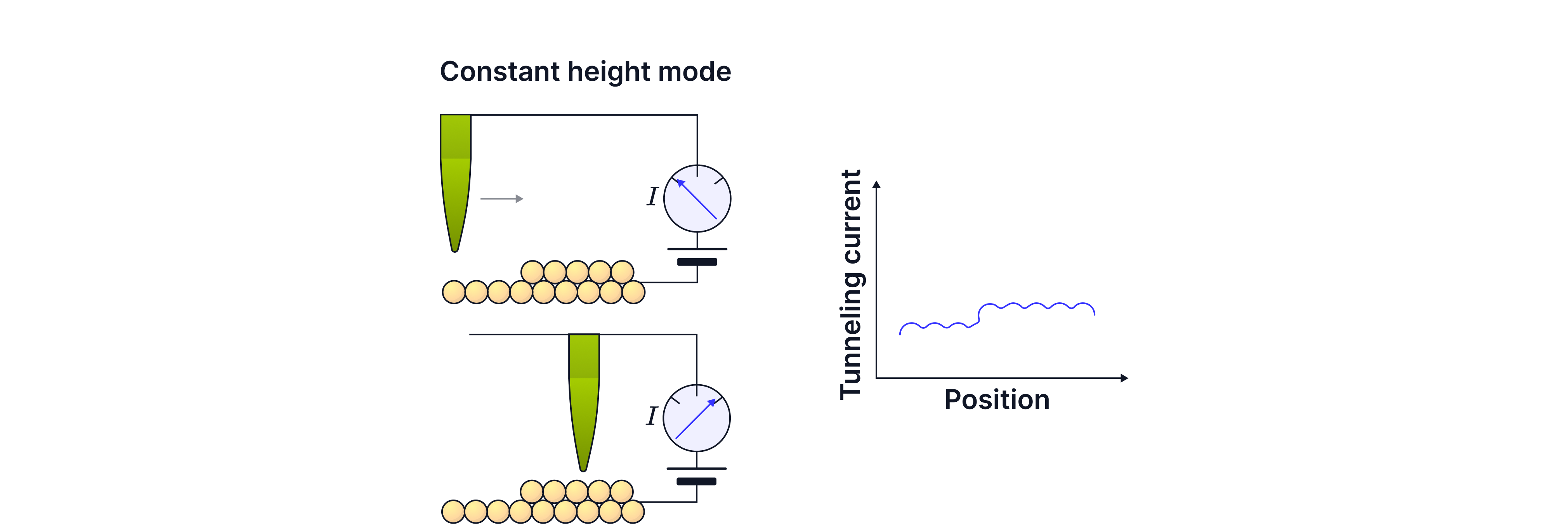
The tip height stays (approximately) constant while scanning. Changes in surface height change the gap so the tunnelling current changes. The STM records current vs position to map the surface.
Constant current mode
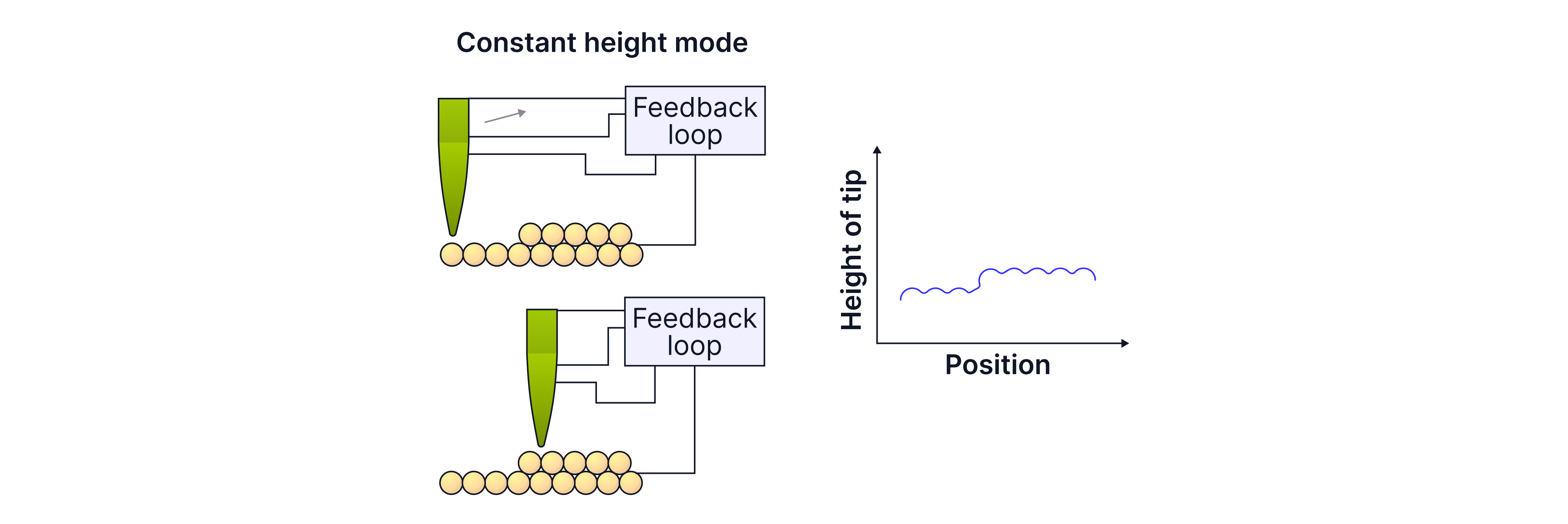
A feedback loop adjusts the tip height continuously to keep the current constant. The STM records tip height vs position, producing a surface profile.
Worked Example:
Compare constant height mode and constant current mode in an STM. State one similarity and two differences.
Answer:
Similarity: both use tunnelling current between a sharp tip and the surface; current depends strongly on tip–surface separation.
Constant height: tip height fixed; current varies with position and is recorded to map the surface.
Constant current: feedback adjusts tip height to keep current constant; height varies with position and is recorded.
Practice Questions
How can we see things that are closer together than the wavelength of light?
-> Check out Hannah's video explanation for more help.
Answer:
Electrons have a de Broglie wavelength . When accelerated through a large p.d., their momentum is large, so their is much smaller than visible light. Smaller wavelength allows finer detail to be resolved. In practice, resolution is limited by lens aberrations and sample effects.
What is quantum tunnelling and why is it useful?
-> Check out Hannah's video explanation for more help.
Answer:
With the tip about from the surface, electrons have a finite probability of tunnelling across the gap due to their wave nature. A small p.d. drives electrons across in one direction, producing a tunnelling current. In constant height mode the tip height is kept constant; changes in surface height change the gap width, which changes tunnelling probability and therefore the current. Recording current versus position produces a surface map.